Abstract
Purpose
A life course perspective to cancer incidence is important for understanding effects of the environment during early life on later cancer risk. We assessed spatial clusters of cancer incidence based on early life location defined as 1940 US Census Enumeration District (ED).
Methods
A cohort of 260,585 individuals aged 0–40 years in 1940 was selected. Individuals were followed from 1940 to cancer diagnosis, death, or last residence in Utah. We geocoded ED centroids in Utah for the 1940 Census. Spatial scan statistics with purely spatial elliptic scanning window were used to identify spatial clusters of EDs with excess cancer rates across 26 cancer types, assuming a discrete Poisson model.
Results
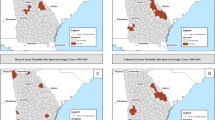
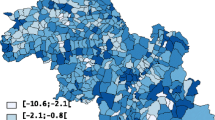
Cancer was diagnosed in 66,904 (25.67%) individuals during follow-up across 892 EDs. Average follow-up was 50.9 years. We detected 15 clusters of excess risk for bladder, breast, cervix, colon, lung, melanoma, oral, ovary, prostate, and soft tissue cancers. An urban area had dense overlap of multiple cancer types, including two EDs at increased risk for five cancer types each.
Conclusions
Early environments may contribute to cancer risk later in life. Life course perspectives applied to the study of cancer incidence can provide insights for increasing understanding of cancer etiology.

Similar content being viewed by others
References
Barker DJ, Martyn CN (1992) The maternal and fetal origins of cardiovascular disease. J Epidemiol Community Health 46(1):8–11
Ben-Shlomo Y, Cooper R, Kuh D (2016) The last two decades of life course epidemiology, and its relevance for research on ageing. Int J Epidemiol 45(4):973–988
Ben-Shlomo Y, Kuh D (2002) A life course approach to chronic disease epidemiology: conceptual models, empirical challenges and interdisciplinary perspectives. Int J Epidemiol 31(2):285–293
Hanson HA, Smith KR, Zimmer Z (2015) Reproductive history and later-life comorbidity trajectories: a medicare-linked cohort study from the Utah Population Database. Demography 52(6):2021–2049
Hanson M, Gluckman P (1758S) Developmental origins of noncommunicable disease: population and public health implications. Am J Clin Nutr 94(6):1754S–1758S
Lekkas P, Paquet C, Howard NJ, Daniel M (2017) Illuminating the lifecourse of place in the longitudinal study of neighbourhoods and health. Soc Sci Med 177:239–247
Estampador AC, Franks PW (2018) Precision medicine in obesity and type 2 diabetes: the relevance of early-life exposures. Clin Chem 64(1):130–141
Fan B, Yang Y, Dayimu A, Zhou G, Liu Y, Li S, Chen W, Zhang T, Xue F (2019) Body mass index trajectories during young adulthood and incident hypertension: a longitudinal cohort in Chinese population. J Am Heart Assoc 8(8):e011937
Pollock BD, Stuchlik P, Harville EW, Mills KT, Tang W, Chen W, Bazzano LA (2019) Life course trajectories of cardiovascular risk: impact on atherosclerotic and metabolic indicators. Atherosclerosis 280:21–27
Rogers NT, Power C, Pinto Pereira SM (2019) Birthweight, lifetime obesity and physical functioning in mid-adulthood: a nationwide birth cohort study. Int J Epidemiol. https://doi.org/10.1093/ije/dyz120
Hansen S, Strøm M, Olsen SF, Dahl R, Hoffmann HJ, Granström C et al (2016) Prenatal exposure to persistent organic pollutants and offspring allergic sensitization and lung function at 20 years of age. Clin Exp Allergy 46(2):329–336
De Rubeis V, Cotterchio M, Smith BT, Griffith LE, Borgida A, Gallinger S, Cleary S, Anderson LN (2019) Trajectories of body mass index, from adolescence to older adulthood, and pancreatic cancer risk; a population-based case–control study in Ontario, Canada. Cancer Causes Control 30(9):955–966
Stroup AM, Herget KA, Hanson HA, Reed DL, Butler JT, Henry KA, Harrell CJ, Sweeney C, Smith KR (2017) Baby boomers and birth certificates: early-life socioeconomic status and cancer risk in adulthood. Cancer Epidemiol Biomark Prev 26(1):75–84
Phillips DIW, Osmond C, Williams ML, Jones A (2017) Air pollution in early life and adult mortality from chronic rheumatic heart disease. Int J Epidemiol 46(4):1107–1114
Yoshida T, Yamauchi H, Fan Sun G (2004) Chronic health effects in people exposed to arsenic via the drinking water: dose–response relationships in review. Toxicol Appl Pharmacol 198(3):243–252
Hanson HA, Hay WW Jr, Tobin JN, Barkin SL, Atkins M, Karagas MR, Dozier AM, Wetmore C, Konstan MW, Heubi JE (2018) Opportunities for life course research through the integration of data across Clinical and Translational Research Institutes. J Clin Transl Sci 2(3):156–162
Kulldorff M (1997) A spatial scan statistic. Commun Statist Theory Methods 26(6):1481–1496
Kulldorff M, Feuer EJ, Miller BA, Freedma LS (1997) Breast cancer clusters in the Northeast United States: a geographic analysis. Am J Epidemiol 146(2):161–170
Chu J, Zhou C, Guo X, Sun J, Xue F, Zhang J, Zilong L, Zhentao F, Aiqiang X (2017) Female breast cancer mortality clusters in Shandong Province, China: a spatial analysis. Sci Rep 7(1):105
The US National Archives and Records Administration (2019) 1940 Census. United States National Archives https://1940census.archives.gov/. Accessed Oct 2019
Kulldorff M (2016) SaTScan user guide version 9.0. https://www.satscan.org.
