Abstract
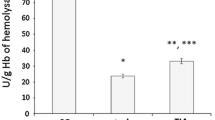
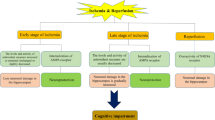
Free radicals play an important role in the pathogenesis of brain injury. This study evaluates the potential relationship between ischaemia/reperfusion (I/R)-induced brain injury, peripheral oxidative stress (lymphocyte DNA damage), plasma antioxidant potential and uric acid levels. We observed that 15 min of ischaemia were sufficient to significantly increase lymphocyte DNA damage that remained elevated at the end of early (3 h) reperfusion and at later (72 h) reperfusion time; this parameter was not significantly increased, when compared to preoperated levels. In parallel, antioxidant potential was elevated after 15 min of ischaemia, remained high at early (3 h) reperfusion and decreased again with longer (72 h) reperfusion. A close association between the plasma antioxidant status and the uric acid content has been confirmed by findings that changes in TRAP values positively correlate with uric acid concentration in rat plasma after ischaemic injury. Moreover, results of in vitro experiments with extra uric acid addition to control plasma have shown that uric acid contributes to a greater part of TRAP values. These results indicate a similar time course of brain I/R-associated oxidative stress and peripheral antioxidant defence status and/or oxidative stress in animal experiments.




Similar content being viewed by others
References
Aabdallah DM, Eid NI (2004) Possible neuroprotective effects of lecithin and alpha-tocopherol alone or in combination against ischemia/reperfusion insult in rat brain. J Biochem Mol Toxicol 18:273–278
Ahmad S, Yousuf S, Ishrat T, Khan MB, Bhatia K, Fazli IS, Khan JS, Ansari NH, Islam F (2006) Effect of dietary sesame oil as antioxidant on brain hippocampus of rat in focal cerebral ischemia. Life Sci 79:921–1928
Becker BF (1993) Towards the physiological function of uric acid. Free Radic Biol Med 14:615–631
Beckman JS, Beckman TW, Chen J, Marshall PA, Freeman BA (1990) Apparent hydroxyl radical production by peroxynitrite: implications for endothelial injury from nitric oxide and superoxide. Proc Natl Acad Sci USA 87:1620–1624
Burda J, Danielisova V, Nemethova M, Gottlieb M, Matiasova M, Domorakova I, Mechirova E, Ferikova M, Salinas M, Burda R (2006) Delayed postconditioning initiates additive mechanism necessary for survival of selectively vulnerable neurons after transient ischemia in rat brain. Cell Mol Neurobiol 26:1141–1151
Collins AR (2005) Assays for oxidative stress and antioxidant status: applications to research into the biological effectiveness of polyphenols. Am J Clin Nutr 81:261–267
Collins AR, Dusinska M, Franklin M, Somorovska M, Petrovska H, Duthie S, Fillion L, Panayiotidis M, Raslova K, Vaughan N (1997) Comet assay in human biomonitoring studies: reliability, validation, and applications. Environ Mol Mutagen 30:139–146
Collins AR, Dusinska M, Gedik CM, Stetina R (1996) Oxidative damage to DNA: do we have a reliable biomarker? Environ Health Perspect 104:465–469
Chamorro A, Obach V, Cervera A, Revilla M, Deulofeu R, Aponte JH (2002) Prognostic significance of uric acid serum concentration in patients with acute ischemic stroke. Stroke 33:1048–1052
Chen J, Jin K, Chen M, Pei W, Kawaguchi K, Greenberg DA, Simon RP (1997) Early detection of DNA strand breaks in the brain after transient focal ischemia: implications for the role of DNA damage in apoptosis and neuronal cell death. J Neurochem 69:232–245
Cherubini A, Polidori MC, Bregnocchi M, Pezzuto S, Cecchetti R, Ingegni T, di Iorio A, Senin U, Mecocci P (2000) Antioxidant profile and early outcome in stroke patients. Stroke 31:2295–2300
