Abstract
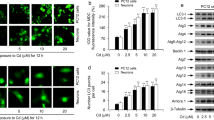
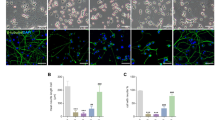
Dopamine oxidation and divalent cations have been reported to induce neuronal cell death. Although autophagy is involved in neuronal cell death, it has also been suggested to facilitate cell survival. We sought to investigate the role of autophagy in PC12 cells and cultured neurons treated with dopamine and Zn2+. Cells expressing EGFP-LC3 were treated with high concentrations of dopamine and Zn2+, and the formation of EGFP-LC3 fluorescence aggregates was monitored. Our results showed a significant increase in the number of fluorescent puncta in the cytosol of PC12 cells treated with these chemicals. These treatments enhanced LC3 lipidation levels in PC12 cells. Decreasing the ATG7 protein level using specific small interference RNA (siRNA) and pretreating with phosphatidylinositol 3-phosphate kinase blockers, wortmannin and LY294002, inhibited puncta formation. Dopamine or Zn2+ treatment significantly elevated the intracellular Zn2+ concentration ([Zn2+] i ); however, inhibiting the [Zn2+] i elevation in dopamine-treated cells suppressed the puncta formation. LY294002 or siRNA-directed members of the autophagy pathway increased the fraction of phosphatidylserine present on the outer membrane leaflet in PC12 cells treated with dopamine or Zn2+, suggesting an increase in apoptosis. Primary embryonic midbrain neurons expressing EGFP-LC3 also displayed a significant increase in the number of fluorescent aggregates in cells upon treatment with dopamine or Zn2+. Dopamine or Zn2+ treatment significantly elevated the [Zn2+] i in neurons and caused neuronal death. Our results indicate that treating cells with dopamine and Zn2+ results in the activation of the autophagy pathway in an effort to enhance cell survival.







Similar content being viewed by others
Abbreviations
- Annexin V-FITC:
-
Annexin V-fluorescein isothiocyanate
- Atg8-PE:
-
Atg8-phosphatidylethanolamine
- BafA1:
-
Bafilomycin A1
- DAPI:
-
4′-6-Diamidino-2-phenylindole
- HBSS:
-
Hank’s buffered salt solution
- MTT:
-
3-(4,5-cimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide
- PAS:
-
Pre-autophagosomal structure
- PBS:
-
Phosphate-buffered saline
- PD:
-
Parkinson’s disease
- PI:
-
Propidium iodide
- PI3K:
-
Phosphatidylinositol 3-phosphate kinase
- PS:
-
Phosphatidylserine
- TPEN:
-
N,N,N′,N′-tetrakis-(2-pyridylmethyl)-thylenediamine
References
Ahmadi FA, Grammatopoulos TN, Poczobutt AM, Jones SM, Snell LD, Das M, et al. Dopamine selectively sensitizes dopaminergic neurons to rotenone-induced apoptosis. Neurochem Res. 2008;33:886–901.
Ahn YH, Kim YH, Hong SH, Koh JY. Depletion of intracellular zinc induces protein synthesis-dependent neuronal apoptosis in mouse cortical culture. Exp Neurol. 1998;154:47–56.
Baehrecke EH. Autophagy: dual roles in life and death? Nat Rev Mol Cell Biol. 2005;6:505–10.
Bains M, Florez-McClure ML, Heidenreich KA. Insulin-like growth factor-I prevents the accumulation of autophagic vesicles and cell death in Purkinje neurons by increasing the rate of autophagosome-to-lysosome fusion and degradation. J Biol Chem. 2009;284:20398–407.
Barnham KJ, Bush AI. Metals in Alzheimer’s and Parkinson’s diseases. Curr Opin Chem Biol. 2008;12:222–8.
Budde T, Minta A, White JA, Kay AR. Imaging free zinc in synaptic terminals in live hippocampal slices. Neuroscience. 1997;79:347–58.
Chang CL, Liao JJ, Huang WP, Lee H. Lysophosphatidic acid inhibits serum deprivation-induced autophagy in human prostate cancer PC-3 cells. Autophagy. 2007;3:268–70.
Chinta SJ, Mallajosyula JK, Rane A, Andersen JK. Mitochondrial alpha-synuclein accumulation impairs complex I function in dopaminergic neurons and results in increased mitophagy in vivo. Neurosci Lett. 2010;486:235–9.
