Abstract
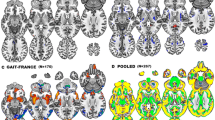
Greater gait variability predicts dementia. However, little is known about the neural correlates of gait variability. The aims of this study were to determine (1) grey matter volume covariance patterns associated with gait variability and (2) whether these patterns were associated with specific cognitive domains. Participants (n = 351; mean age 71.9 ± 7.1) were randomly selected from the Southern Tasmanian electoral roll. Step time, step length, step width and double support time were measured using an electronic walkway. Gait variability was calculated as the standard deviation of all steps for each gait measure. Voxel-based morphometry and multivariate covariance-based analyses were used to identify grey matter patterns associated with each gait variability measure. The individual expressions of grey matter patterns were correlated with processing speed, memory, executive and visuospatial functions. The grey matter covariance pattern of double support time variability included frontal, medial temporal, anterior cingulate, insula, cerebellar and striatal regions. Greater expression of this pattern was correlated with poorer performance in all cognitive functions (p < 0.001). The covariance pattern of step length variability included frontal, temporal, insula, occipital and cerebellar regions and was correlated with all cognitive functions (p < 0.05), except memory (p = 0.76). The covariance pattern of step width variability was limited to the cerebellum and correlated only with memory (p = 0.047). No significant pattern was identified for step time variability. In conclusion, different grey matter covariance patterns were associated with individual gait variability measures. These patterns were also correlated with specific cognitive functions, suggesting common neural networks may underlie both gait and cognition.

Similar content being viewed by others
Data Availability
Data will be available upon request to the corresponding author.
References
Allali G, Montembeault M, Brambati SM, Bherer L, Blumen HM, Launay CP et al (2018) Brain structure covariance associated with gait control in aging. J Gerontol Ser A. https://doi.org/10.1093/gerona/gly123
Annweiler C, Beauchet O, Bartha R, Wells JL, Borrie MJ, Hachinski V, Montero-Odasso M (2013) Motor cortex and gait in mild cognitive impairment: a magnetic resonance spectroscopy and volumetric imaging study. Brain 136(3):859–871. https://doi.org/10.1093/brain/aws373
Ashburner J (2007) A fast diffeomorphic image registration algorithm. Neuroimage 38(1):95–113. https://doi.org/10.1016/j.neuroimage.2007.07.007
Ashburner J, Friston KJ (2005) Unified segmentation. Neuroimage 26(3):839–851. https://doi.org/10.1016/j.neuroimage.2005.02.018
Ashburner J, Barnes G, Chen C, Daunizeau J, Flandin G, Friston K et al (2014) SPM12 manual. Wellcome Trust Centre for Neuroimaging, London
Baars B, Gage NM (2012) Fundamentals of cognitive neuroscience: a beginner’s guide, 2nd edn. Academic Press, Cambridge
Beauchet O, Annweiler C, Celle S, Bartha R, Barthélémy J-C, Roche F (2014) Higher gait variability is associated with decreased parietal gray matter volume among healthy older adults. Brain Topogr 27(2):293–295. https://doi.org/10.1007/s10548-013-0293-y
Beauchet O, Launay CP, Annweiler C, Allali G (2015) Hippocampal volume, early cognitive decline and gait variability: which association? Exp Gerontol 61:98–104. https://doi.org/10.1016/j.exger.2014.11.002
Beauchet O, Launay CP, Sekhon H, Montembeault M, Allali G (2019) Association of hippocampal volume with gait variability in pre-dementia and dementia stages of Alzheimer disease: results from a cross-sectional study. Exp Gerontol 115:55–61. https://doi.org/10.1016/j.exger.2018.11.010
Blumen HM, Verghese J (2018) Gray matter volume covariance networks associated with social networks in older adults. Soc Neurosci. https://doi.org/10.1080/17470919.2018.1535999
Blumen HM, Allali G, Beauchet O, Lipton RB, Verghese J (2018a) A gray matter volume covariance network associated with the motoric cognitive risk syndrome: a multi-cohort MRI study. J Gerontol A Biol Sci Med Sci. https://doi.org/10.1016/j.jalz.2017.06.2401
Blumen HM, Brown LL, Habeck C, Allali G, Ayers E, Beauchet O et al (2018b) Gray matter volume covariance patterns associated with gait speed in older adults: a multi-cohort MRI study. Brain Imaging Behav. https://doi.org/10.1007/s11682-018-9871-7
Brach JS, Studenski SA, Perera S, VanSwearingen JM, Newman AB (2007) Gait variability and the risk of incident mobility disability in community-dwelling older adults. J Gerontol A Biol Sci Med Sci 62(9):983–988. https://doi.org/10.1093/gerona/62.9.983
