Abstract
Purpose
Low-density lipoprotein cholesterol (LDL-C) recommendations differ between the 2018 American College of Cardiology/American Heart Association (ACC/AHA) and 2019 European Society of Cardiology/European Atherosclerosis Society (ESC/EAS) guidelines for patients with atherosclerotic cardiovascular disease (ASCVD) (< 70 vs. < 55 mg/dl, respectively). In the DA VINCI study, residual cardiovascular risk was predicted in ASCVD patients. The extent to which relative and absolute risk might be lowered by achieving ACC/AHA versus ESC/EAS LDL-C recommended approaches was simulated.
Methods
DA VINCI was a cross-sectional observational study of patients prescribed lipid-lowering therapy (LLT) across 18 European countries. Ten-year cardiovascular risk (CVR) was predicted among ASCVD patients receiving stabilized LLT. For patients with LDL-C ≥ 70 mg/dl, the absolute LDL-C reduction required to achieve an LDL-C of < 70 or < 55 mg/dl (LDL-C of 69 or 54 mg/dl, respectively) was calculated. Relative and absolute risk reductions (RRRs and ARRs) were simulated.
Results
Of the 2039 patients, 61% did not achieve LDL-C < 70 mg/dl. For patients with LDL-C ≥ 70 mg/dl, median (interquartile range) baseline LDL-C and 10-year CVR were 93 (81–115) mg/dl and 32% (25–43%), respectively. Median LDL-C reductions of 24 (12–46) and 39 (27–91) mg/dl were needed to achieve an LDL-C of 69 and 54 mg/dl, respectively. Attaining ACC/AHA or ESC/EAS goals resulted in simulated RRRs of 14% (7–25%) and 22% (15–32%), respectively, and ARRs of 4% (2–7%) and 6% (4–9%), respectively.
Conclusion
In ASCVD patients, achieving ESC/EAS LDL-C goals could result in a 2% additional ARR over 10 years versus the ACC/AHA approach.
Graphical abstract

Similar content being viewed by others
Introduction
Patients with established atherosclerotic cardiovascular disease (ASCVD) are at the highest risk of cardiovascular events, with high low-density lipoprotein cholesterol (LDL-C) levels being one of the major determinants of the magnitude of their residual risk [1, 2]. As such, these individuals benefit from lipid-lowering therapy (LLT) with statins irrespective of the vascular territory affected (e.g., coronary artery disease [CAD], cerebrovascular disease [CBD], or peripheral artery disease [PAD]) [3,4,5,6]. Recent data from large cardiovascular outcomes trials in individuals with ASCVD of add-on LLT (such as proprotein convertase subtilisin/kexin type 9 inhibitors [PCSK9i] and ezetimibe) have demonstrated the benefits of additional lowering of LDL-C levels previously unattainable with statin monotherapy [7,8,9,10,11,12]. These findings have been incorporated into the current guidelines for patients with ASCVD, with the recommendation that LDL-C should be managed more aggressively with combination LLTs if LDL-C levels are not adequately controlled [13, 14]. However, international guidelines differ with respect to these recommendations, with the 2019 European Society of Cardiology/European Atherosclerosis Society (ESC/EAS) recommendations [13] advocating a more stringent LDL-C goal of < 55 mg/dl in ASCVD patients, compared with their 2016 iteration, which had advocated the less stringent goal of < 70 mg/dl [15]. In contrast, the 2018 American College of Cardiology/American Heart Association (ACC/AHA) guidelines [14] recommend aiming for an LDL-C of < 70 mg/dl, using 70 mg/dl as a threshold to guide additional LLT in ASCVD patients at very-high risk.
The recent EU-Wide Cross-Sectional Observational Study of Lipid-Modifying Therapy Use in Secondary and Primary Care (DA VINCI) [16] provides a valuable opportunity to assess contemporary information regarding how LLTs are used in current practice and their impact on LDL-C levels. Although patients being managed for CAD have been studied extensively in bespoke registries, contemporary data for individuals being managed for CBD or PAD are more limited. Through design, approximately four-fifths of the patients with ASCVD in the DA VINCI study [16] were being managed for CBD or PAD, allowing comparisons across the spectrum of ASCVD phenotypes. The present analysis assesses the use of LLT across these populations, the implications of current practice on the future risk of cardiovascular events, and, through a simulation study, to what extent risk might be mitigated if different guidelines were adopted.
Methods
Study Design
The methods and primary results of the DA VINCI study have been described in detail elsewhere [16]. Briefly, DA VINCI was a cross-sectional, observational study of routine clinical management that enrolled 5888 adults (aged ≥ 18 years) in primary and secondary prevention (overall ratio of 1:1) across 18 European countries [16]. Participants must have been prescribed LLT within the 12 months before enrolment and must have had an LDL-C measurement in the 14 months before enrolment. Among secondary prevention participants with established ASCVD, patients being managed for CAD, CBD, or PAD were enrolled in an overall ratio of approximately 1:2:2, respectively. Data were extracted from medical records at a single (enrolment) visit between June 2017 and November 2018. The study protocol (available online at ENCePP; registration no. EUPAS22075 [17]) was approved by the institutional review board or independent ethics committee from each site participating in the study [16].
Aims and Outcomes
In the present analysis, we assessed LLT use and LDL-C control among patients with established ASCVD, stratified by ASCVD type (by definition, all patients were at very high cardiovascular risk according to 2019 ESC/EAS guidelines [13]). Next, we compared the proportion of patients who were above the LDL-C level of 70 mg/dl recommended by 2018 ACC/AHA guidelines for very high-risk ASCVD patients, versus the attainment of the 2019 ESC/EAS goal of < 55 mg/dl. By means of comparison, we additionally explored, as an extension of the attainment of the 2019 ESC/EAS goal, the proportion of patients achieving LDL-C levels < 40 mg/dl. Finally, we evaluated the implications of current practice and LDL-C goal achievement on the future risk of cardiovascular events using the REduction of Atherothrombosis for Continued Health (REACH) equation [18] and simulation methods. The Cholesterol Treatment Trialists' Collaboration meta-analysis has demonstrated that a 1.0 mmol/l reduction in LDL-C levels leads to a 22% relative risk reduction [4]. For this analysis, we modelled what the anticipated potential added benefit on cardiovascular risk reduction would be if patients not on target were to achieve the recommended LDL-C levels according to 2019 ESC/EAS or 2018 ACC/AHA guidelines.
