Abstract
Background
Nuclear receptor 4A1 (NR4A1) is overexpressed in mammary tumors, and the methylene-substituted bis-indole derivative 1,1-bis(3′-indolyl)-1-(p-hydroxyphenyl)methane (DIM-C-pPhOH) acts as an NR4A1 antagonist (inverse agonist) and inhibits NR4A1-regulated pro-oncogenic pathways/genes in breast and other cancer cells.
Methods
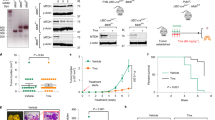
Buttressed analogs of DIM-C-pPhOH were synthesized by condensation of the substituted p-hydroxybenzaldehydes with indole. Breast cancer cell growth, survival, and migration assays were carried out by cell counting, Annexin V staining, and Boyden chamber assays, respectively. Changes in RNA and protein expression were determined by RT-PCR and western blots, respectively. Analysis of RNAseq results was carried out using Ingenuity Pathway Analysis, and in vivo potencies of NR4A1 antagonists were determined in athymic nude mice bearing MDA-MB-231 cells in an orthotopic model.
Results
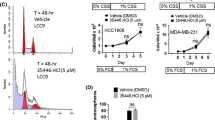
Ingenuity Pathway analysis of common genes modulated by NR4A1 knockdown or treatment with DIM-C-pPhOH showed that changes in gene expression were consistent with the observed decreased functional responses, namely inhibition of growth and migration and increased apoptosis. DIM-C-pPhOH is rapidly metabolized and the effects and potencies of buttressed analogs of DIM-C-pPhOH which contain one or two substituents ortho to the hydroxyl groups were investigated using NR4A1-regulated gene/gene products as endpoints. The buttressed analogs were more potent than DIM-C-pPhOH in both in vitro assays and as inhibitors of mammary tumor growth. Moreover, using 1,1-bis(3′-indolyl)-1-(3-chloro-4-hydroxy-5-methoxyphenyl)methane (DIM-C-pPhOh-3-Cl-5-OCH3) significant tumor growth inhibition was observed at doses as low as 2 mg/kg/d which was at least an order of magnitude more potent than DIM-C-pPhOH.
Conclusions
These buttressed analogs represent a more potent set of second generation NR4A1 antagonists as inhibitors of breast cancer.






Similar content being viewed by others
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- C-DIMs:
-
Methylene-substituted DIMs
- DIM-C-pPhOH:
-
1,1-bis(3′-indolyl)-1-(p-hydroxyphenyl)methane
- DIM-C-pPhOh-3-Cl-5-OCH3 :
-
1,1-bis(3′-indolyl)-1-(3-chloro-4-hydroxy-5-methoxyphenyl)methane
- DMSO:
-
Dimethyl sulfoxide
- EGFR:
-
Epidermal growth factor receptor
- NR4A1:
-
Nuclear receptor 4A1
- SERMs:
-
Selective estrogen receptor modulators
References
Santos R, Ursu O, Gaulton A, Bento AP, Donadi RS, Bologa CG, Karlsson A, Al-Lazikani B, Hersey A, Oprea TI, Overington JP (2017) A comprehensive map of molecular drug targets. Nat Rev Drug Discov 16(1):19–34. https://doi.org/10.1038/nrd.2016.230
Schierle S, Merk D (2018) Development of nuclear receptor modulators. In: Mavromoustakos T, Kellici TF (eds) Rational drug design: methods and protocols, vol 1824. Methods in molecular biology. Springer, New york, pp 245–260
Jordan VC (2003) Antiestrogens and selective estrogen receptor modulators as multifunctional medicines. 2. Clinical considerations and new agents. J Med Chem 46(7):1081–1111. https://doi.org/10.1021/jm020450x
Jordan VC (2007) SERMs: meeting the promise of multifunctional medicines. J Natl Cancer Inst 99(5):350–356. https://doi.org/10.1093/jnci/djk062
Jeong Y, Xie Y, Xiao G, Behrens C, Girard L, Wistuba II, Minna JD, Mangelsdorf DJ (2010) Nuclear receptor expression defines a set of prognostic biomarkers for lung cancer. PLoS Med 7(12):e1000378. https://doi.org/10.1371/journal.pmed.1000378
Holbeck S, Chang J, Best AM, Bookout AL, Mangelsdorf DJ, Martinez ED (2010) Expression profiling of nuclear receptors in the NCI60 cancer cell panel reveals receptor-drug and receptor-gene interactions. Mol Endocrinol 24(6):1287–1296. https://doi.org/10.1210/me.2010-0040