Cuervo AM, Stefanis L, Fredenburg R, Lansbury PT, Sulzer D. Impaired degradation of mutant alpha-synuclein by chaperone-mediated autophagy. Science. 2004;305:1292–5.
Dexter DT, Wells FR, Lees AJ, Agid F, Agid Y, Jenner P, et al. Increased nigral iron content and alterations in other metal ions occurring in brain in Parkinson’s disease. J Neurochem. 1989;52:1830–6.
Eibl JK, Abdallah Z, Ross GM. Zinc-metallothionein: a potential mediator of antioxidant defence mechanisms in response to dopamine-induced stress. Can J Physiol Pharmacol. 2010;88:305–12.
Eller M, Williams DR. alpha-Synuclein in Parkinson disease and other neurodegenerative disorders. Clin Chem Lab Med. 2011;49:403–8.
Gimenez-Xavier P, Francisco R, Santidrian AF, Gil J, Ambrosio S. Effects of dopamine on LC3-II activation as a marker of autophagy in a neuroblastoma cell model. Neurotoxicology. 2009;30:658–65.
Gonzalez-Hernandez T, Barroso-Chinea P, De La Cruz MI, Del Mar Perez-Delgado M, Rodriguez M. Expression of dopamine and vesicular monoamine transporters and differential vulnerability of mesostriatal dopaminergic neurons. J Comp Neurol. 2004;479:198–215.
Haque ME, Asanuma M, Higashi Y, Miyazaki I, Tanaka K, Ogawa N. Overexpression of Cu-Zn superoxide dismutase protects neuroblastoma cells against dopamine cytotoxicity accompanied by increase in their glutathione level. Neurosci Res. 2003;47:31–7.
Jana S, Sinha M, Chanda D, Roy T, Banerjee K, Munshi S, et al. Mitochondrial dysfunction mediated by quinone oxidation products of dopamine: implications in dopamine cytotoxicity and pathogenesis of Parkinson’s disease. Biochim Biophys Acta. 2011;1812:663–73.
Kabeya Y, Mizushima N, Ueno T, Yamamoto A, Kirisako T, Noda T, et al. LC3, a mammalian homologue of yeast Apg8p, is localized in autophagosome membranes after processing. EMBO J. 2000;19:5720–8.
Klionsky DJ, Abdalla FC, Abeliovich H, Abraham RT, Acevedo-Arozena A, Adeli K, et al. Guidelines for the use and interpretation of assays for monitoring autophagy. Autophagy. 2012;8:445–544.
Lee SJ, Koh JY. Roles of zinc and metallothionein-3 in oxidative stress-induced lysosomal dysfunction, cell death, and autophagy in neurons and astrocytes. Mol Brain. 2010;3:30.
Lo HS, Chiang HC, Lin AM, Chiang HY, Chu YC, Kao LS. Synergistic effects of dopamine and Zn2+ on the induction of PC12 cell death and dopamine depletion in the striatum: possible implication in the pathogenesis of Parkinson’s disease. Neurobiol Dis. 2004;17:54–61.
Lozier ER, Stelmashook EV, Uzbekov RE, Novikova SV, Zorov SD, Alieva IB, et al. Stimulation of kainate toxicity by zinc in cultured cerebellar granule neurons and the role of mitochondria in this process. Toxicol Lett. 2012;208:36–40.
Medvedeva YV, Lin B, Shuttleworth CW, Weiss JH. Intracellular Zn2+ accumulation contributes to synaptic failure, mitochondrial depolarization, and cell death in an acute slice oxygen-glucose deprivation model of ischemia. J Neurosci. 2009;29:1105–14.
Mizuno K, Kurokawa K, Ohkuma S. Regulation of type 1 IP(3) receptor expression by dopamine D2-like receptors via AP-1 and NFATc4 activation. Neuropharmacology. 2013;71:264–72.
Napolitano A, Manini P, d’Ischia M. Oxidation chemistry of catecholamines and neuronal degeneration: an update. Curr Med Chem. 2011;18:1832–45.
Ohsumi Y. Molecular dissection of autophagy: two ubiquitin-like systems. Nat Rev Mol Cell Biol. 2001;2:211–6.
Pasquali L, Ruggieri S, Murri L, Paparelli A, Fornai F. Does autophagy worsen or improve the survival of dopaminergic neurons? Parkinsonism Relat Disord. 2009;15 Suppl 4:S24–7.
Qian L, Flood PM, Hong JS. Neuroinflammation is a key player in Parkinson’s disease and a prime target for therapy. J Neural Transm. 2010;117:971–9.