Statistical Analysis
Our analysis included all participants with established ASCVD who were receiving stabilized LLT at LDL-C measurement (defined as no change in dose or regimen for at least 28 days prior to LDL-C measurement). The overall ASCVD population was categorized into individuals with LDL-C levels ≥ 70 mg/dl and those with levels < 70 mg/dl. Results are presented for the overall ASCVD group, and further stratified by ASCVD type, namely, CAD, CBD, and PAD (for participants with known atherosclerotic involvement of more than one vascular bed, categorization was made based on the most recent manifestation of vascular disease at enrolment). Results are reported as mean ± standard deviation or median and interquartile range (IQR) for normally and not normally distributed continuous variables, respectively, and as absolute and relative frequencies (n [%]) for categorical variables.
Calculation of Residual Risk
For each patient, we predicted their (baseline) risk of a subsequent cardiovascular event in the next 10 years (10-year cardiovascular risk) using the REACH equation, based on the individual patient demographics and medical history. The REACH equation predicts the risk of recurrent cardiovascular events and cardiovascular death among outpatients with established ASCVD [18]. The 10-year cardiovascular risk of a subsequent cardiovascular event was calculated by converting the 20-month risk predicted from the REACH equation [18], assuming a constant rate over time (exponential survival function).
For patients with LDL-C levels ≥ 70 mg/dl, we calculated (at an individual level) the absolute and relative reductions in LDL-C required to achieve an LDL-C < 70 mg/dl (2018 ACC/AHA-recommended approach [14]) and LDL-C < 55 mg/dl (2019 ESC/EAS approach), defined for the purposes of this simulation as LDL-C levels of 69 mg/dl and 54 mg/dl, respectively (conservative approach, since the actual LDL-C levels achieved in real life would be expected to be lower if a clinician were to try and lower LDL-C levels to < 70 or < 55 mg/dl, respectively). By means of comparison (as an extension of the attainment of the 2019 ESC/EAS goal), we also explored the reductions in LDL-C required to achieve an LDL-C < 40 mg/dl (defined as 39 mg/dl). In addition, we simulated the relative risk reduction (RRR) by randomly sampling from the inverse probability distribution of the rate ratio per 39 mg/dl from the Cholesterol Treatment Trialists' Collaboration meta-analysis [5]. Finally, we simulated the absolute risk reduction (ARR) and 10-year cardiovascular risk if LDL-C levels of 69, 54, and 39 mg/dl were attained. For comparison, we also assessed the residual risk of cardiovascular events among patients with LDL-C levels < 70 mg/dl.
Results
Patient Characteristics
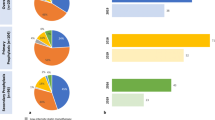
Among participants enrolled in the DA VINCI study, 2039 patients with ASCVD were on stabilized LLT at LDL-C measurement and had data available to allow estimation of risk using the REACH equation; this included 470 (23%), 751 (37%), and 818 (40%) patients being managed for CAD, CBD, and PAD, respectively. Patient characteristics, cardiovascular risk factors, and comorbidities are shown in Table 1, overall and stratified by ASCVD type and by LDL-C < 70 mg/dl or ≥ 70 mg/dl.
LLT Use and LDL-C Levels in Patients with ASCVD
LLT use is shown in Table 2 and consisted mostly of statin monotherapy. Use of moderate-intensity statins as monotherapy ranged from 35% among patients with CAD to 47% in patients with CBD. High-intensity statin monotherapy was used in 44% of patients with CAD and 36% of patients with either CBD or PAD. Ezetimibe was used in combination with statins in 15% of patients with CAD, 7% of patients with CBD, and 8% of patients with PAD. In each group, PCSK9i was used in combination with statins and/or ezetimibe in less than 2% of patients.
The proportion of individuals, overall and within each ASCVD group, who achieved LDL-C levels of < 70 mg/dl and < 55 mg/dl are shown in Fig. 1. Overall, 39% of patients achieved an LDL-C level < 70 mg/dl and 19% achieved an LDL-C level < 55 mg/dl (Fig. 1a). The achievement of an LDL-C level < 70 mg/dl was more likely in patients receiving combination therapy with either ezetimibe or a PCSK9i than in patients on statin monotherapy. Although used infrequently, 58% of patients receiving PCSK9i in combination with other LLTs attained an LDL-C level < 55 mg/dl. The proportion of patients across ASCVD subtypes with LDL-C levels < 70 mg/dl was 44%, 36%, and 40% for the CAD, CBD, and PAD groups, respectively, and 20%, 16%, and 19% for LDL-C levels < 55 mg/dl (Fig. 1b–d).
