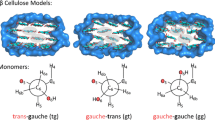
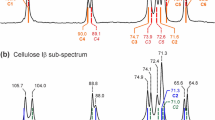
Abstract
Plane wave and molecular orbital density functional theory calculations with periodic models and oligomer fragments based on Iβ cellulose showed relationships among hydrogen bond (H-bond) lengths, angles, energies, and vibrational frequencies. Significantly, the 13C NMR chemical shifts (δ13C), glycosidic and hydroxymethyl torsion angles, H-bond vibrational frequencies, and H-bond geometries results all suggest the predominance of the crystallographic structure C rather than structure A of Iβ cellulose as reported by Nishiyama et al. (J Am Chem Soc 124(31):9074–9082, 2002). The results reported herein also clarified that the δ13C and δ13C′ data from Erata et al. (Cellul Commun 4:128–131, 1997) correspond to δ13C from the origin and center chains of cellulose, respectively. Moreover, this work discusses the use of cellulose oligomer fragments for their potential use in understanding cellulose assembly.










Similar content being viewed by others
References
Adamo C, Barone V, Introduction I (1998) Exchange functionals with improved long-range behavior and adiabatic connection methods without adjustable parameters: the mPW and mPW1PW models. J Chem Phys 108:664–675
Atalla RH (1999) Celluloses. In: Meth-Cohn O, Barton D, Nakanishi K (eds) Comprehensive natural products chemistry. Elsevier, New York, pp 529–598
Blackwell J (1977) Infrared and Raman spectroscopy of cellulose. In: Arthur J (ed) Cellulose chemistry and technology: ACS symposium series. ACS Publications, Washington, DC, pp 206–218
Blackwell J, Vasko PD, Koenig JL (1970) Infrared and Raman spectra of the cellulose from the cell wall of Valonia ventricosa. J Appl Phys 41:4375–4379
Boys SF, Bernardi F (2002) The calculation of small molecular interactions by the differences of separate total energies. Some procedures with reduced errors. Mol Phys 100:65–73
Bućko T, Hafner J (2005) No geometry optimization of periodic systems using internal coordinates. J Chem Phys 122:124508–124517
Buhl M, Kaupp M, Malkina OL, Malkin VG (1999) The DFT route to NMR chemical shifts. J Comput Chem 20:91–105
Cancès E, Mennucci B, Tomasi J (1997) A new integral equation formalism for the polarizable continuum model: theoretical background and applications to isotropic and anisotropic dielectrics. J Chem Phys 107:3032–3041
Cheeseman JR, Trucks GW, Keith TA, Frisch MJ (1996) A comparison of models for calculating nuclear magnetic resonance shielding tensors. J Chem Phys 104:5497–5509
Clark T, Chandrasekhar J, Spitznagel GW, Schleyer PVR (1983) Efficient diffuse function-augmented basis sets for anion calculations. III. The 3-21 + G basis set for first-row elements, Li–F. J Comput Chem 4:294–301
Cosgrove DJ (2005) Growth of the plant cell wall. Nat Rev Mol Cell Biol 6:850–861
Cremer D, Pople J (1975) A general definition of ring puckering coordinates. J Am Chem Soc 97:1354–1358
Davidson TC, Newman RH, Ryan MJ (2004) Variations in the fibre repeat between samples of cellulose I from different sources. Carbohydr Res 339:2889–2893
Erata T, Shikano T, Yunoki S, Takai M (1997) The complete assignment of the 13C CP/MAS NMR spectrum of native cellulose by using 13C labeled glucose. Cellul Commun 4:128–131
Fernandes AN, Thomas LH, Altaner CM, Callow P, Forsyth VT, Apperely DC, Kennedy CJ, Jarvis MC (2011) Nanostructure of cellulose microfibrils in spruce wood. Proc Natl Acad Sci USA 108:E1195–E1203
Fiadzomor PAY, Keen AM, Grant RB, Orr-Ewing AJ (2008) Interaction energy of water dimers from pressure broadening of near-IR absorption lines. Chem Phys Lett 462:188–191