Muscat GE, Eriksson NA, Byth K, Loi S, Graham D, Jindal S, Davis MJ, Clyne C, Funder JW, Simpson ER, Ragan MA, Kuczek E, Fuller PJ, Tilley WD, Leedman PJ, Clarke CL (2013) Research resource: nuclear receptors as transcriptome: discriminant and prognostic value in breast cancer. Mol Endocrinol 27(2):350–365. https://doi.org/10.1210/me.2012-1265
Lee SO, Andey T, Jin UH, Kim K, Singh M, Safe S (2012) The nuclear receptor TR3 regulates mTORC1 signaling in lung cancer cells expressing wild-type p53. Oncogene 31(27):3265–3276. https://doi.org/10.1038/onc.2011.504
Cho SD, Yoon K, Chintharlapalli S, Abdelrahim M, Lei P, Hamilton S, Khan S, Ramaiah SK, Safe S (2007) Nur77 agonists induce proapoptotic genes and responses in colon cancer cells through nuclear receptor-dependent and nuclear receptor-independent pathways. Cancer Res 67(2):674–683. https://doi.org/10.1158/0008-5472.can-06-2907
Wang JR, Gan WJ, Li XM, Zhao YY, Li Y, Lu XX, Li JM, Wu H (2014) Orphan nuclear receptor Nur77 promotes colorectal cancer invasion and metastasis by regulating MMP-9 and E-cadherin. Carcinogenesis 35(11):2474–2484. https://doi.org/10.1093/carcin/bgu157
Zhou F, Drabsch Y, Dekker TJ, de Vinuesa AG, Li Y, Hawinkels LJ, Sheppard KA, Goumans MJ, Luwor RB, de Vries CJ, Mesker WE, Tollenaar RA, Devilee P, Lu CX, Zhu H, Zhang L, Dijke PT (2014) Nuclear receptor NR4A1 promotes breast cancer invasion and metastasis by activating TGF-beta signalling. Nat Commun 5:3388. https://doi.org/10.1038/ncomms4388
Wu H, Bi J, Peng Y, Huo L, Yu X, Yang Z, Zhou Y, Qin L, Xu Y, Liao L, Xie Y, Conneely OM, Jonkers J, Xu J (2017) Nuclear receptor NR4A1 is a tumor suppressor down-regulated in triple-negative breast cancer. Oncotarget 8(33):54364–54377. https://doi.org/10.18632/oncotarget.17532
Alexopoulou AN, Leao M, Caballero OL, Da Silva L, Reid L, Lakhani SR, Simpson AJ, Marshall JF, Neville AM, Jat PS (2010) Dissecting the transcriptional networks underlying breast cancer: NR4A1 reduces the migration of normal and breast cancer cell lines. Breast Cancer Res 12(4):R51. https://doi.org/10.1186/bcr2610
Hedrick E, Lee SO, Doddapaneni R, Singh M, Safe S (2015) Nuclear receptor 4A1 as a drug target for breast cancer chemotherapy. Endocr Relat Cancer 22(5):831–840. https://doi.org/10.1530/erc-15-0063
Hedrick E, Lee SO, Doddapaneni R, Singh M, Safe S (2016) NR4A1 antagonists inhibit β1-integrin-dependent breast cancer cell migration. Mol Cell Biol 36(9):1383–1394. https://doi.org/10.1128/mcb.00912-15
Hedrick E, Safe S (2017) Transforming growth factor beta/NR4A1-inducible breast cancer cell migration and epithelial-to-mesenchymal transition Is p38alpha (mitogen-activated protein kinase 14) dependent. Mol Cell Biol 37(18):e00306–e00317. https://doi.org/10.1128/mcb.00306-17
Hedrick E, Lee SO, Kim G, Abdelrahim M, Jin UH, Safe S, Abudayyeh A (2015) Nuclear receptor 4A1 (NR4A1) as a drug target for renal cell adenocarcinoma. PLoS ONE 10(6):e0128308. https://doi.org/10.1371/journal.pone.0128308
Lacey A, Rodrigues-Hoffman A, Safe S (2017) PAX3-FOXO1A expression in rhabdomyosarcoma is driven by the targetable nuclear receptor NR4A1. Cancer Res 77(3):732–741. https://doi.org/10.1158/0008-5472.can-16-1546
Lee SO, Jin UH, Kang JH, Kim SB, Guthrie AS, Sreevalsan S, Lee JS, Safe S (2014) The orphan nuclear receptor NR4A1 (Nur77) regulates oxidative and endoplasmic reticulum stress in pancreatic cancer cells. Mol Cancer Res 12(4):527–538. https://doi.org/10.1158/1541-7786.mcr-13-0567
Lee SO, Li X, Hedrick E, Jin UH, Tjalkens RB, Backos DS, Li L, Zhang Y, Wu Q, Safe S (2014) Diindolylmethane analogs bind NR4A1 and are NR4A1 antagonists in colon cancer cells. Mol Endocrinol 28(10):1729–1739. https://doi.org/10.1210/me.2014-1102
Lee SO, Abdelrahim M, Yoon K, Chintharlapalli S, Papineni S, Kim K, Wang H, Safe S (2010) Inactivation of the orphan nuclear receptor TR3/Nur77 inhibits pancreatic cancer cell and tumor growth. Cancer Res 70(17):6824–6836. https://doi.org/10.1158/0008-5472.can-10-1992