Achievement of LDL-C levels < 70 mg/dl and < 55 mg/dl in (a) the overall cohort of patients with established ASCVD and stratified by type of ASCVD, namely, (b) coronary artery disease, (c) cerebrovascular disease, and (d) peripheral artery disease. The proportion of individuals within each ASCVD group who achieved LDL-C levels of < 70 mg/dl and < 55 mg/dl are described. Overall, 39% of patients achieved an LDL-C level < 70 mg/dl and 19% of patients achieved an LDL-C level < 55 mg/dl. The achievement of an LDL-C level < 70 mg/dl was more likely in patients receiving combination therapy with either ezetimibe or a PCSK9i than patients on statin monotherapy. ASCVD, atherosclerotic cardiovascular disease; LDL-C, low-density lipoprotein cholesterol; LLT, lipid-lowering therapy; PCSK9i, proprotein convertase subtilisin/kexin type 9 inhibitor
Residual Risk Among Those with LDL-C ≥ 70 mg/dl and < 70 mg/dl
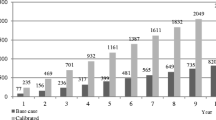
Overall, 61% (1238/2039) of patients had an LDL-C level ≥ 70 mg/dl (Fig. 2), with a median (IQR) LDL-C level of 93 (81–115) mg/dl. The median (IQR) predicted 10-year cardiovascular risk for these patients was 32% (25–43%). Among patients with LDL-C < 70 mg/dl (39% [801/2039]), median (IQR) LDL-C levels were 56 (46–63) mg/dl. Among these patients, the median (IQR) predicted 10-year cardiovascular risk was 35% (26–46%). Demographic characteristics varied between those who had LDL-C levels ≥ 70 mg/dl or < 70 mg/dl, with a slightly higher prevalence of risk factors such as a history of smoking and diabetes in the group with levels < 70 mg/dl (Table 1). The proportion of participants with diabetes was higher in patients with LDL-C < 70 mg/dl (51% of patients with a median LDL-C of 56 [46–63] mg/dl) compared with those with LDL-C levels ≥ 70 mg/dl (39% with a median LDL-C of 93 [81–115] mg/dl) (Table 1).
Simulated risk reduction in patients with established ASCVD. Simulated risk reductions associated with attainment of 2019 ESC/EAS LDL-C goal of < 55 mg/dl and 2018 American College of Cardiology/American Heart Association (ACC/AHA) recommended approach of LDL-C < 70 mg/dl, respectively. Ten-year CV risk was lower with attainment of 2019 ESC/EAS goals (25%) versus the 2018 ACC/AHA approach (28%). LDL-C reductions of 24 (12–46) and 39 (27–91) mg/dl were needed to achieve LDL-C of 69 and 54 mg/dl, respectively. AR, absolute reduction; ASCVD, atherosclerotic cardiovascular disease; ARR, absolute risk reduction; CV, cardiovascular; EAS, European Atherosclerosis Society; ESC, European Society of Cardiology; LDL-C, low-density lipoprotein cholesterol; RR, risk reduction; RRR, relative risk reduction
Simulated Risk Reduction on Achieving an LDL-C < 70 mg/dl
The cohort of patients with LDL-C levels ≥ 70 mg/dl required a median (IQR) 26% (15–40%) reduction (absolute reduction of 24 [12–46] mg/dl) to lower their LDL-C levels to 69 mg/dl (Fig. 2). The median (IQR) simulated RRR and ARR of cardiovascular events were 14% (7–25%) and 4% (2–7%), respectively. The median (IQR) simulated residual 10-year cardiovascular risk was 28% (20–37%).
In the CAD group, patients with LDL-C levels ≥ 70 mg/dl required a median (IQR) 22% (12–35%) reduction (absolute reduction of 19 [9–37] mg/dl) to lower their LDL-C levels to 69 mg/dl (Fig. 3a). The median (IQR) simulated RRR and ARR of cardiovascular events were 11% (6–22%) and 3% (1–6%), respectively. For patients with CAD, attaining an LDL-C level < 70 mg/dl would result in a lower median (IQR) simulated residual 10-year cardiovascular risk of 25% (19–34%).
Simulated risk reduction in the (a) coronary artery disease, (b) cerebrovascular disease, and (c) peripheral artery disease groups. Simulated risk reductions associated with attainment of 2019 ESC/EAS (< 55 mg/dl) and 2018 ACC/AHA (< 70 mg/dl) LDL-C recommendations stratified by ASCVD subtype. Patients being managed for coronary artery disease, cerebrovascular disease, and peripheral artery disease each had similar 10-year CV risk estimates. ACC American College of Cardiology; AHA, American Heart Association; AR, absolute reduction; ARR, absolute risk reduction; CV, cardiovascular; EAS, European Atherosclerosis Society; ESC, European Society of Cardiology; LDL-C, low-density lipoprotein cholesterol; RR, risk reduction; RRR, relative risk reduction
In the CBD group, patients with LDL-C levels ≥ 70 mg/dl required a median (IQR) 25% (15–39%) reduction to lower their LDL-C levels to 69 mg/dl, with a median (IQR) absolute reduction of 23 (12–45) mg/dl (Fig. 3b). The median (IQR) simulated RRR and ARR of cardiovascular events were 14% (7–25%) and 3% (2–7%), respectively. The median (IQR) simulated residual 10-year cardiovascular risk was 26% (30–35%).
In the PAD group, patients with LDL-C levels ≥ 70 mg/dl required a median (IQR) 28% (15–43%) reduction to lower their LDL-C levels to 69 mg/dl, with a median (IQR) absolute reduction of 27 (12–52) mg/dl (Fig. 3c). The median (IQR) simulated RRR and ARR of cardiovascular events were 15% (7–28%) and 4% (2–8%), respectively. The median (IQR) simulated residual 10-year cardiovascular risk was 30% (22–41%).
Simulated Risk Reduction on Achieving an LDL-C < 55 mg/dl
In order for patients with LDL-C levels ≥ 70 mg/dl to reach levels of 54 mg/dl, a median (IQR) 42% (33–53%) reduction in LDL-C was required (absolute reduction of 39 [27–91] mg/dl) (Fig. 2). The median (IQR) simulated RRR and ARR of cardiovascular events were 22% (15–32%) and 6% (4–9%), respectively. The median (IQR) simulated residual 10-year cardiovascular risk was 25% (18–35%).
In the CAD group, patients with LDL-C levels ≥ 70 mg/dl required a median (IQR) 42% (33–53%) reduction (absolute reduction of 39 [31–49] mg/dl) to lower their LDL-C levels to 54 mg/dl (Fig. 3). The median (IQR) simulated RRR and ARR of cardiovascular events were 20% (14–29%) and 5% (3–8%), respectively. The resulting median (IQR) simulated residual 10-year cardiovascular risk was 23% (17–32%).
