Abstract
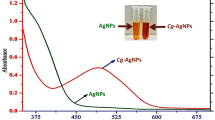
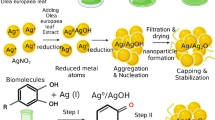
The ‘sacred leaf’ or “Hoja Santa” (Piper auritum Kunth) has a great value for Mexican culture and has gained popularity worldwide for its excellent properties from culinary to remedies. To contribute to its heritage, in this project we proposed the green synthesis of silver oxide nanoparticles (Ag2O NPs) using an extract of “Hoja Santa” (Piper auritum) as a reducing and stabilizing agent. The synthesized Ag2O NPs were characterized by UV–Visible spectroscopy (plasmon located at 405 nm), X-ray diffraction (XRD) (particle size diameter of 10 nm), scanning electron microscopy (SEM) (particle size diameter of 13.62 ± 4.61 nm), and Fourier-transform infrared spectroscopy (FTIR) (functional groups from “Hoja Santa” attached to nanoparticles). Antioxidant capacity was evaluated using DPPH, ABTS and FRAP methods. Furthermore, the antimicrobial activity of NPs against a panel of clinically relevant bacterial strains, including both Gram-positive (Staphylococcus aureus) and Gram-negative bacteria (Salmonella Enteritidis and Escherichia coli O157:H7), was over 90% at concentrations of 200 µg/mL. Additionally, we assessed the antibiofilm activity of the NPs against Pseudomonas aeruginosa (reaching 98% of biofilm destruction at 800 µg/mL), as biofilm formation plays a crucial role in bacterial resistance and chronic infections. Moreover, we investigated the impact of Ag2O NPs on immune cell viability, respiratory burst, and phagocytic activity to understand their effects on the immune system.









Similar content being viewed by others
References
Aguilar-Urquizo E, Itza-Ortiz MF, Sangines-Garcia JR, Pineiro-Vázquez AT, Reyes-Ramirez A, Pinacho-Santana B (2020) Phytobiotic activity of Piper auritum and Ocimum basilicum on avian E. coli. Braz J Poult Sci https://doi.org/10.1590/1806-9061-2019-1167
Ahmadi F (2012) Impact of different levels of silver nanoparticles (Ag-NPs) on performance, oxidative enzymes and blood parameters in broiler chicks. Pak Vet J 26(3):325–328
Akar Z, Akay S, Ejder N, Düzgün AO (2023) Determination of the cytotoxicity and antibiofilm potential effect of Equisetum arvense silver nanoparticles. Appl Biochem Biotechnol. https://doi.org/10.1007/s12010-023-04587-7
Al-Rajhi AMH, Salem SS, Alharbi AA, Abdelghany TM (2022) Ecofriendly synthesis of silver nanoparticles using Kei-apple (Dovyalis caffra) fruit and their efficacy against cancer cells and clinical pathogenic microorganisms. Arab J Chem 15(7):03927. https://doi.org/10.1016/j.arabjc.2022.103927
Alharthi AF, Gouda M, Khalaf MM, Elmushyakhi A, Abou Taleb MF, Abd El-Lateef HM (2023) Cellulose-acetate-based films modified with Ag2O and ZnS as nanocomposites for highly controlling biological behavior for wound. Healing Appl Mater 16:777. https://doi.org/10.3390/ma16020777
Ali F, Younas U, Nazir A, Hassan F, Iqbal M, Hamza B, Mukhtar S, Khalid A, Ishfaq A (2022) Biosynthesis and characterization of silver nanoparticles using strawberry seed extract and evaluation of their antibacterial and antioxidant activities. J Saudi Chem Soc 26(6):101558. https://doi.org/10.1016/j.jscs.2022.101558
Amaliyah S, Sabarudin A, Masruri M, Sumitro SB (2022) Characterization and antibacterial application of biosynthesized silver nanoparticles using Piper retrofractum vahl fruit extract as bioreductor. J Appl Pharm Sci 12(3):103–114. https://doi.org/10.7324/JAPS.2022.120311
Angulo A, Maldonado M, Delgado K, Reyes-Becerril M (2017) Debaryomyces hansenii up regulates superoxide dismutase gene expression and enhances the immune response and survival in pacific red snapper (Lutjanus peru) leukocytes after Vibrio parahaemolyticus infection. Dev Comp Immunol 71:18–27. https://doi.org/10.1016/j.dci.2017.01.020