Brach JS, Studenski S, Perera S, VanSwearingen JM, Newman AB (2008) Stance time and step width variability have unique contributing impairments in older persons. Gait Posture 27(3):431–439. https://doi.org/10.1016/j.gaitpost.2007.05.016
Burgess N, Maguire EA, O’Keefe J (2002) The human hippocampus and spatial and episodic memory. Neuron 35(4):625–641. https://doi.org/10.1016/S0896-6273(02)00830-9
Bush G, Luu P, Posner MI (2000) Cognitive and emotional influences in anterior cingulate cortex. Trends Cogn Sci 4(6):215–222. https://doi.org/10.1016/S1364-6613(00)01483-2
Callisaya ML, Blizzard L, McGinley JL, Schmidt MD, Srikanth VK (2009) Sensorimotor factors affecting gait variability in older people—a population-based study. J Gerontol Ser A Biomed Sci Med Sci 65(4):386–392. https://doi.org/10.1093/gerona/glp184
Callisaya ML, Blizzard L, Schmidt MD, McGinley JL, Srikanth VK (2010) Ageing and gait variability—a population-based study of older people. Age Ageing 39(2):191–197. https://doi.org/10.1093/ageing/afp250
Callisaya ML, Blizzard L, Schmidt MD, Martin KL, McGinley JL, Sanders LM, Srikanth VK (2011) Gait, gait variability and the risk of multiple incident falls in older people: a population-based study. Age Ageing 40(4):481–487. https://doi.org/10.1093/ageing/afr055
Callisaya ML, Beare R, Phan TG, Chen J, Srikanth VK (2014) Global and regional associations of smaller cerebral gray and white matter volumes with gait in older people. PLoS ONE 9(1):e84909. https://doi.org/10.1371/journal.pone.0084909
Dietz V (2003) Spinal cord pattern generators for locomotion. Clin Neurophysiol 114(8):1379–1389. https://doi.org/10.1016/S1388-2457(03)00120-2
Efron B, Tibshirani RJ (1994) An introduction to the bootstrap. CRC Press, Boca Raton
Habeck C, Stern Y (2007) Neural network approaches and their reproducibility in the study of verbal working memory and Alzheimer’s disease. Clin Neurosci Res 6(6):381–390. https://doi.org/10.1016/j.cnr.2007.05.004
Hausdorff JM (2007) Gait dynamics, fractals and falls: finding meaning in the stride-to-stride fluctuations of human walking. Hum Mov Sci 26(4):555–589. https://doi.org/10.1016/j.humov.2007.05.003
Holtzer R, Wang C, Verghese J (2012) The relationship between attention and gait in aging: facts and fallacies. Mot Control 16(1):64–80. https://doi.org/10.1123/mcj.16.1.64
Jahn K, Deutschländer A, Stephan T, Strupp M, Wiesmann M, Brandt T (2004) Brain activation patterns during imagined stance and locomotion in functional magnetic resonance imaging. Neuroimage 22(4):1722–1731. https://doi.org/10.1016/j.neuroimage.2004.05.017
Jayakody O, Breslin M, Srikanth V, Callisaya ML (2018) Medical, sensorimotor and cognitive factors associated with gait variability: a longitudinal population-based study. Front Aging Neurosci 10:419. https://doi.org/10.3389/fnagi.2018.00419
Jayakody O, Breslin M, Srikanth V, Callisaya M (2019) Gait characteristics and cognitive decline: a longitudinal population-based study. J Alzheimer’s Dis (Preprint). https://doi.org/10.3233/JAD-181157
Kang HG, Dingwell JB (2008) Separating the effects of age and walking speed on gait variability. Gait Posture 27(4):572–577. https://doi.org/10.1016/j.gaitpost.2007.07.009
Kikkert LH, Vuillerme N, van Campen JP, Hortobagyi T, Lamoth CJ (2016) Walking ability to predict future cognitive decline in old adults: a scoping review. Ageing Res Rev 27:1–14. https://doi.org/10.1016/j.arr.2016.02.001
La Fougere C, Zwergal A, Rominger A, Förster S, Fesl G, Dieterich M et al (2010) Real versus imagined locomotion: a [18F]-FDG PET-fMRI comparison. Neuroimage 50(4):1589–1598. https://doi.org/10.1016/j.neuroimage.2009.12.060
Lord S, Galna B, Verghese J, Coleman S, Burn D, Rochester L (2012) Independent domains of gait in older adults and associated motor and nonmotor attributes: validation of a factor analysis approach. J Gerontol Ser A Biomed Sci Med Sci 68(7):820–827. https://doi.org/10.1093/gerona/gls255
Martin KL, Blizzard L, Wood AG, Srikanth V, Thomson R, Sanders LM, Callisaya ML (2012) Cognitive function, gait, and gait variability in older people: a population-based study. J Gerontol Ser A Biomed Sci Med Sci 68(6):726–732. https://doi.org/10.1093/gerona/gls224
Menon V, Uddin LQ (2010) Saliency, switching, attention and control: a network model of insula function. Brain Struct Funct 214(5–6):655–667. https://doi.org/10.1007/s00429-010-0262-0
Miller EK, Cohen JD (2001) An integrative theory of prefrontal cortex function. Annu Rev Neurosci 24(1):167–202. https://doi.org/10.1146/annurev.neuro.24.1.167
Modarresi S, Divine A, Grahn JA, Overend TJ, Hunter SW (2018) Gait parameters and characteristics associated with increased risk of falls in people with dementia: a systematic review. Int Psychogeriatr. https://doi.org/10.1017/S1041610218001783