In the CBD group, those with LDL-C ≥ 70 mg/dl required a median (IQR) 41% (33–53%) reduction (absolute reduction of 38 [27–60] mg/dl) to lower their LDL-C levels to 54 mg/dl (Fig. 3b). The median (IQR) simulated RRR and ARR of cardiovascular events were 22% (15–32%) and 6% (4–9%), respectively. The resulting median (IQR) simulated residual 10-year cardiovascular risk was 24% (18–33%).
In the PAD group, patients with LDL-C levels ≥ 70 mg/dl required a median (IQR) 44% (33–55%) reduction (absolute reduction of 42 [27–67] mg/dl) to lower their LDL-C levels to 54 mg/dl (Fig. 3c). The median (IQR) simulated RRR and ARR of cardiovascular events were 23% (16–34%) and 7% (5–10%), respectively. The resulting median (IQR) simulated residual 10-year cardiovascular risk was 28% (20–38%).
Simulated Risk Reduction on Achieving an LDL-C < 40 mg/dl
In order for patients with LDL-C levels ≥ 70 mg/dl to reach levels of 39 mg/dl, a median (IQR) 58% (52–66%) reduction in LDL-C was required (absolute reduction of 54 [42–76] mg/dl) (Fig. 2). The median (IQR) simulated RRR and ARR of cardiovascular events were 29% (23–28%) and 8% (6–12%), respectively. The resulting median (IQR) simulated residual 10-year cardiovascular risk was 23% (17–32%). The corresponding values for each subgroup are presented in Fig. 3.
Discussion
The DA VINCI study, conducted across 18 countries and 128 sites in Europe, provided an opportunity to assess the patterns of LLT use in contemporary practice, as well as gaps between guidelines and their implementation in patients with manifestations of ASCVD. Of the 2039 patients with ASCVD in this study, the majority (61%) did not achieve an LDL-C goal of < 70 mg/dl and only approximately one-fifth of patients (19%) reached the more stringent goal of < 55 mg/dl. Simulations from this study indicate that median LDL-C reductions of 24 (12–46) and 39 (27–61) mg/dl would be needed in this population to achieve an LDL-C level of 69 and 54 mg/dl, respectively. If LDL-C levels of 54 mg/dl were achieved, the estimated ARR would be expected to be 6% over 10 years as compared with an estimated ARR of 4% if LDL-C levels of 69 mg/dl were achieved. These results have implications for current clinical practice and highlight the extent that current residual risk among ASCVD patients receiving statins might be mitigated if the 2019 ESC/EAS guidelines were achieved in a population with ASCVD similar to the DA VINCI cohort.
The DA VINCI study shows that, in patients with ASCVD, LLT use consisted of monotherapy-based treatments in approximately 80–85% of cases. Use of combination therapy (e.g., statins with ezetimibe and/or PCSK9i) was low (< 20%), but use of combination therapy was twice as frequent among patients being managed for CAD than among those with CBD or PAD. The result of this monotherapy-based approach is that, among patients with major manifestations of ASCVD, the majority failed to attain either the 2019 ESC/EAS or the 2018 ACC/AHA LDL-C recommendations with current LLT use. In addition, among patients not achieving the 2018 ACC/AHA LDL-C recommendation of < 70 mg/dl), mean LDL-C levels were approximately 93 mg/dl (or 24 mg/dl higher than the recommended figure), with an estimated 10-year risk of cardiovascular events of 32%. Notably, even among patients who had achieved an LDL-C < 70 mg/dl (with a mean LDL-C of 56 mg/dl), the estimated 10-year risk of cardiovascular events was 35%.
The higher risk observed in these patients is likely due to additional comorbidities such as diabetes or a history of smoking. This observation highlights the importance of absolute risk when making clinical decisions about optimal LDL-C control for individual patients [19].
Large-scale Mendelian randomization genetic studies that simulate LLT use suggest that the cardiovascular benefits of LDL-C lowering should be similar, irrespective of the mechanism by which lowering is achieved when standardized for the same absolute difference in LDL-C [20, 21]. Furthermore, comparisons of pharmacological approaches to LDL-C lowering suggest that conflicting findings from trials can be harmonized when the observed RRR in cardiovascular events is standardized per an approximately 39 mg/dl (1 mmol/l) lowering in LDL-C and the number of years of treatment [20, 22]. Current guidelines recommend a stepwise approach strategy [13,14]. This strategy is based on the iterative development of evidence from randomized trials and economic considerations, with statins being the cornerstone of LDL-C lowering and subsequent therapies being added on. An impact of this approach is that, in routine clinical practice, there is an inevitable delay in the use of evidence-based therapies. For instance, although ezetimibe is widely available as a generic therapy, it remains underutilized. In the present study, the enrolment centres with participating physicians with a primary interest in lipid management only used ezetimibe in combination with statins in 9% of patients. This proportion is even lower when ezetimibe use is measured in unselected cohorts available through assessment of electronic health records [23].
There are notable differences between the 2019 ESC/EAS and 2018 ACC/AHA guidelines in their clinical approach to LLT. The 2019 ESC/EAS guidelines recommend that all patients with ASCVD are classified as very high risk and that an LDL-C goal of < 55 mg/dl for these patients is to be achieved. In contrast, the 2018 ACC/AHA guidelines classify high and very high-risk patients by the presence or absence of additional very high-risk characteristics and recommend an LDL-C ≥ 70 mg/dl to guide intensification of therapy among those at very high risk. The 2016 ESC/EAS guidelines [15] were closer to the current 2018 ACC/AHA guidelines in the sense of recommending an LDL-C goal of < 70 mg/dl for patients with ASCVD.