Sensi SL, Paoletti P, Koh JY, Aizenman E, Bush AI, Hershfinkel M. The neurophysiology and pathology of brain zinc. J Neurosci. 2011;31:16076–85.
Shacka JJ, Klocke BJ, Shibata M, Uchiyama Y, Datta G, Schmidt RE, et al. Bafilomycin A1 inhibits chloroquine-induced death of cerebellar granule neurons. Mol Pharmacol. 2006;69:1125–36.
Sharif R, Thomas P, Zalewski P, Fenech M. The role of zinc in genomic stability. Mutat Res. 2011;16:16.
Stork CJ, Li YV. Zinc release from thapsigargin/IP3-sensitive stores in cultured cortical neurons. J Mol Signal. 2010;5:5.
Strozyk D, Launer LJ, Adlard PA, Cherny RA, Tsatsanis A, Volitakis I, et al. Zinc and copper modulate Alzheimer Abeta levels in human cerebrospinal fluid. Neurobiol Aging. 2009;30:1069–77.
Tarohda T, Ishida Y, Kawai K, Yamamoto M, Amano R. Regional distributions of manganese, iron, copper, and zinc in the brains of 6-hydroxydopamine-induced parkinsonian rats. Anal Bioanal Chem. 2005;383:224–34.
Thompson RB, Whetsell Jr WO, Maliwal BP, Fierke CA, Frederickson CJ. Fluorescence microscopy of stimulated Zn(II) release from organotypic cultures of mammalian hippocampus using a carbonic anhydrase-based biosensor system. J Neurosci Methods. 2000;96:35–45.
Toth K. Zinc in neurotransmission. Annu Rev Nutr. 2011;31:139–53.
Vidailhet M. Movement disorders in 2010: Parkinson disease-symptoms and treatments. Nat Rev Neurol. 2011;7:70–2.
Watabe-Uchida M, Zhu L, Ogawa SK, Vamanrao A, Uchida N. Whole-brain mapping of direct inputs to midbrain dopamine neurons. Neuron. 2012;74:858–73.
Wu MP, Kao LS, Liao HT, Pan CY. Reverse mode Na+/Ca2+ exchangers trigger the release of Ca2+ from intracellular Ca2+ stores in cultured rat embryonic cortical neurons. Brain Res. 2008;1201:41–51.
Wurmser AE, Emr SD. Novel PtdIns(3)P-binding protein Etf1 functions as an effector of the Vps34 PtdIns 3-kinase in autophagy. J Cell Biol. 2002;158:761–72.
Xie Z, Nair U, Klionsky DJ. Atg8 controls phagophore expansion during autophagosome formation. Mol Biol Cell. 2008;19:3290–8.
Yang Z, Klionsky DJ. Mammalian autophagy: core molecular machinery and signaling regulation. Curr Opin Cell Biol. 2010;22:124–31.
Yang JY, Yang WY. Spatiotemporally controlled initiation of Parkin-mediated mitophagy within single cells. Autophagy. 2011;7:1230–8.
Zeng Y, Yang X, Wang J, Fan J, Kong Q, Yu X. Aristolochic acid I induced autophagy extenuates cell apoptosis via ERK 1/2 pathway in renal tubular epithelial cells. PLoS One. 2012;7:e30312.
Zhu L, Ji XJ, Wang HD, Pan H, Chen M, Lu TJ. Zinc neurotoxicity to hippocampal neurons in vitro induces ubiquitin conjugation that requires p38 activation. Brain Res. 2012;1438:1–7.
Acknowledgments
We would like to thank Professor Chun-Jen Chen for his help with the flow cytometry, Professor Hsin-Yu Lee for his help in MTT assay, and Technology Commons, College of Life Science, National Taiwan University for their help in the confocal microscopy. We thank Mr. Chih-Chen Yang and Dr. Yung-Feng Liao (Academia Sinica, Taipei, Taiwan) for their help with the PC12 cell cultures and our time-course experiment. This study was supported by the National Science Council of Taiwan (http://web1.nsc.gov.tw/mp.aspx?mp=7) under grant nos. NSC-99-2628-B-002-054-MY3 and NSC-100-2627-M-002-013.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 917 kb)
Rights and permissions
About this article
Cite this article
Hung, HH., Huang, WP. & Pan, CY. Dopamine- and zinc-induced autophagosome formation facilitates PC12 cell survival. Cell Biol Toxicol 29, 415–429 (2013). https://doi.org/10.1007/s10565-013-9261-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-013-9261-2