Morris R, Lord S, Bunce J, Burn D, Rochester L (2016) Gait and cognition: mapping the global and discrete relationships in ageing and neurodegenerative disease. Neurosci Biobehav Rev 64:326–345. https://doi.org/10.1016/j.neubiorev.2016.02.012
Morton SM, Bastian AJ (2004) Cerebellar control of balance and locomotion. Neuroscientist 10(3):247–259. https://doi.org/10.1177/1073858404263517
Palliyath S, Hallett M, Thomas SL, Lebiedowska MK (1998) Gait in patients with cerebellar ataxia. Mov Disord Off J Mov Disord Soc 13(6):958–964. https://doi.org/10.1002/mds.870130616
Pieruccini-Faria F, Black SE, Masellis M, Smith EE, Almeida QJ, Li KZ et al (2021) Gait variability across neurodegenerative and cognitive disorders: results from the Canadian Consortium of Neurodegeneration in Aging (CCNA) and the Gait and Brain Study. Alzheimer’s & Dement. https://doi.org/10.1002/alz.12298
Rosano C, Brach J, Studenski S, Longstreth W Jr., Newman AB (2007) Gait variability is associated with subclinical brain vascular abnormalities in high-functioning older adults. Neuroepidemiology 29(3–4):193–200. https://doi.org/10.1159/000111582
Rosso AL, Studenski SA, Chen WG, Aizenstein HJ, Alexander NB, Bennett DA et al (2013) Aging, the central nervous system, and mobility. J Gerontol Ser A Biomed Sci Med Sci 68(11):1379–1386. https://doi.org/10.1093/gerona/glt089
Schmahmann JD (1997) The cerebellum and cognition, vol 41. Academic Press, Cambridge
Schmahmann JD, Caplan D (2006) Cognition, emotion and the cerebellum. Brain 129(2):290–292. https://doi.org/10.1093/brain/awh729
Srikanth V, Beare R, Blizzard L, Phan T, Stapleton J, Chen J et al (2009) Cerebral white matter lesions, gait, and the risk of incident falls. Stroke 40(1):175–180. https://doi.org/10.1161/STROKEAHA.108.524355
Tripathi S, Verghese J, Blumen HM (2019) Gray matter volume covariance networks associated with dual-task cost during walking-while-talking. Hum Brain Mapp. https://doi.org/10.1002/hbm.24520
Verghese J, Wang C, Lipton RB, Holtzer R, Xue X (2007) Quantitative gait dysfunction and risk of cognitive decline and dementia. J Neurol Neurosurg Psychiatry 78(9):929–935. https://doi.org/10.1136/jnnp.2006.106914
Zimmerman ME, Lipton RB, Pan JW, Hetherington HP, Verghese J (2009) MRI-and MRS-derived hippocampal correlates of quantitative locomotor function in older adults. Brain Res 1291:73–81. https://doi.org/10.1016/j.brainres.2009.07.043
Zwergal A, Linn J, Xiong G, Brandt T, Strupp M, Jahn K (2012) Aging of human supraspinal locomotor and postural control in fMRI. Neurobiol Aging 33(6):1073–1084. https://doi.org/10.1016/j.neurobiolaging.2010.09.022
Acknowledgements
Study participants: TASCOG research staff and volunteers. The computational resources provided by the Australian Government through MASSIVE under the National Computational Merit Allocation Scheme.
Funding
National Health and Medical Research Council (Grant Nos. 403000BH and 491109). Physiotherapy Research Foundation (Grant No. BH036/05). Perpetual Trustees. Brain Foundation. Royal Hobart Hospital Research Foundation (Grant No. 341 M). ANZ Charitable Trust. Masonic Centenary Medical Research Foundation. VS is funded by NHMRC Practitioner Fellowship (APP1137837). MC is funded by NHMRC Boosting Dementia Research Leadership Fellowship (1135761).
Author information
Authors and Affiliations
Contributions
Author contributions included conception and study design (VS, MC), data collection or acquisition (VS,MC), statistical analysis (OJ, HB, RB, MC), interpretation of results (OJ, HB, RB, MC), drafting the manuscript work or revising it critically for important intellectual content (OJ, HB, RB, MC, MB, VS) and approval of final version to be published and agreement to be accountable for the integrity and accuracy of all aspects of the work (All authors).
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that he/she has no conflict of interest.
Ethical Approval
All procedures performed in this study were in accordance with the ethical standards of the Southern Tasmanian Health and Medical Human Research Ethics committee and with the 1964 Helsinki declaration and its amendments.
Informed Consent
Informed written consent was obtained from all participants.
Additional information
Handling Editor: Glenn Wylie.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jayakody, O., Breslin, M., Beare, R. et al. The Associations Between Grey Matter Volume Covariance Patterns and Gait Variability—The Tasmanian Study of Cognition and Gait. Brain Topogr 34, 478–488 (2021). https://doi.org/10.1007/s10548-021-00841-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10548-021-00841-5