In the DA VINCI study, full implementation of the 2016 ESC/EAS or 2018 ACC/AHA guidelines would mean that an average LDL-C reduction of 24 mg/dl (relative reduction of 26%) would be required for patients not achieving an LDL-C < 70 mg/dl. It has been reported that doubling statin dosing produces approximately a further 6% reduction in LDL-C [24] and the addition of ezetimibe results in up to 25% further lowering [25]. As the IQR for absolute reductions in LDL-C ranged from 12 to 46 mg/dl, the majority of patients might achieve LDL-C < 70 mg/dl if moderate-intensity statins were optimized to high-intensity statins and ezetimibe was prescribed more frequently, with a smaller proportion of patients requiring PCSK9i. If LDL-C levels of 69 mg/dl were achieved, the estimated ARR would be expected to be 4%, with a simulated 10-year risk of 28% after attainment of that LDL-C level. These data are consistent with simulations conducted using US claims data [26]. In contrast, achieving the 2019 ESC/EAS goal of < 55 mg/dl would require greater absolute reductions of 39 mg/dl (IQR 27–91 mg/dl), with a relative reduction of 42%, to achieve an LDL-C level of 54 mg/dl. As a result, an increased use of PCSK9i with higher-intensity statins would likely be required to reach lower LDL-C goals. Inevitably, there may be some patients who may not be able to tolerate higher intensity regimens, and intensive statin therapy has been previously shown to be associated with an increased risk of developing new-onset diabetes with approximately one additional case per 1000 patients per year. However, this risk was outweighed by three predicted cardiovascular events being prevented during the same time period [27]. It is worth noting that from the HEYMANS real-world registry in Europe, when evolocumab treatment is used as part of combination therapy with an oral LLT such as statins and/or ezetimibe, this combination resulted in more patients attaining LDL-C levels of < 55 mg/dl in comparison with PCSK9i monotherapy, further reinforcing the importance of combination therapies [28]. Future DA VINCI analyses will examine the simulated LDL-C reductions associated with following specific treatment intensification pathways (e.g., treatment with high-intensity statins alone, adding ezetimibe, or adding ezetimibe and PCSK9i). Our observations are consistent with recent findings from the SWEDEHEART Registry, in which a population of patients who experienced a recent post-myocardial infarction (MI) was studied [29]. If LDL-C levels of 54 mg/dl were achieved, the estimated ARR would be expected to be 6%, with a simulated 10-year cardiovascular risk of 25%. It should be noted that, while our simulations were based on the conservative approach of patients achieving LDL-C levels of 69 mg/dl and 54 mg/dl, the actual LDL-C levels that would be achieved in clinical practice would likely be lower (and the associated benefits higher) if clinicians were to reduce LDL-C levels to < 70 mg/dl or < 55 mg/dl, particularly if optimized combination LLT was used.
Overall, patients being managed for CAD, CBD, and PAD each had similar 10-year cardiovascular risk estimates when receiving mainly monotherapy regimens, and baseline characteristics were largely similar between these groups. Historically, populations with CBD and PAD are understudied in large registries that usually focus on patients with MI and CAD. In patients with CAD, the benefits of LDL-C lowering with the use of ezetimibe and PCSK9i have been demonstrated in clinical trials [9–11]. As most trials recruited patients on the basis of CAD, and thus few patients have CBD or PAD only, it is possible that this has contributed to delays in evidence-based LLT use in these patient groups. Nevertheless, a recent study in patients with stroke showed that lower LDL-C levels achieved through more intensive LLT regimens (including ezetimibe alone, or with statin combination therapies) resulted in reductions in cardiovascular events [30].
The current 2018 ACC/AHA guidance to aid intensification of LLT is largely qualitative; the guidance identifies multiple different univariate very high-risk conditions [11, 31] but does not allow quantitative estimation of cardiovascular risk and thus does not allow absolute benefit estimation and re-estimation of risk after further LDL-C lowering. In this regard, our simulation study does not allow direct comparisons between the implementation of the 2018 ACC/AHA and 2019 ESC/EAS guidelines. Nevertheless, our study provides information that, at a population level, adoption of the lower LDL-C goal for patients with ASCVD as recommended by the 2019 ESC/EAS guidelines is expected to result in greater ARR and lower residual risk once the more stringent goals have been achieved.
Another interesting observation from our study is that, using quantitative approaches to estimate 10-year cardiovascular risk, the estimated 10-year cardiovascular risk was 35% in patients who achieved an LDL-C < 70 mg/dl (2018 ACC/AHA approach) with an average LDL-C of 56 mg/dl. These patients were more likely to have additional comorbidities such as diabetes or a history of smoking but also had increased use of more potent LLT regimens, including combination therapy. This may suggest that physicians are already identifying certain very high-risk comorbidity or lifestyle factors and treating such patients more aggressively. However, our observations suggest that achieving LDL-C goals without due consideration to absolute risk is only partly informative and may result in potentially modifiable risks being unaddressed [19]. A practical implication of this simulation is that the relative benefit from LLT is proportional to absolute reduction in LDL-C. A patient who is receiving LLT with an LDL-C level > 55 mg/dl would require more potent add-on therapies such as PCSK9i, which reduce LDL-C by 50–60%, thus likely achieving a further 28–34 mg/dl reduction in LDL-C levels. This is in contrast to simply ensuring that all patients achieve an LDL-C of 39 mg/dl, which could be achieved with additional oral therapies but would result on average in only a further 17 mg/dl lowering of LDL-C levels in that population. Both approaches have merits from a population health standpoint in terms of cardiovascular events prevented, but there are implications for drug acquisition costs, which also need to be considered.