Awad MA, Hendi AA, Ortashi KMO, Alanazi AB, ALZahrani BA, Soliman DA (2019) Greener synthesis, characterization, and antimicrobiological effects of Helba silver nanoparticle-PMMA nanocomposite. Int J Polym Sci 4379507:7. https://doi.org/10.1155/2019/4379507
Ayromlou A, Masoudi S, Mirzaie A (2019) Scorzonera calyculata aerial part extract mediated synthesis of silver nanoparticles: evaluation of their antibacterial, antioxidant and anticancer activities. J Clust Sci 30:1037–1050. https://doi.org/10.1007/s10876-019-01563-2
Banua J, Han JI (2020) Biogenesis of prism-like silver oxide nanoparticles using nappa cabbage extract and their p-nitrophenol sensing activity. Molecules 25(10):2298. https://doi.org/10.3390/molecules25102298
Benzie IF, Strain JJ (1996) The ferric reducing ability of plasma (FRAP) as a measure of antioxidant power: the FRAP assay. Anal Biochem 239(1):70–76. https://doi.org/10.1006/abio.1996.0292
Bhakya S, Muthukrishnan S, Sukumaran M, Muthukumar M (2022) Biogenic synthesis of silver nanoparticles and their antioxidant and antibacterial activity. Appl Nanosci 6:755–766. https://doi.org/10.1007/s13204-015-0473-z
Brand-Williams W, Cuvelier ME, Berset C (1995) Use of a free radical method to evaluate antioxidant activity. LWT-Food Sci Technol 28(1):25–30. https://doi.org/10.1016/S0023-6438(95)80008-5
Castorena-Sánchez A, Velázquez-Carriles CA, López-Álvarez MA, Serrano-Niño JC, Cavazos-Garduño A, Garay-Martínez LE, Silva-Jara JM (2023) Magnesium nanohydroxide (2D brucite) as a host matrix for thymol and carvacrol: synthesis, characterization, and inhibition of foodborne pathogens. Green Proc Synth 12(1):20230145. https://doi.org/10.1515/gps-2023-0145
Chacón C, Bojórquez-Quintal E, Caamal-Chan G, Ruíz-Valdiviezo VM, Montes-Molina JA, Garrido-Ramírez ER, Rojas-Abarca LM, Ruiz-Lau N (2021) In vitro antifungal activity and chemical composition of Piper auritum Kunth essential oil against Fusarium oxysporum and Fusarium equiseti. Agronomy 11(6):1098. https://doi.org/10.3390/agronomy11061098
Corciova A, Mircea C, Burlec AF, Cioanca O, Tuchilus C, Fifere A, Lungoci AL, Marangoci N, Hancianu M (2019) Antioxidant, antimicrobial and photocatalytic activities of silver nanoparticles obtained by bee propolis extract assisted biosynthesis. Farmacia 67(3):482–489. https://doi.org/10.31925/farmacia.2019.3.16
Dawadi S, Katuwal S, Gupta A, Lamichhane U, Thapa R, Jaisi S, Lamichhane G, Bhattarai DP, Parajuli N (2021) Current research on silver nanoparticles: synthesis, characterization, and applications. J Nanomater. https://doi.org/10.1155/2021/6687290
de Lacerda Coriolano D, Souza JB, Bueno Ev, Medeiros SMDFRDS, Cavalcanti IDL, Cavalcanti IMF (2021) Antibacterial and antibiofilm potential of silver nanoparticles against antibiotic-sensitive and multidrug-resistant Pseudomonas aeruginosa strains. Braz J Microbiol 52:267–278. https://doi.org/10.1007/s42770-020-00406-x
Dewi FRP, Lim V, Rosyidah AA, Fatimah, Wahyuningsih SPA, Zubaidah U (2023) Characterization of silver nanoparticles (AgNPs) synthesized from Piper ornatum leaf extract and its activity against food borne pathogen Staphylococcus aureus. Biodivers J Biol Divers 24(3):1742–1748. https://doi.org/10.13057/biodiv/d240348
Dharmaraj D, Krishnamoorthy M, Rajendran K, Karuppiah K, Annamalai J, Durairaj KR, Santhiyagu P, Ethiraj K (2021) Antibacterial and cytotoxicity activities of biosynthesized silver oxide (Ag2O) nanoparticles using Bacillus paramycoides. J Drug Deliv Sci Technol 61:102111. https://doi.org/10.1016/j.jddst.2020.102111
Dhoondia ZH, Chakraborty H (2012) Lactobacillus mediated synthesis of silver oxide nanoparticles. Nanomater Nanotechnol 2(15):1. https://doi.org/10.5772/55741
Essghaier B, Ben Khedher G, Hannachi H, Dridi R, Zid MF, Chaffei C (2022) Green synthesis of silver nanoparticles using mixed leaves aqueous extract of wild olive and pistachio: characterization, antioxidant, antimicrobial and effect on virulence factors of Candida. Arch Microbiol 204(4):203. https://doi.org/10.1007/s00203-022-02810-3