Kulldorf M, Huang L, Pickle L, Duczmal L (2006) An elliptic spatial scan statistic. Statist Med 25:329–3943
Han J, Zhu L, Kulldorff M, Hostovich S, Stinchcomb DG, Tatalovich Z, Lewis DR, Feuer EJ (2016) Using Gini coefficient to determining optimal cluster reporting sizes for spatial scan statistics. Int J Health Geogr 15(1):27
Ray GT, Kulldorff M, Asgari MM (2016) Geographic clusters of basal cell carcinoma in a Northern California health plan population. JAMA Dermatol 152(11):1218–1224
Konstantinoudis G, Kreis C, Ammann RA, Niggli F, Kuehni CE, Spycher BD, Swiss Paediatric Ongology Group, Swiss National Cohort Study Group (2018) Spatial clustering of childhood cancers in Switzerland: a nationwide study. Cancer Causes Control 29(3):353–362
Dockerty JD, Sharples KJ, Borman B (1999) An assessment of spatial clustering of leukaemias and lymphomas among young people in New Zealand. J Epidemiol Community Health 53(3):154–158
Carpenter DO, Bushkin-Bedient S (2013) Exposure to chemicals and radiation during childhood and risk for cancer later in life. J Adolesc Health 52(5):S9–S21
Meyer TE, Coker AL, Sanderson M, Symanski E (2007) A case-control study of farming and prostate cancer in African-American and Caucasian men. Occup Environ Med 64(3):155–160
Sritharan J, MacLeod J, Harris S, Cole DC, Harris A, Tjepkema M, Peters PA, Demers PA (2018) Prostate cancer surveillance by occupation and industry: the Canadian Census Health and Environment Cohort (CanCHEC). Cancer Med 7(4):1468–1478
Gandini S, Sera F, Cattaruzza MS, Pasquini P, Picconi O, Boyle P, Melchi CF (2005) Meta-analysis of risk factors for cutaneous melanoma: II. Sun exposure. Eur J Cancer 41(1):45–60
Taeger D, Pesch B, Kendzia B, Behrens T, Jöckel K-H, Dahmann D et al (2015) Lung cancer among coal miners, ore miners and quarrymen: smoking-adjusted risk estimates from the synergy pooled analysis of case–control studies. Scand J Work Environ Health 41(5):467–477
CDC (2011) Vital Signs: current cigarette smoking among adults aged %3e= 18 Years—United States, 2005–2010. MMWR Morb Mortal Wkly Rep 60:1207–1212
Alexandrov LB, Kim J, Haradvala NJ, Huang MN, Ng AWT, Wu Y, Boot A et al (2020) The repertoire of mutational signatures in human cancer. Nature 578:95–101
Ahern J, Galea S, Hubbard A, Syme SL (2009) Neighborhood smoking norms modify the relation between collective efficacy and smoking behavior. Drug Alcohol Depend 100(1–2):138–145
Stimpson JP, Hyunsu J, Mukaila RA, Eschbach K (2007) Neighborhood deprivation and health risk behaviors in NHANES III. Am J Health Behav 31(2):215–222
American Cancer Society (2020) Arsenic and cancer risk. American Cancer Society Inc. https://www.cancer.org/cancer/cancer-causes/arsenic.html. Accessed 28 Feb 2020.
Cogliano VJ, Bean R, Straif K, Grosse Y, Lauby-Secretan B, Ghissassi FE et al (2011) Preventable exposures associated with human cancers. J Natl Cancer Inst 103:1827–1839
International Agency for Research on Cancer (IARC) (2014) IARC monographs on the evaluation of carcinogenic risks to humans. Lyon, France
Guo J, Kauppinen T, Kyyrönen HP, Lindbohm ML, Pukkala E (2004) Risk of esophageal, ovarian, testicular, kidney, and bladder cancers and leukemia among finnish workers exposed to diesel or gasoline engine exhaust. Int J Cancer 111:286–292
Mahabir S, Aagaard K, Anderson LM, Herceg Z, Hiatt RA, Hoover RN et al (2012) Challenges and opportunities in research on early-life events/exposures and cancer development later in life. Cancer Causes Control 23(6):983–990
Wray AJD, Minaker LM (2019) Is cancer prevention influenced by the built environment? A multidisciplinary scoping review. Cancer 125(19):3299–3311
Ozonoff A, Jeffery C, Manjourides J, White LF, Pagano M (2007) Effect of spatial resolution on cluster detection: a simulation study. Int J Health Geogr 6:52
Acknowledgments
This study was supported by the National Institutes of Health (1K12HD085852-01).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by CLL, MT, RH, and HAH. The first draft of the manuscript was written by CL and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Leiser, C.L., Taddie, M., Hemmert, R. et al. Spatial clusters of cancer incidence: analyzing 1940 census data linked to 1966–2017 cancer records. Cancer Causes Control 31, 609–615 (2020). https://doi.org/10.1007/s10552-020-01302-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10552-020-01302-3