The strengths and limitations of the present study merit consideration. Although a systematic approach was used to assess patients, the participating sites were likely to have focused on lipid management and prevention of cardiovascular events, so the findings may represent a better than average management scenario. The use of LLT may vary across countries, reflecting differences in healthcare systems, prevalence of other major cardiovascular risk factors, drug acquisition costs, and local guidelines, and may not necessarily reflect practice in other regions. Despite this, goal attainment is generally determined by the starting level of LDL-C prior to treatment, which would not be expected to vary significantly across regions. Furthermore, the LLT regimens used may vary across countries, but as the effects of LLT on percentage reduction in LDL-C should not vary across the same populations, the findings are meaningful to other populations. We used a simulation approach to provide inferences about potential treatment benefits rather than using observational longitudinal data. These simulations were based on the REACH equation, which has been derived from a large, global cohort [18], albeit with a relatively short follow-up (20 months). While we acknowledge that other risk equations are available, REACH is well established for examining risk in ASCVD patients [32]. We approximated the predictions from the REACH equation to 10-year cardiovascular risk, which could under- or overestimate risk. This under- or overestimation would apply equally to all individuals included, and hence, our study results could be considered as illustrative, exploring the concepts of different LDL-C goals within the caveats of the simulation and population studied. It is worth noting that the DA VINCI cohort described in this study included both very high-risk and high-risk ASCVD patients according to the current ACC/AHA criteria [14]. Hence, the present simulation may have underestimated cardiovascular risk and therefore absolute risk reduction among very high-risk patients (according to the ACC/AHA criteria), as the REACH equation was derived from a cohort that included patients who would not be considered as very high risk [33,34,35]. However, baseline risk varies across populations, whether one uses ESC/EAS or ACC/AHA criteria for very high risk, and these categories are qualitative rather than quantitative in providing measures of absolute risk and benefit. Therefore, the present study should be considered as illustrative within the limitations described. Finally, as with many studies, an additional limitation of our study was the homogeneous ethnicity of the DA VINCI cohort, in which over 90% of participants were white [33,34,35].
Conclusions
In a multi-country broad cohort of patients with ASCVD, the majority of patients who were mostly treated with statin monotherapy did not achieve 2019 ESC/EAS or 2018 ACC/AHA LDL-C recommended goals. Implementation of these guidelines would require greater use of combination therapies, with the more stringent 2019 ESC/EAS goal expected to yield better health outcomes than the 2018 ACC/AHA LDL-C approach.
Data Availability
Qualified researchers may request data from Amgen clinical studies. Complete data are available at the following: https://wwwext.amgen.com/science/clinical-trials/clinical-data-transparency-practices/
References
World Health Organization (2007). Prevention of cardiovascular disease: guidelines for assessment and management of total cardiovascular risk. Geneva: World Health Organization. http://www.who.int/iris/handle/10665/43685. Accessed 30 July 2021.
Dhindsa DS, Sandesara PB, Shapiro MD, Wong ND. The evolving understanding and approach to residual cardiovascular risk management. Review. Front Cardiovasc Med. 2020;7(88).
Amarenco P, Bogousslavsky J, Callahan A 3rd, et al. High-dose atorvastatin after stroke or transient ischemic attack. N Engl J Med. 2006;355(6):549–59.
Baigent C, Keech A, Kearney PM, et al. Efficacy and safety of cholesterol-lowering treatment: prospective meta-analysis of data from 90,056 participants in 14 randomised trials of statins. Lancet. 2005;366(9493):1267–78.
Cholesterol Treatment Trialists’ (CTT) Collaboration, Baigent C, Blackwell L, et al. Efficacy and safety of more intensive lowering of LDL cholesterol: a meta-analysis of data from 170,000 participants in 26 randomised trials. Lancet. 2010;376(9753):1670–81.
Heart Protection Study Collaborative Group. Randomized trial of the effects of cholesterol-lowering with simvastatin on peripheral vascular and other major vascular outcomes in 20,536 people with peripheral arterial disease and other high-risk conditions. J Vasc Surg. Apr 2007;45(4):645–54.
Sabatine MS, Giugliano RP, Keech AC, et al. Evolocumab and clinical outcomes in patients with cardiovascular disease. N Engl J Med. 2017;376(18):1713–22.
Schwartz GG, Steg PG, Szarek M, et al. Alirocumab and cardiovascular outcomes after acute coronary syndrome. N Engl J Med. 2018;379(22):2097–107.
Jukema JW, Szarek M, Zijlstra LE, et al. Alirocumab in patients with polyvascular disease and recent acute coronary syndrome: ODYSSEY OUTCOMES trial. J Am Coll Cardiol. 2019;74(9):1167–76.
Giugliano RP, Cannon CP, Blazing MA, et al. Benefit of adding ezetimibe to statin therapy on cardiovascular outcomes and safety in patients with versus without diabetes mellitus: results from IMPROVE-IT (IMProved Reduction of Outcomes: Vytorin Efficacy International Trial). Circulation. 2018;137(15):1571–82.
Bonaca MP, Nault P, Giugliano RP, et al. Low-density lipoprotein cholesterol lowering with evolocumab and outcomes in patients with peripheral artery disease: insights from the FOURIER Trial (Further cardiovascular OUtcomes Research with PCSK9 Inhibition in subjects with Elevated Risk). Circulation. 2018;137(4):338–50.
Bohula EA, Giugliano RP, Cannon CP, et al. Achievement of dual low-density lipoprotein cholesterol and high-sensitivity C-reactive protein targets more frequent with the addition of ezetimibe to simvastatin and associated with better outcomes in IMPROVE-IT. Circulation. 2015;132(13):1224–33.
Mach F, Baigent C, Catapano AL, et al. 2019 ESC/EAS guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Eur Heart J. 2020;41(1):111–88.
Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the management of blood cholesterol: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2019;139(25):e1082–143.
Catapano AL, Graham I, De Backer G, et al. 2016 ESC/EAS guidelines for the management of dyslipidaemias. Eur Heart J. 2016;37(39):2999–3058.
Ray KK, Molemans B, Schoonen WM, et al. EU-wide cross-sectional observational study of lipid-modifying therapy use in secondary and primary care: the DA VINCI study. Eur J Prev Cardiol. 2020 .
European network of Centres for Pharmacoepidemiology and Pharmacovigilance. EU PAS Register: EU-wide cross-sectional observational study of lipid-modifying therapy use in secondary and primary care (20150333). http://www.encepp.eu/encepp/viewResource.htm?id=31479. Accessed 19 Feb 2020.
Wilson PW, D'Agostino R, Sr., Bhatt DL, et al. An international model to predict recurrent cardiovascular disease. Am J Med. 2012;125(7):695–703.e1.