Crack PJ, Taylor JM (2005) Reactive oxygen species and the modulation of stroke. Free Radic Biol Med 38:1433–1444
Danielisova V, Nemethova M, Gottlieb M, Burda J (2006) The changes in endogenous antioxidant enzyme activity after postconditioning. Cell Mol Neurobiol 26:1181–1191
Enrico P, Esposito G, Mura MA, Migheli R, Serra PA, Desole MS, Miele E, De Natale G, Miele M (1997) Effects of allopurinol on striatal dopamine, ascorbate and uric acid during an acute morphine challenge: ex vivo and in vivo studies. Pharmacol Res 35:577–585
ESCODD (2005) Establishing the background level of base oxidation in human lymphocyte DNA: results of an interlaboratory validation study. FASEB J 19:82–84
Fiskum G, Rosenthal RE, Vereczki V, Martin E, Hoffman GE, Chinopoulos C, Kowaltowski A (2004) Protection against ischemic brain injury by inhibition of mitochondrial oxidative stress. J Bioenerg Biomembr 36:347–352
Fracasso ME, Doria D, Franceschetti P, Perbellini L, Romeo L (2006) DNA damage and repair capacity by comet assay in lymphocytes of white-collar active smokers and passive smokers (non- and ex-smokers) at workplace. Toxicol Lett 167:131–141
Frassetto SS, Schetinger MR, Webber A, Sarkis JJ, Netto CA (1999) Ischemic preconditioning reduces peripheral oxidative damage associated with brain ischemia in rats. Braz J Med Biol Res 32:1295–1302
Gariballa SE, Hutchin TP, Sinclair AJ (2002) Antioxidant capacity after acute ischaemic stroke. QJM 95:685–690
Gerber Y, Tanne D, Medalie JH, Goldbourt U (2006) Serum uric acid and long-term mortality from stroke, coronary heart disease and all causes. Eur J Cardiovasc Prev Rehabil 13:193–198
Ghiselli A, Serafini M, Maiani G, Azzini E, Ferro-Luzzi A (1995) A fluorescence-based method for measuring total plasma antioxidant capability. Free Radic Biol Med 18:29–36
Giovannelli L, Cozzi A, Guarnieri I, Dolara P, Moroni F (2002) Comet assay as a novel approach for studying DNA damage in focal cerebral ischemia: differential effects of NMDA receptor antagonists and poly(ADP-ribose) polymerase inhibitors. J Cereb Blood Flow Metab 22:697–704
Glantz L, Avramovich A, Trembovler V, Gurvitz V, Kohen R, Eidelman LA, Shohami E (2005) Ischemic preconditioning increases antioxidants in the brain and peripheral organs after cerebral ischemia. Exp Neurol 192:117–124
Glantzounis GK, Tsimoyiannis EC, Kappas AM, Galaris DA (2005) Uric acid and oxidative stress. Curr Pharm Des 11:4145–4151
Green AR, Ashwood TF (2005) Free radical trapping as a therapeutic approach to neuroprotection in stroke: experimental and clinical studies with NXY-059 and free radical scavengers. Curr Drug Targets CNS Neurol Disord 4:109–118
Heiss WD (2002) Stroke-acute interventions. J Neural Transm 63:37–57
Homi HM, Freitas JJ, Curi R, Velasco IT, Junior BA (2002) Changes in superoxide dismutase and catalase activities of rat brain regions during early global transient ischemia/reperfusion. Neurosci Lett 333:37–40
Hosseinzadeh H, Sadeghnia HR (2005) Safranal, a constituent of Crocus sativus (saffron), attenuated cerebral ischemia induced oxidative damage in rat hippocampus. J Pharm Pharm Sci 8:394–399
Kolesarova M, Pavel J, Lukacova N, Kolesar D, Marsala J (2006) Effect of ischemia in vivo and oxygen-glucose deprivation in vitro on NOS pools in the spinal cord: comparative study. Cell Mol Neurobiol 26:1281–1294
Lehotsky J, Murin R, Strapkova A, Urikova A, Tatarkova Z, Kaplan P (2004) Time course of ischemia/reperfusion-induced oxidative modification of neural proteins in rat forebrain. Gen Physiol Biophys 23:401–415
