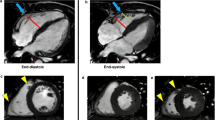
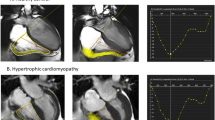
Abstract
Right ventricular (RV) fibrosis is increasingly recognized as the underlying pathological substrate in a variety of clinical conditions. We sought to employ cardiac magnetic resonance (CMR) techniques of strain imaging and longitudinal relaxation time (T1) mapping to better examine the relationship between RV function and structure. Our aim was to initially evaluate the feasibility of these techniques to evaluate the right ventricle. We then sought to explore the relationship between RV function and underlying fibrosis, along with examining the evolution of RV remodeling according to the amount of baseline fibrosis. Echocardiography was performed in 102 subjects with non-ischemic cardiomyopathy. Right ventricular parameters were assessed including: fractional area change (FAC) and longitudinal strain. The same cohort underwent CMR. Post-contrast T1 mapping was performed as a marker of fibrosis with a Look-Locker technique using inversion recovery imaging. Mid-ventricular post-contrast T1 values of the RV free wall, RV septum and lateral LV were calculated using prototype analysis software. Biventricular volumetric data including ejection fraction was measured by CMR using a cine short axis stack. CMR strain analysis was also performed to assess 2D RV longitudinal and radial strain. Simultaneous biochemical and anthropometric data were recorded. Subjects were followed over a median time of 29 months (IQR 20–37 months) with echocardiography to evaluate temporal change in RV FAC according to baseline post-contrast T1 values. Longitudinal data analysis was performed to adjust for patient loss during follow-up. Subjects (62% men, 51 ± 15 years) had mild to moderately impaired global RV systolic function (RVEF = 39 ± 15%; RVEDV = 187 ± 69 ml; RVESV = 119 ± 68 ml) and moderate left ventricular dysfunction at baseline (LVEF 30 ± 17%). Good correlation was observed between mean LV and RV post-contrast T1 values (r = 0.652, p < 0.001), with similar post-contrast T1 values maintained in both the RV free wall and septum (r = 0.761, p < 0.001). CMR RVEF demonstrated a proportional correlation with echocardiographic measures of RV longitudinal function and CMR RV strain (longitudinal r = −0.449, p = 0.001; radial r = −0.549, p < 0.001). RVEF was related to RV post-contrast T1 values, particularly in those with RV dysfunction (free wall T1 r = 0.259 p = 0.027; septal T1 r = 0.421 p < 0.001). RV strain was also related to RV post-contrast T1 values (r = −0.417, p = 0.002). Linear regression analysis demonstrated strain and post-contrast T1 values to be independently associated with RVEF. Subjects with severe RV dysfunction (CMR RVEF <25%) demonstrated lower RV CMR strain (longitudinal p = 0.018; radial p < 0.001), RV T1 values (free wall p = 0.013; septum <0.001) and RV longitudinal echocardiography parameters despite no difference in afterload. During follow-up, those with RV free wall post-contrast T1 values ≥ 350 ms demonstrated ongoing improvement in FAC (Δ6%), whilst values <350 ms were associated with deterioration in RV function (ΔFAC = −5%) (p = 0.026). CMR provides a comprehensive method by which to evaluate right ventricular function. Post-contrast T1 mapping and CMR strain imaging are technically feasible and provide incremental information regarding global RV function and structure. The proportional relationship between RV function and post-contrast T1 values supports that myocardial fibrosis is a causative factor of RV dysfunction in NICM, irrespective of RV afterload. This same structural milieu also appears integral to the propensity for both positive and negative RV remodeling long-term, suggestive that this is also determined by the degree of underlying RV fibrosis.






