Abstract
Purpose
Acute pulmonary embolism (APE) is a critical cardiopulmonary condition associated with right ventricular (RV) failure and death. While pharmacological inhibition of matrix metalloproteinases (MMPs) attenuated APE-induced hemodynamic alterations, no previous study has evaluated whether this approach decreases APE-induced mortality and RV deformation. We tested this hypothesis in rats.
Methods
Wistar rats received an intraperitoneal injection of 30 mg/kg doxycycline (or saline) and after 30 min a sterile suspension of 300 μm microsphere (21 mg/kg or saline) was injected into the tail vein. After 24 h, surviving animals were killed and the RVs were collected and used for histological and morphometric analyses. RV samples were also homogenized and assayed by SDS-polyacrilamide gel electrophoresis gelatin zymography to evaluate MMP-2 and MMP-9 activity. In situ zymography was carried out in RV to assess MMP activity and neutrophil accumulation in myocardial tissue was determined by myeloperoxidase activity measurement. Dihydroethidium was used to assess RV reactive oxygen species concentrations.
Results
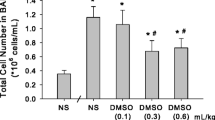
APE caused 72.5 % mortality during the first hour of follow up. Pretreatment with doxycycline was associated with significant decrease in APE-induced mortality rate to 50 % (P < 0.05). Embolized animals showed significant RV dilation, and pretreatment with doxycycline blunted this alteration (P < 0.05). APE increased the number of RV neutrophils and MMP-9 levels (P < 0.05). Pretreatment with doxycycline blunted APE-induced increases in RV myocardial ROS concentrations and MMP gelatinolytic activity (both P < 0.05).
Conclusions
These findings show that MMP inhibition with doxycycline protects against APE-induced mortality and RV enlargement. These beneficial effects are probably due to attenuation of APE-induced oxidative stress and increases in ventricular proteolytic activity and suggest that doxycycline may have promising protective effects in patients with APE.






Similar content being viewed by others
References
Wood KE. Major pulmonary embolism: Review of a pathophysiologic approach to the golden hour of hemodynamically significant pulmonary embolism. Chest. 2002;121:877–905.
Watts JA, Marchick MR, Kline JA. Right ventricular heart failure from pulmonary embolism: Key distinctions from chronic pulmonary hypertension. J Card Fail. 2010;16:250–9.
Lualdi JC, Goldhaber SZ. Right ventricular dysfunction after acute pulmonary embolism: Pathophysiologic factors, detection, and therapeutic implications. Am Heart J. 1995;130:1276–82.
Watts JA, Gellar MA, Obraztsova M, Kline JA, Zagorski J. Role of inflammation in right ventricular damage and repair following experimental pulmonary embolism in rats. Int J Exp Pathol. 2008;89:389–99.
Watts JA, Zagorski J, Gellar MA, Stevinson BG, Kline JA. Cardiac inflammation contributes to right ventricular dysfunction following experimental pulmonary embolism in rats. J Mol Cell Cardiol. 2006;41:296–307.
Zagorski J, Gellar MA, Obraztsova M, Kline JA, Watts JA. Inhibition of cinc-1 decreases right ventricular damage caused by experimental pulmonary embolism in rats. J Immunol. 2007;179:7820–6.
Iwadate K, Doi M, Tanno K, et al. Right ventricular damage due to pulmonary embolism: Examination of the number of infiltrating macrophages. Forensic Sci Int. 2003;134:147–53.
Vinten-Johansen J. Involvement of neutrophils in the pathogenesis of lethal myocardial reperfusion injury. Cardiovasc Res. 2004;61:481–97.
Hobeika MJ, Thompson RW, Muhs BE, Brooks PC, Gagne PJ. Matrix metalloproteinases in peripheral vascular disease. J Vasc Surg. 2007;45:849–57.
Schulz R. Intracellular targets of matrix metalloproteinase-2 in cardiac disease: Rationale and therapeutic approaches. Annu Rev Pharmacol Toxicol. 2007;47:211–42.
Chow AK, Cena J, Schulz R. Acute actions and novel targets of matrix metalloproteinases in the heart and vasculature. Br J Pharmacol. 2007;152:189–205.
Ali MA, Cho WJ, Hudson B, et al. Titin is a target of matrix metalloproteinase-2: Implications in myocardial ischemia/reperfusion injury. Circulation. 2010;122:2039–47.
Palei AC, Zaneti RA, Fortuna GM, Gerlach RF, Tanus-Santos JE. Hemodynamic benefits of matrix metalloproteinase-9 inhibition by doxycycline during experimental acute pulmonary embolism. Angiology. 2005;56:611–7.
Neto-Neves EM, Dias-Junior CA, Rizzi E, et al. Metalloproteinase inhibition protects against cardiomyocyte injury during experimental acute pulmonary thromboembolism. Crit Care Med. 2011;39:349–56.