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Montgomery Jr JA, Vreven T Kudin KN, Burant JC et al (2009) Gaussian 09 Revision B.01. Wallingford, CT
Gallina ME, Sassi P, Paolantoni M, Morresi A, Cataliotti RS (2006) Vibrational analysis of molecular interactions in aqueous glucose solutions. Temperature and concentration effects. J Phys Chem B 110:8856–8864
Glendening ED, Weinhold F (1997) Natural resonance theory: II. Natural bond order and valency. J Comput Chem 19:610–627
Glendening ED, Weinhold F (1998) Natural resonance theory: II. Natural bond order and valency. J Comput Chem 19:610–627
Gogonea V (1998) Self-consistent reaction field methods: cavities. In: von Rague Schleyer P, Allinger NL, Clark T, Gastiger J, Kollman PA, Schaefer HF, Schreiner PR (eds) Encyclopedia of computational chemistry. Wiley, New York, NY, pp 2560–2574
Gonzalez-Outeiriño J, Kirschner KN, Thobhani S, Woods RJ (2006) Reconciling solvent effects on rotamer populations in carbohydrates: a joint MD and NMR analysis. Can J Chem 84:569–579
Gottlieb HE, Kotlyar V, Nudelman A (1997) NMR chemical shifts of common laboratory solvents as trace impurities. J Org Chem 62:7512–7515
Grimme S (2006) Semiempirical GGA-type density functional constructed with a long-range dispersion correction. J Comput Chem 27:1787–1799
Guvench O, Hatcher ER, Venable RM, Pastor RW, MacKerell AD Jr (2009) CHARMM additive all-atom force field for glycosidic linkages. J Chem Theory Comput 5:2353–2370
Harris DM, Corbin K, Wang T, Gutierrez R, Bertolo AL, Petti C, Smilgies D-M, Estevez JM, Bonetta D, Urbanowicz BR et al (2012) Cellulose microfibril crystallinity is reduced by mutating C-terminal transmembrane region residues CESA1A903 V and CESA3T942I of cellulose synthase. Proc Natl Acad Sci USA 109:4098–4103
Himmel ME, Di S-Y, Johnson DK, Adney WS, Nimlos MR, Brady JW, Foust TD (2007) Biomass recalcitrance: engineering plants and enzymes for biofuels. Science 315(80):804–807
Hohenberg P, Kohn W (1964) Inhomgeneous electron gas. Phys Rev 136:B864–B871
Jarvis M (2003) Cellulose stacks up. Nature 426:1–2
Jeffrey GA (1997) An introduction to hydrogen bonding. Oxford University Press, New York
Karadakov PB (2006) Ab initio calculation of NMR shielding constants. In: Webb GA (ed) Modern magnetic resonance. Springer, Netherlands, pp 63–70
Kirschner KN, Woods RJ (2001) Solvent interactions determine carbohydrate conformation. Proc Natl Acad Sci USA 98:10541–10545
Kohn W, Sham LJ (1965) Self-consistent equations including exchange and correlation effects. Phys Rev 140:A1133–A1138
Kresse G, Furthmüller J (1996) Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys Rev B 54:11169–11186
Krishnan R, Brinkley JS, Seeger R, Pople JA (1980) Self-consistent molecular orbital methods. XX. A basis set for correlated wave functions. J Chem Phys 72:650–654
Kubicki JD, Mohamed MN-A, Watts HD (2013) Quantum mechanical modeling of the structures, energetics and spectral properties of Iα and Iβ cellulose. Cellulose 20:9–23
Li Y, Lin M, Davenport JW (2011) Ab initio studies of cellulose I: crystal structure, intermolecular forces, and interactions with water. J Phys Chem C 115:11533–11539
Malm, E., Bulone, V., Wickholm, K., Larsson, P., and Iversen, T. (2010). The surface structure of well-ordered native cellulose fibrils in contact with water. Carbohydr. Res. 345
McNamara JP, Hillier IH (2007) Semi-empirical molecular orbital methods including dispersion corrections for the accurate prediction of the full range of intermolecular interactions in biomolecules. Phys Chem Chem Phys 9:2362–2370