Similar content being viewed by others
References
Hinojar R, Varma N, Child N, Goodman B, Jabbour A, Yu CY, Gebker R, Doltra A, Kelle S, Khan S, Rogers T, Arroyo Ucar E, Cummins C, Carr-White G, Nagel E, Puntmann VO (2015) T1 mapping in discrimination of hypertrophic phenotypes: hypertensive heart disease and hypertrophic cardiomyopathy: findings from the international t1 multicenter cardiovascular magnetic resonance study. Circ Cardiovasc Imaging 8(12)
Puntmann VO, Voigt T, Chen Z, Mayr M, Karim R, Rhode K, Pastor A, Carr-White G, Razavi R, Schaeffter T, Nagel E (2013) Native t1 mapping in differentiation of normal myocardium from diffuse disease in hypertrophic and dilated cardiomyopathy. JACC Cardiovasc Imaging 6:475–484
Jellis C, Wright J, Kennedy D, Sacre J, Jenkins C, Haluska B, Martin J, Fenwick J, Marwick TH (2011) Association of imaging markers of myocardial fibrosis with metabolic and functional disturbances in early diabetic cardiomyopathy. Circ Cardiovasc Imaging 4:693–702
Iles L, Pfluger H, Phrommintikul A, Cherayath J, Aksit P, Gupta SN, Kaye DM, Taylor AJ (2008) Evaluation of diffuse myocardial fibrosis in heart failure with cardiac magnetic resonance contrast-enhanced t1 mapping. J Am Coll Cardiol 52:1574–1580
Hong YJ, Park CH, Kim YJ, Hur J, Lee HJ, Hong SR, Suh YJ, Greiser A, Paek MY, Choi BW, Kim TH (2015) Extracellular volume fraction in dilated cardiomyopathy patients without obvious late gadolinium enhancement: comparison with healthy control subjects. Int J Cardiovasc Imaging 31(Suppl 1):115–122
Burke AP, Farb A, Tashko G, Virmani R (1998) Arrhythmogenic right ventricular cardiomyopathy and fatty replacement of the right ventricular myocardium: are they different diseases? Circulation 97:1571–1580
van de Veerdonk MC, Bogaard HJ, Voelkel NF (2016) The right ventricle and pulmonary hypertension. Heart Fail Rev 21(3):259–271
La Gerche A, Roberts T, Claessen G (2014) The response of the pulmonary circulation and right ventricle to exercise: exercise-induced right ventricular dysfunction and structural remodeling in endurance athletes (2013 grover conference series). Pulm Circ 4:407–416
Puchalski MD, Williams RV, Askovich B, Minich LL, Mart C, Tani LY (2007) Assessment of right ventricular size and function: echo versus magnetic resonance imaging. Congenit Heart Dis 2:27–31
Kawel-Boehm N, Dellas Buser T, Greiser A, Bieri O, Bremerich J, Santini F (2014) In-vivo assessment of normal t1 values of the right-ventricular myocardium by cardiac mri. Int J Cardiovasc Imaging 30:323–328
Platts D, Brown M, Javorsky G, West C, Kelly N, Burstow D (2010) Comparison of fluoroscopic versus real-time three-dimensional transthoracic echocardiographic guidance of endomyocardial biopsies. Eur J Echocardiogr 11:637–643
Aquaro GD, Camastra G, Monti L, Lombardi M, Pepe A, Castelletti S, Maestrini V, Todiere G, Masci P, di Giovine G, Barison A, Dellegrottaglie S, Perazzolo Marra M, Pontone G, Di Bella G (2016) Reference values of cardiac volumes, dimensions, and new functional parameters by MR: a multicenter, multivendor study. J Magn Reson Imaging 45(4):1055–1067
Foppa M, Arora G, Gona P, Ashrafi A, Salton CJ, Yeon SB, Blease SJ, Levy D, O’Donnell CJ, Manning WJ, Chuang ML (2016) Right ventricular volumes and systolic function by cardiac magnetic resonance and the impact of sex, age, and obesity in a longitudinally followed cohort free of pulmonary and cardiovascular disease: the framingham heart study. Circ Cardiovasc Imaging 9:e003810