Mohankumar K, Lee J, Wu CS, Sun Y, Safe S (2018) Bis-indole-derived NR4A1 ligands and metformin exhibit NR4A1-dependent glucose metabolism and uptake in C2C12 cells. Endocrinology 159(5):1950–1963. https://doi.org/10.1210/en.2017-03049
De Miranda BR, Miller JA, Hansen RJ, Lunghofer PJ, Safe S, Gustafson DL, Colagiovanni D, Tjalkens RB (2013) Neuroprotective efficacy and pharmacokinetic behavior of novel anti-inflammatory para-phenyl substituted diindolylmethanes in a mouse model of Parkinson’s disease. J Pharmacol Exp Ther 345(1):125–138. https://doi.org/10.1124/jpet.112.201558
Bichlmaier I, Finel M, Sippl W, Yli-Kauhaluoma J (2007) Stereochemical and steric control of the UDP-glucuronosyltransferase-catalyzed conjugation reaction: a rational approach for the design of inhibitors for the human UGT2B7. ChemMedChem 2(12):1730–1740. https://doi.org/10.1002/cmdc.200700122
Dobin A, Davis CA, Schlesinger F, Drenkow J, Zaleski C, Jha S, Batut P, Chaisson M, Gingeras TR (2013) STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29(1):15–21. https://doi.org/10.1093/bioinformatics/bts635
Robinson MD, McCarthy DJ, Smyth GK (2010) edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26(1):139–140. https://doi.org/10.1093/bioinformatics/btp616
Francis PA, Pagani O, Fleming GF, Walley BA, Colleoni M, Lang I, Gomez HL, Tondini C, Ciruelos E, Burstein HJ, Bonnefoi HR, Bellet M, Martino S, Geyer CE Jr, Goetz MP, Stearns V, Pinotti G, Puglisi F, Spazzapan S, Climent MA, Pavesi L, Ruhstaller T, Davidson NE, Coleman R, Debled M, Buchholz S, Ingle JN, Winer EP, Maibach R, Rabaglio-Poretti M, Ruepp B, Di Leo A, Coates AS, Gelber RD, Goldhirsch A, Regan MM, Soft, Investigators T, the International Breast Cancer Study G (2018) Tailoring adjuvant endocrine therapy for premenopausal breast cancer. N Engl J Med 379(2):122–137. https://doi.org/10.1056/nejmoa1803164
Sledge GW, Mamounas EP, Hortobagyi GN, Burstein HJ, Goodwin PJ, Wolff AC (2014) Past, present, and future challenges in breast cancer treatment. J Clin Oncol 32(19):1979–1986. https://doi.org/10.1200/jco.2014.55.4139
Hedrick E, Lee SO, Safe S (2017) The nuclear orphan receptor NR4A1 regulates beta1-integrin expression in pancreatic and colon cancer cells and can be targeted by NR4A1 antagonists. Mol Carcinog. https://doi.org/10.1002/mc.22662
Hedrick E, Li X, Safe S (2017) Penfluridol represses integrin expression in breast cancer through induction of reactive oxygen species and downregulation of Sp transcription factors. Mol Cancer Ther 16(1):205–216. https://doi.org/10.1158/1535-7163.mct-16-0451
Acknowledgement
The financial assistance of the Sid Kyle Chair endowment, Texas AgriLife and the National Institutes of Health (P30-ES023512, S. Safe) is gratefully acknowledged.
Author information
Authors and Affiliations
Contributions
SS conceived the study and wrote the manuscript. EH, XL, YC, AL and KM were involved with collection of data. EH, XL, YC, AL, KM and MZ carried out all data analysis. EH, XL, YC, AL, KM, MZ and SS were all involved in data interpretation, critical revisions of the manuscript, and approval of the final version.
Corresponding author
Ethics declarations
Conflict of interest
The authors do not have any conflicts of interests.
Ethical approval
The Texas A&M University Institutional Animal Care and Use Committee reviewed and approved our animal treatment and use protocols.
Research involving human and animal participants
This article does not contain any studies with human participants performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hedrick, E., Li, X., Cheng, Y. et al. Potent inhibition of breast cancer by bis-indole-derived nuclear receptor 4A1 (NR4A1) antagonists. Breast Cancer Res Treat 177, 29–40 (2019). https://doi.org/10.1007/s10549-019-05279-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-019-05279-9