Liu H, Uno M, Kitazato KT, Suzue A, Manabe S, Yamasaki H, Shono M, Nagahiro S (2004) Peripheral oxidative biomarkers constitute a valuable indicator of the severity of oxidative brain damage in acute cerebral infarction. Brain Res 1025:43–50
Margaill I, Plotkine M, Lerouet D (2005) Antioxidant strategies in the treatment of stroke. Free Radic Biol Med 39:429–443
Moor E, Kohen R, Reiter RJ, Shohami E (2001) Closed head injury increases extracellular levels of antioxidants in rat hippocampus in vivo: an adaptive mechanism? Neurosci Lett 316:169–172
Nagayama T, Lan J, Henshall DC, Chen D, O’Horo C, Simon RP, Chen J (2000) Induction of oxidative DNA damage in the peri-infarct region after permanent focal cerebral ischemia. J Neurochem 75:1716–1728
Nihei H, Kanemitsu H, Tamura A, Oka H, Sano K (1989) Cerebral uric acid, xanthine, and hypoxanthine after ischemia: the effect of allopurinol. Neurosurgery 25:6173–6177
Nita DA, Nita V, Spulber S, Moldovan M, Popa DP, Zagrean AM, Zagrean L (2001) Oxidative damage following cerebral ischemia depends on reperfusion-a biochemical study in rat. J Cell Mol Med 5:163–170
Pulsinelli WA, Brierley JB (1979) A new model of bilateral hemispheric ischemia in the unanesthetized rat. Stroke 10:268–272
Repine JE, Fox RB, Berger EM (1981) Hydrogen peroxide kills Staphylococcus aureus by reacting with staphylococcal iron to form hydroxyl radical. J Biol Chem 256:7094–7096
Ryan M, Grayson L, Clarke DJ (1997) The total antioxidant capacity of human serum measured using enhanced chemiluminescence is almost completely accounted for by urate. Ann Clin Biochem 34:688–689
Romanos E, Planas AM, Amaro S, Chamorro A (2007) Uric acid reduces brain damage and improves the benefits of rt-PA in a rat model of thromboembolic stroke. J Cereb Blood Flow Metab 27:14–20
Shi H, Liu KJ (2007) Cerebral tissue oxygenation and oxidative brain injury during ischemia and reperfusion. Front Biosci 12:1318–1328
Sies H (1993) Strategies of antioxidant defense. Eur J Biochem 215:213–219
Slavikova H, Lojek A, Hamar J, Duskova M, Kubala L, Vondracek J, Ciz M (1998) Total antioxidant capacity of serum increased in early but not late period after intestinal ischemia in rats. Free Radic Biol Med 25:9–18
Urikova A, Babusikova E, Dobrota D, Drgova A, Kaplan P, Tatarkova Z, Lehotsky J (2006) Impact of Ginkgo biloba extract EGb 761 on ischemia/reperfusion—induced oxidative stress products formation in rat forebrain. Cell Mol Neurobiol 26:1343–1353
Valko M, Leibfritz D, Moncol J, Cronin MT, Mazur M, Telser J (2007) Free radicals and antioxidants in normal physiological functions and human disease. Int J Biochem Cell Biol 39:44–84
Waring WS, Webb DJ, Maxwell SR (2001) Systemic uric acid administration increases serum antioxidant capacity in healthy volunteers. J Cardiovasc Pharmacol 38:365–371
Yu ZF, Bruce-Keller AJ, Goodman Y, Mattson MP (1998) Uric acid protects neurons against excitotoxic and metabolic insults in cell culture, and against focal ischemic brain injury in vivo. J Neurosci Res 53:613–625
Acknowledgements
This work was supported by Grants VEGA 3380/06, UK/38/2005, MVTS 39, MVTS-COST B30, APVV 51-027404, from the Ministry of Education and Science of the Slovak Republic.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sivoňová, M., Kaplán, P., Ďuračková, Z. et al. Time Course of Peripheral Oxidative Stress as Consequence of Global Ischaemic Brain Injury in Rats. Cell Mol Neurobiol 28, 431–441 (2008). https://doi.org/10.1007/s10571-007-9246-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-007-9246-x