Gai N, Turkbey EB, Nazarian S, van der Geest RJ, Liu CY, Lima JA, Bluemke DA (2011) T1 mapping of the gadolinium-enhanced myocardium: adjustment for factors affecting interpatient comparison. Magn Reson Med 65:1407–1415
Puntmann VO, Carr-White G, Jabbour A, Yu CY, Gebker R, Kelle S, Hinojar R, Doltra A, Varma N, Child N, Rogers T, Suna G, Arroyo Ucar E, Goodman B, Khan S, Dabir D, Herrmann E, Zeiher AM, Nagel E (2016) T1-mapping and outcome in nonischemic cardiomyopathy: all-cause mortality and heart failure. JACC Cardiovasc Imaging 9:40–50
Rudski LG, Lai WW, Afilalo J, Hua L, Handschumacher MD, Chandrasekaran K, Solomon SD, Louie EK, Schiller NB (2010) Guidelines for the echocardiographic assessment of the right heart in adults: a report from the american society of echocardiography endorsed by the european association of echocardiography, a registered branch of the european society of cardiology, and the canadian society of echocardiography. J Am Soc Echocardiogr 23:685–713
Dabir D, Child N, Kalra A, Rogers T, Gebker R, Jabbour A, Plein S, Yu CY, Otton J, Kidambi A, McDiarmid A, Broadbent D, Higgins DM, Schnackenburg B, Foote L, Cummins C, Nagel E, Puntmann VO (2014) Reference values for healthy human myocardium using a t1 mapping methodology: results from the international t1 multicenter cardiovascular magnetic resonance study. J Cardiovasc Magn Reson 16:69
Freed BHSE, Benefield BC, Spottiswoode B, Brady C, Allen B, Shah SJ, Carr JC, Markl M, Collins JD (2016) Right ventricular t1 mapping: a feasibility study. J Heart Lung Transplant 35:S275–S276
Garcia-Lunar IPP, Pozo E, Calcagno C, Ramachandran S, Adapoe CM, Nair A, Jacobi A, Fuster V, Sanz J (2013) Myocardial fibrosis with t1 mapping and right ventricular performance in pulmonary hypertension. J Cardiovasc Magn Reson 15(Suppl 1):E102
Gardner BI, Bingham SE, Allen MR, Blatter DD, Anderson JL (2009) Cardiac magnetic resonance versus transthoracic echocardiography for the assessment of cardiac volumes and regional function after myocardial infarction: an intrasubject comparison using simultaneous intrasubject recordings. Cardiovasc Ultrasound 7:38
Farsalinos KE, Daraban AM, Unlu S, Thomas JD, Badano LP, Voigt JU (2015) Head-to-head comparison of global longitudinal strain measurements among nine different vendors: the EACVI/ASE inter-vendor comparison study. J Am Soc Echocardiogr 28:1171–1181, e1172
Yingchoncharoen T, Jellis C, Popovic ZB, Wang L, Gai N, Levy WC, Tang WH, Flamm S, Kwon DH (2016) Focal fibrosis and diffuse fibrosis are predictors of reversed left ventricular remodeling in patients with non-ischemic cardiomyopathy. Int J Cardiol 221:498–504
Lemarie J, Huttin O, Girerd N, Mandry D, Juilliere Y, Moulin F, Lemoine S, Beaumont M, Marie PY, Selton-Suty C (2015) Usefulness of speckle-tracking imaging for right ventricular assessment after acute myocardial infarction: a magnetic resonance imaging/echocardiographic comparison within the relation between aldosterone and cardiac remodeling after myocardial infarction study. J Am Soc Echocardiogr 28:818–827 e814
Wong C, Marwick TH (2007) Obesity cardiomyopathy: pathogenesis and pathophysiology. Nat Clin Pract Cardiovasc Med 4:436–443
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No disclosures or conflicts.
Rights and permissions
About this article
Cite this article
Jellis, C.L., Yingchoncharoen, T., Gai, N. et al. Correlation between right ventricular T1 mapping and right ventricular dysfunction in non-ischemic cardiomyopathy. Int J Cardiovasc Imaging 34, 55–65 (2018). https://doi.org/10.1007/s10554-017-1113-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-017-1113-3