Abstract
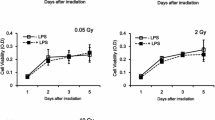
The perception of toxicity to nitric oxide (NO) and irradiation (IR) by three different cell types has been studied. The three cell types are the macrophage like RAW264.7 cells, EL4 lymphoma cells, and splenocytes, which represent the different components of a tumor. These three cell types respond differently to NO donors (SNP and SNAP) and radiation treatment. The macrophages were found to be most radio-resistant and insensitive to NO donors. The innate resistance of the macrophages was not due to its antioxidant defense system since there was no significant activation of the enzymes (superoxide dismutases, catalase, and glutathione peroxidase) in RAW264.7 cells after NO donor and irradiation. But the cell cycle arrest of the three cell types was different from each other. The EL4 cells were found to arrest in the G2/M phase while the macrophages were found arrested in the G1 phase of the cell cycle. Such specific killing of the tumor cell in response to NO donor while sparing the macrophages can be of immense importance to radiotherapy.






Similar content being viewed by others
References
Beckman JS, Crow JP. Pathological implications of nitric oxide, superoxide and peroxynitrite formation. Biochem Soc Trans. 1993;21(2):330–4.
Brockhaus F, Brune B. Overexpression of CuZn superoxide dismutase protects RAW 264.7 macrophages against nitric oxide cytotoxicity. Biochem J. 1999;338(Pt 2):295–303.
Brune B. Nitric oxide: NO apoptosis or turning it ON? Cell Death Differ. 2003;10(8):864–9.
Chang CI, Liao JC, et al. Macrophage arginase promotes tumor cell growth and suppresses nitric oxide-mediated tumor cytotoxicity. Cancer Res. 2001;61(3):1100–6.
Crowell JA, Steele VE, et al. Is inducible nitric oxide synthase a target for chemoprevention? Mol Cancer Ther. 2003;2(8):815–23.
Dewey DL. Effect of oxygen and nitric oxide on the radio-sensitivity of human cells in tissue culture. Nature. 1960;186:780–2.
Epperly MW, Defilippi S, et al. Intratracheal injection of manganese superoxide dismutase (MnSOD) plasmid/liposomes protects normal lung but not orthotopic tumors from irradiation. Gene Ther. 2000;7(12):1011–8.
Griffin RJ, Makepeace CM, et al. Radiosensitization of hypoxic tumor cells in vitro by nitric oxide. Int J Radiat Oncol Biol Phys. 1996;36(2):377–83.
Harris ED. Regulation of antioxidant enzymes. FASEB J. 1992;6(9):2675–83.
Hartwell LH, Kastan MB. Cell cycle control and cancer. Science. 1994;266(5192):1821–8.
Hartwell LH, Weinert TA. Checkpoints: controls that ensure the order of cell cycle events. Science. 1989;246(4930):629–34.
Hirose K, Longo DL, et al. Overexpression of mitochondrial manganese superoxide dismutase promotes the survival of tumor cells exposed to interleukin-1, tumor necrosis factor, selected anticancer drugs, and ionizing radiation. FASEB J. 1993;7(2):361–8.
Hofseth LJ, Hussain SP, et al. Nitric oxide in cancer and chemoprevention. Free Radic Biol Med. 2003;34(8):955–68.
Howard-Flanders P. Effect of nitric oxide on the radiosensitivity of bacteria. Nature. 1957;180(4596):1191–2.
Jansen S, Arning J, et al. S-Nitroso compounds interfere with zinc probing by Zinquin. Anal Biochem. 2004;332(1):145–52.
Jenkins DC, Charles IG, et al. Roles of nitric oxide in tumor growth. Proc Natl Acad Sci USA. 1995;92(10):4392–6.
Kim YM, Talanian RV, et al. Nitric oxide inhibits apoptosis by preventing increases in caspase-3-like activity via two distinct mechanisms. J Biol Chem. 1997;272(49):31138–48.
Kim PK, Zamora R, et al. The regulatory role of nitric oxide in apoptosis. Int Immunopharmacol. 2001;1(8):1421–41.
Kumar KS, Vaishnav YN, et al. Radioprotection by antioxidant enzymes and enzyme mimetics. Pharmacol Ther. 1988;39(1–3):301–9.