Nishikido T, Fayyad R, Melamed S, Ray KK. TRS2P and LDL-C alone or in combination for predicting absolute benefits from additional LDL-C lowering: analysis from the TNT trial. Atherosclerosis. 2021;322:8–14.
Ference BA, Ginsberg HN, Graham I, et al. Low-density lipoproteins cause atherosclerotic cardiovascular disease. 1. Evidence from genetic, epidemiologic, and clinical studies. A consensus statement from the European Atherosclerosis Society Consensus Panel. Eur Heart J. 2017;38(32):2459–72.
Ference BA, Ray KK, Catapano AL, et al. Mendelian randomization study of ACLY and cardiovascular disease. N Engl J Med. 2019;380(11):1033–42.
Ference BA, Cannon CP, Landmesser U, Lüscher TF, Catapano AL, Ray KK. Reduction of low density lipoprotein-cholesterol and cardiovascular events with proprotein convertase subtilisin-kexin type 9 (PCSK9) inhibitors and statins: an analysis of FOURIER, SPIRE, and the Cholesterol Treatment Trialists Collaboration. Eur Heart J. 2018;39(27):2540–5.
Khunti K, Danese MD, Kutikova L, et al. Association of a combined measure of adherence and treatment intensity with cardiovascular outcomes in patients with atherosclerosis or other cardiovascular risk factors treated with statins and/or ezetimibe. JAMA Netw Open. 2018;1(8):e185554.
Nicholls SJ, Brandrup-Wognsen G, Palmer M, Barter PJ. Meta-analysis of comparative efficacy of increasing dose of atorvastatin versus rosuvastatin versus simvastatin on lowering levels of atherogenic lipids (from VOYAGER). Am J Cardiol. 2010;105(1):69–76.
Ballantyne CM, Laufs U, Ray KK, et al. Bempedoic acid plus ezetimibe fixed-dose combination in patients with hypercholesterolemia and high CVD risk treated with maximally tolerated statin therapy. Eur J Prev Cardiol. 2020;27(6):593–603.
Cannon CP, Khan I, Klimchak AC, Reynolds MR, Sanchez RJ, Sasiela WJ. Simulation of lipid-lowering therapy intensification in a population with atherosclerotic cardiovascular disease. JAMA Cardiology. 2017;2(9):959–66.
Preiss D, Seshasai SRK, Welsh P, et al. Risk of incident diabetes with intensive-dose compared with moderate-dose statin therapy: a meta-analysis. JAMA. 2011;305(24):2556–64.
Ray KK, Dhalwani N, Sibartie M, et al. Low density lipoprotein cholesterol levels exceed the recommended European threshold for PCSK9i initiation: lessons from the HEYMANS study. Eur Heart J Qual Care Clin Outcomes. 2022.https://doi.org/10.1093/ehjqcco/qcac009
Schubert J, Lindahl B, Melhus H, et al. Low-density lipoprotein cholesterol reduction and statin intensity in myocardial infarction patients and major adverse outcomes: a Swedish nationwide cohort study. Eur Heart J. 2020;42(3):243–52.
Amarenco P, Kim JS, Labreuche J, et al. A comparison of two LDL cholesterol targets after ischemic stroke. N Engl J Med. 2020;382(1):9.
Sabatine MS, De Ferrari GM, Giugliano RP, et al. Clinical benefit of evolocumab by severity and extent of coronary artery disease: analysis from FOURIER. Circulation. 2018;138(8):756–66.
Betts MB, Milev S, Hoog M, et al. Comparison of recommendations and use of cardiovascular risk equations by health technology assessment agencies and clinical guidelines. Value Health. 2019;22(2):210–19.
Al-Shamsi S, Govender RD. Validation of the REduction of Atherothrombosis for Continued Health (REACH) prediction model for recurrent cardiovascular disease among United Arab Emirates Nationals. BMC Res Notes. 2020;13(1):484.
Taylor B, Lothgren M, Villa G, Lindgren P, Van Hout B, Mark D. Differences between observed and predicted cardiovascular event rates using the Framingham and REACH equations: the case of high-intensity statin users in the United Kingdom. Circulation. 2015;132(Suppl 3):A18114.
Danese MD, Pemberton-Ross P, Catterick D, Villa G. Estimation of the increased risk associated with recurrent events or polyvascular atherosclerotic cardiovascular disease in the United Kingdom. Eur J Prev Cardiol. 2021;28(3):335–43.
Acknowledgements
The authors would like to thank Sinéad Flannery PhD (PharmaGenesis London, London, UK) and Ryan Woodrow (Aspire Scientific, Bollington, UK) for medical writing and editorial support, which was funded by Amgen. KKR acknowledges support from the National Institute for Health Research (NIHR) Imperial Biomedical Research Centre and from the NIHR ARC for North West London. AJVV acknowledges support from the “Programa Beatriz Galindo” from the Ministry of Universities, Spain, and University of Seville, Spain.
Funding
This study was funded by Amgen Europe (GmbH).
Author information
Authors and Affiliations
Consortia
Contributions
KKR and NRP contributed substantially to the study design and concept; KKR, AP, MV, HKI, SK, IGB, CV, DT, RK, VM, SDS, WJJ, JJJ, DG, KR, LM, SR, PC, OP, and NRP were involved in data acquisition; SB and GV conducted the data analyses; and all authors assisted with interpretation of the data. All authors were involved in drafting of the manuscript, provided critical revisions for important intellectual content, approved the final version submitted for publication, and agreed to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Competing Interests
AJV-V acknowledges past or current participation in research grants to Imperial College London from Pfizer, Amgen, MSD, Sanofi-Aventis, Daiichi Sankyo, and Regeneron; and received personal fees for consulting from Bayer and Regeneron and honoraria for lectures from Amgen, Mylan, and Akcea; all outside the submitted work.
SB is an employee of Amgen Ltd and holds stock in Amgen.
GV is an employee of Amgen (Europe) GmbH and holds stock in Amgen.
JB has received speaker honoraria from Amgen and research grant support from AstraZeneca.