McQueen-Mason S, Cosgrove DJ (1994) Disruption of hydrogen bonding between plant cell wall polymers by proteins that induce wall extension. Proc Natl Acad Sci USA 91:6574–6578
Newman RH, Davidson TC (2004) Molecular conformations at the cellulose:water interface. Cellulose 11:23–32
Nishiyama Y, Langan P, Chanzy H (2002) Crystal structure and hydrogen-bonding system in cellulose Iβ from synchrotron X-ray and neutron fiber diffraction. J Am Chem Soc 124:9074–9082
Nishiyama Y, Johnson GP, French AD, Forsyth VT, Langan P (2008) Neutron crystallography, molecular dynamics, and quantum mechanics studies of the nature of hydrogen bonding in cellulose Iβ. Biomacromolecules 9:3133–3140
Panduranga S, Chamundeeswari V, Rajan E, Jebaseelan J, Sundaraganesan N (2011) Theoretical and experimental studies on 2-(2-methyl-5-nitro-1-imidazoyl)ethanol. Eur J Chem 2:136–145
Papajak E, Zheng J, Xu X, Leverentz HR, Truhlar DG (2011) Perspectives on basis sets beautiful: seasonal plantings of diffuse basis functions. J Chem Theory Comput 7:3027–3034
Raman EP, Guvench O, MacKerell AD (2010) CHARMM additive all-atom force field for glycosidic linkages in carbohydrates involving furanoses. J Phys Chem B 114:12981–12994
Sarotti AM, Pellegrinet SC (2009) A multi-standard approach for GIAO (13)C NMR calculations. J Org Chem 74:7254–7260
Schaftenaar G, Noordik JH (2000) Molden: a pre- and post-processing program for molecular and electronic structures. J Comput Aided Mol Des 14:123–134
Schreckenbach G, Ziegler T (1995) Calculation of NMR shielding tensors using gauge-including atomic orbitals and modern density functional theory. J Phys Chem 99:606–611
Shen T, Gnanakaran S (2009) The stability of cellulose: a statistical perspective from a coarse-grained model of hydrogen-bond networks. Biophys J 96:3032–3040
Sternberg U, Koch F, Prieß W, Witter R (2003) Crystal structure refinements of cellulose polymorphs using solid state 13C chemical shifts. Cellulose 10:189–199
Watts HD, Mohamed MNA, Kubicki JD (2011) Comparison of multistandard and TMS-standard calculated NMR shifts for coniferyl alcohol and application of the multistandard method to lignin dimers. J Phys Chem B 115:1958–1970
Wickholm K, Larsson PT, Iversen T (1998) Assignment of non-crystalline forms in cellulose I by CP/MAS 13C NMR spectroscopy. Carbohydr Res 312:123–129
Wolinski K, Hinton JF, Pulay P (1990) Efficient implementation of the gauge-independent atomic orbital method for NMR chemical shift calculations. J Am Chem Soc 112:8251–8260
Zhao Y, Schultz NE, Truhlar DG (2006) Design of density functionals by combining the method of constraint satisfaction with parametrization for thermochemistry, thermochemical kinetics, and noncovalent interactions. J Chem Theory Comput 2:364–382
Zhao H, Kwak JH, Zhang ZC, Brown HM, Arey BW, Holladay JE (2007) Studying cellulose fiber structure by SEM, XRD, NMR and acid hydrolysis. Carbohydr Polym 68:235–241
Zhao Z, Shklyaev OE, Nili A, Mohamed MNA, Kubicki JD, Crespi VH, Zhong L (2013) Cellulose microfibril twist, mechanics, and implication for cellulose biosynthesis. J Phys Chem A 117:2580–2589
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Watts, H.D., Mohamed, M.N.A. & Kubicki, J.D. A DFT study of vibrational frequencies and 13C NMR chemical shifts of model cellulosic fragments as a function of size. Cellulose 21, 53–70 (2014). https://doi.org/10.1007/s10570-013-0128-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-013-0128-8