Lancaster Jr JR, Xie K. Tumors face NO problems? Cancer Res. 2006;66(13):6459–62.
MacMicking J, Xie QW, et al. Nitric oxide and macrophage function. Annu Rev Immunol. 1997;15:323–50.
Marks GS, McLaughlin BE, et al. Time-dependent increase in nitric oxide formation concurrent with vasodilation induced by sodium nitroprusside, 3-morpholinosydnonimine, and S-nitroso-N-acetylpenicillamine but not by glyceryl trinitrate. Drug Metab Dispos. 1995;23(11):1248–52.
Mitchell JB, DeGraff W, et al. Redox generation of nitric oxide to radiosensitize hypoxic cells. Int J Radiat Oncol Biol Phys. 1998;42(4):795–8.
Narang H, Dhariwala FA, et al. Effect of nitric oxide donor and gamma irradiation on modifications of ERK and JNK in murine peritoneal macrophages. J Cell Commun Signal. 2007;1(3–4):219–26.
Nathan CF, Hibbs Jr JB. Role of nitric oxide synthesis in macrophage antimicrobial activity. Curr Opin Immunol. 1991;3(1):65–70.
O’Connell MJ, Walworth NC, et al. The G2-phase DNA-damage checkpoint. Trends Cell Biol. 2000;10(7):296–303.
Pawlik TM, Keyomarsi K. Role of cell cycle in mediating sensitivity to radiotherapy. Int J Radiat Oncol Biol Phys. 2004;59(4):928–42.
Phoa N, Epe B. Influence of nitric oxide on the generation and repair of oxidative DNA damage in mammalian cells. Carcinogenesis. 2002;23(3):469–75.
Pietarinen P, Raivio K, et al. Catalase and glutathione reductase protection of human alveolar macrophages during oxidant exposure in vitro. Am J Respir Cell Mol Biol. 1995;13(4):434–41.
Schmidtke JR, Dixon FJ. The functional capacity of x-irradiated macrophages. J Immunol. 1972;108(6):1624–30.
St Clair DK, Wan XS, et al. Suppression of radiation-induced neoplastic transformation by overexpression of mitochondrial superoxide dismutase. Mol Carcinog. 1992;6(4):238–42.
Sturgeon CM, Knight ZA, et al. Effect of combined DNA repair inhibition and G2 checkpoint inhibition on cell cycle progression after DNA damage. Mol Cancer Ther. 2006;5(4):885–92.
Sun J, Chen Y, et al. Role of antioxidant enzymes on ionizing radiation resistance. Free Radic Biol Med. 1998;24(4):586–93.
Takahashi S, Kubota Y, et al. The effect of external gamma-irradiation on 59Fe release in vitro from alveolar macrophages previously having ingested 59Fe-iron hydroxide colloid. J Radiat Res (Tokyo). 1990;31(3):263–9.
Thomas DD, Ridnour LA, et al. The chemical biology of nitric oxide: implications in cellular signaling. Free Radic Biol Med. 2008;45(1):18–31.
Warner B, Papes R, et al. Expression of human Mn SOD in Chinese hamster ovary cells confers protection from oxidant injury. Am J Physiol. 1993;264(6 Pt 1):L598–605.
Wassmann S, Wassmann K, et al. Modulation of oxidant and antioxidant enzyme expression and function in vascular cells. Hypertension. 2004;44(4):381–6.
Weinert TA, Hartwell LH. The RAD9 gene controls the cell cycle response to DNA damage in Saccharomyces cerevisiae. Science. 1988;241(4863):317–22.
Xie K, Huang S, et al. Transfection with the inducible nitric oxide synthase gene suppresses tumorigenicity and abrogates metastasis by K-1735 murine melanoma cells. J Exp Med. 1995;181(4):1333–43.
Xu W, Liu LZ, et al. The role of nitric oxide in cancer. Cell Res. 2002;12(5–6):311–20.
Yoshioka Y, Kitao T, et al. Nitric oxide protects macrophages from hydrogen peroxide-induced apoptosis by inducing the formation of catalase. J Immunol. 2006;176(8):4675–81.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dhariwala, F.A., Narang, H. & Krishna, M. Differential response of three cell types to dual stress of nitric oxide and radiation. Cell Biol Toxicol 28, 161–173 (2012). https://doi.org/10.1007/s10565-012-9213-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-012-9213-2