Griffin MO, Ceballos G, Villarreal FJ. Tetracycline compounds with non-antimicrobial organ protective properties: Possible mechanisms of action. Pharmacol Res. 2011;63:102–7.
Souza-Costa DC, Figueiredo-Lopes L, Alves-Filho JC, et al. Protective effects of atorvastatin in rat models of acute pulmonary embolism: Involvement of matrix metalloproteinase-9. Crit Care Med. 2007;35:239–45.
Jacob-Ferreira AL, Palei AC, Cau SB, et al. Evidence for the involvement of matrix metalloproteinases in the cardiovascular effects produced by nicotine. Eur J Pharmacol. 2010;627:216–22.
Rizzi E, Castro MM, Prado CM, et al. Matrix metalloproteinase inhibition improves cardiac dysfunction and remodeling in 2-kidney, 1-clip hypertension. J Card Fail. 2010;16:599–608.
Sousa-Santos O, Neto-Neves EM, Ferraz KC, et al. Antioxidant treatment protects against matrix metalloproteinase activation and cardiomyocyte injury during acute pulmonary thromboembolism. Naunyn Schmiedebergs Arch Pharmacol. 2012;385:685–96.
Rizzi E, Castro MM, Ceron CS, et al. Tempol inhibits tgf-beta and mmps upregulation and prevents cardiac hypertensive changes. Int J Cardiol 2011
Sanchez O, Trinquart L, Colombet I, et al. Prognostic value of right ventricular dysfunction in patients with haemodynamically stable pulmonary embolism: A systematic review. Eur Heart J. 2008;29:1569–77.
Faurschou M, Borregaard N. Neutrophil granules and secretory vesicles in inflammation. Microbes Infect. 2003;5:1317–27.
Rouet-Benzineb P, Buhler JM, Dreyfus P, et al. Altered balance between matrix gelatinases (mmp-2 and mmp-9) and their tissue inhibitors in human dilated cardiomyopathy: Potential role of mmp-9 in myosin-heavy chain degradation. Eur J Heart Fail. 1999;1:337–52.
Uzuelli JA, Dias-Junior CA, Tanus-Santos JE. Severity dependent increases in circulating cardiac troponin i and mmp-9 concentrations after experimental acute pulmonary thromboembolism. Clin Chim Acta. 2008;388:184–8.
Fortuna GM, Figueiredo-Lopes L, Dias-Junior CA, Gerlach RF, Tanus-Santos JE. A role for matrix metalloproteinase-9 in the hemodynamic changes following acute pulmonary embolism. Int J Cardiol. 2007;114:22–7.
Souza-Costa DC, Zerbini T, Palei AC, Gerlach RF, Tanus-Santos JE. L-arginine attenuates acute pulmonary embolism-induced increases in lung matrix metalloproteinase-2 and matrix metalloproteinase-9. Chest. 2005;128:3705–10.
Fernandez-Patron C, Radomski MW, Davidge ST. Vascular matrix metalloproteinase-2 cleaves big endothelin-1 yielding a novel vasoconstrictor. Circ Res. 1999;85:906–11.
Fernandez-Patron C, Zouki C, Whittal R, et al. Matrix metalloproteinases regulate neutrophil-endothelial cell adhesion through generation of endothelin-1[1–32]. FASEB J. 2001;15:2230–40.
Tanus-Santos JE, Gordo WM, Udelsmann A, Cittadino MH, Moreno Jr H. Nonselective endothelin-receptor antagonism attenuates hemodynamic changes after massive pulmonary air embolism in dogs. Chest. 2000;118:175–9.
Tanus-Santos JE, Gordo WM, Udelsmann A, Moreno Jr H. The hemodynamic effects of endothelin receptor antagonism during a venous air infusion in dogs. Anesth Analg. 2000;90:102–6.
Kraus RL, Pasieczny R, Lariosa-Willingham K, et al. Antioxidant properties of minocycline: Neuroprotection in an oxidative stress assay and direct radical-scavenging activity. J Neurochem. 2005;94:819–27.
Castro MM, Tanus-Santos JE, Gerlach RF. Matrix metalloproteinases: Targets for doxycycline to prevent the vascular alterations of hypertension. Pharmacol Res. 2011;64:567–72.
Acknowledgments
We gratefully acknowledge to the excellent technical support of Maria Elena Riul. This study was funded by Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP-Brazil) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq-Brazil).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cau, S.B.A., Barato, R.C., Celes, M.R. et al. Doxycycline Prevents Acute Pulmonary Embolism-Induced Mortality and Right Ventricular Deformation in Rats. Cardiovasc Drugs Ther 27, 259–267 (2013). https://doi.org/10.1007/s10557-013-6458-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10557-013-6458-9