GK has no relevant disclosures.
JM has no relevant disclosures.
MB has participated in speakers bureau for Akcea, Amgen, Daiichi Sankyo, KRKA, MSD, Mylan, Polpharma, Sanofi-Aventis, Servier, and Valeant; served as a consultant to Abbott Vascular, Akcea, Amgen, Daiichi Sankyo, Esperion, Freia Pharmaceuticals, MSD, Polfarmex, Resverlogix, and Sanofi-Aventis; and received grants from Mylan, Sanofi, and Valeant.
SDS has no relevant disclosures.
DG has received honoraria for educational activities from Amgen, AstraZeneca, Berlin-Chemie, Boehringer Ingelheim, Merck, Mylan, Novartis, Novo Nordisk, Pfizer, Sanofi, and Servier.
IG-B has received personal fees and non-financial support from Akcea, Amgen and Sanofi, and personal fees from Aegerion, Amarin, Daiichi Sankyo, Novartis, and Regeneron, outside the submitted work.
GKH has received funding for clinical trial activities and/or lecture fees from Aegerion, Amgen, AstraZeneca, Eli Lilly, Genzyme, Kowa, Pfizer, Regeneron Pharmaceuticals, Roche, Sanofi, The Medicines Company, and Ionis until April 2019, and fees were paid to his institute. Since April 2019 GKH is part-time employed by Novo Nordisk.
JJJ has received research grant/support from Valeant, and has served as a consultant or speaker for ALAB Laboratories, Amgen, Bioton, Boehringer Ingelheim, Celgene, Microlife, Servier, Teva, and Valeant.
JWJ or his institution department has received research grants from, and/or was a speaker (with or without lecture fees) at meetings sponsored by Amgen, AstraZeneca, Athera, Biotronik, Boston Scientific, Dalcor, Daiichi Sankyo, Lilly, Medtronic, Merck-Schering-Plough, Pfizer, Roche, Sanofi-Aventis, The Medicine Company, the Netherlands Heart Foundation, CardioVascular Research the Netherlands (CVON), the Netherlands Heart Institute and the European Community Framework KP7 Programme.
RGK has received honoraria from, and has been a member of the speakers bureau for, Amgen, AstraZeneca, Bayer, Boehringer Ingelheim, Merck, Pfizer, and Sanofi-Aventis.
SK has received grant/research support from Amgen, AstraZeneca, and Bayer; and consulting fees/honoraria from Amgen, Bayer, BMS, Boehringer Ingelheim, MSD, Mesi, Pfizer, Philips Healthcare, Sanofi, and Servier.
HKI has received honoraria for contributing to advisory boards or oral presentations from Allergan, Amgen, AstraZeneca, Bayer, Boehringer Ingelheim, Bristol Myers Squibb, MSD, Pfizer, and St Jude.
VM has no conflict of interest relevant to this publication.
LM has received fees for lectures and/or advisory work from Amarin, Amgen, Amryt, Mylan, Novartis, Sanofi-Regeneron, and Servier.
APa has received research grants and personal fees from Amgen, AstraZeneca, Bayer, Pfizer, and Sanofi-Aventis.
APe has no relevant disclosures.
PC has no relevant disclosures.
KR has received research grants for clinical trials, and consulting fees and honoraria for presentations, from Amgen, AstraZeneca, Bayer, Boehringer Ingelheim, Eli Lilly, Gilead, Merck, Mylan, Novo Nordisk, Pfizer, Sanofi, and Zentiva.
PS has received honoraria for being a speaker and advisory board member from Amgen, AstraZeneca, MSD, and Sanofi.
SR has received honoraria for being a speaker and advisory board member from Akcea, Amgen, AstraZeneca, and Sanofi; and grants from Amgen, AstraZeneca, and Sanofi.
DT has no relevant disclosures.
CV has received research grant(s)/support and honoraria from Amgen, ELPEN, MSD, Sanofi, and VIANEX.
MV has received grants, personal fees, or non-financial support from Abbott Laboratories, Amgen, AstraZeneca, Boehringer Ingelheim, KRKA, Mylan, Novartis, Novo Nordisk, Pfizer, Sanofi-Aventis, Servier, and Zentiva, outside of the submitted work.
ALC has received research grant(s)/support from Amgen, Eli Lilly, Mylan, Menarini, Sanofi, and Sanofi-Regeneron; and has served as a consultant for or received honoraria from Aegerion, Akcea, Amgen, Amryt, AstraZeneca, Daiichi Sankyo, Esperion, Genzyme, Ionis Pharmaceuticals, Kowa, Medco, Menarini, MSD, Mylan, Novartis, Recordati, Regeneron, and Sanofi.
NRP has received financial support from several pharmaceutical companies that manufacture lipid-lowering agents, for consultancy fees (Amgen and Pfizer), research projects and staff (Amgen and Pfizer), and for arranging and speaking at educational meetings (Amgen, MSD, and Pfizer). He holds no stocks and shares in any such companies. NRP is supported by the National Institute for Health Research Senior Investigator Awards, Biomedical Research Centre funding, and the British Heart Foundation Research Centre Excellence Award.
KKR reports grants from Amgen during the conduct of the study; and has received personal fees from AbbVie, Aegerion, Akcea, Algorithm, AstraZeneca, Bayer, Boehringer Ingelheim, Cerenis, Cipla, Dr Reddys, Esperion, Kowa, Lilly, Novartis, Silence Therapeutics, Takeda, The Medicines Company, and Zuellig Pharma; and grants and personal fees from Amgen, Daiichi Sankyo, MSD, Sanofi-Regeneron, and Pfizer, outside the submitted work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Vallejo-Vaz, A.J., Bray, S., Villa, G. et al. Implications of ACC/AHA Versus ESC/EAS LDL-C Recommendations for Residual Risk Reduction in ASCVD: A Simulation Study From DA VINCI. Cardiovasc Drugs Ther 37, 941–953 (2023). https://doi.org/10.1007/s10557-022-07343-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10557-022-07343-x