Fowsiya J, Madhumitha G (2019) Biomolecules derived from Carissa edulis for the microwave assisted synthesis of Ag2O nanoparticles: A study against S. incertulas, C. medinalis and S. mauritia. J Clust Sci 30(5):1243–1252. https://doi.org/10.1007/s10876-019-01627-3
Hetta HF, Al-Kadmy IMS, Khazaal SS, Abbas S, Suhail A, El-Mokhtar MA, Ellah NHA, Ahmed EA, Abd-ellatief RB, El-Masry EA, Batiha GES, Elkady AA, Mohamed NA, Algammal AM (2021) Antibiofilm and antivirulence potential of silver nanoparticles against multidrug-resistant Acinetobacter baumannii. Sci Rep 11:10751. https://doi.org/10.1038/s41598-021-90208-4
Ibrahim S, Ahmad Z, Manzoor MZ, Mujahid M, Faheem Z, Adnan A (2021) Optimization for biogenic microbial synthesis of silver nanoparticles through response surface methodology, characterization, their antimicrobial, antioxidant, and catalytic potential. Sci Rep 11(1):770. https://doi.org/10.1038/s41598-020-80805-0
Jabbar AH, Al-Janabi HSO, Hamzah MQ, Mezan SO, Tumah AN, Ameruddin ASB, Agam MA (2020) Green synthesis and characterization of silver nanoparticle (AgNPs) using Pandanus atrocarpus extract. Int J Adv Sci Technol 29(3):4913–4922
Jaswal T, Gupta J (2023) A review on the toxicity of silver nanoparticles on human health. Mater Today Proc 81(2):859–863. https://doi.org/10.1016/j.matpr.2021.04.266
Kanniah P, Chelliah P, Thangapandi JR, Gnanadhas G, Mahendran V, Robert M (2021) Green synthesis of antibacterial and cytotoxic silver nanoparticles by Piper nigrum seed extract and development of antibacterial silver based Chitosan Nanocomposite. Int J Biol Macromol 189:18–33. https://doi.org/10.1016/j.ijbiomac.2021.08.056
Kemenade B, Groeneveld A, Rens B, Rombout J (1994) Characterization of macrophages and neutrophilic granulocytes from the pronephros of carp (Cyprinus carpio L). J Exp Biol 187(1):143–158. https://doi.org/10.1242/jeb.187.1.143
Li H, Wang X, Li P, Li Y, Wang H (2008) Comparative study of antioxidant activity of grape (Vitis vinifera) seed powder assessed by different methods. J Food Drug Anal 16(6):67–73. https://doi.org/10.38212/2224-6614.2321
Liu X, Yao H, Zhao X, Ge C (2023) Biofilm formation and control of foodborne pathogenic bacteria. Molecules 28(6):2432. https://doi.org/10.3390/molecules28062432
Manik UP, Nande A, Raut S, Dhoble SJ (2020) Green synthesis of silver nanoparticles using plant leaf extraction of Artocarpus heterophylus and Azadirachta indica. Results Mater 6:100086. https://doi.org/10.1016/j.rinma.2020.100086
Manikandan V, Velmurugan P, Park JH, Chang WS, Park YJ, Jayanthi P, Cho M, Oh BT (2017) Green synthesis of silver oxide nanoparticles and its antibacterial activity against dental pathogens. 3 Biotech 7:1–9. https://doi.org/10.1007/s13205-017-0670-4
Morillo-Castillo JA, Balseca-Ibarra MC (2018) Inhibitory effect of the essential oil of Cymbopogon citratus on the strain of Porphyromona gingivalis: in vitro study. Odontología 20(2):5–13. https://doi.org/10.29166/odontologia.vol20.n2.2018-5-13
Nguyen VP, Trung HL, Nguyen TH, Hoang D, Tran TH (2021) Synthesis of biogenic silver nanoparticles with eco-friendly processes using Ganoderma lucidum extract and evaluation of their theranostic applications. J Nanomater Article ID 6135920:11. https://doi.org/10.1155/2021/6135920
O’Toole GA (2011) Microtiter dish biofilm formation assay. J Vis Exp 47:2437. https://doi.org/10.3791/2437
Ognik K, Cholewińska E, Czech A, Kozlowski K, Wlazlo L, Nowakowicz-Dębek B, Szlązak R, Tutaj K (2016) Effect of silver nanoparticles on the immune, redox, and lipid status of chicken blood. Czech J Anim Sci 61(10):450–461. https://doi.org/10.17221/80/2015-CJAS
Paunovic J, Vucevic D, Radosavljevic T, Mandić-Rajčević S, Pantic I (2020) Iron-based nanoparticles and their potential toxicity: focus on oxidative stress and apoptosis. Chem Biol Interac 316:108935. https://doi.org/10.1016/j.cbi.2019.108935
Perez Gutierrez RM (2016) Antidiabetic andantioxidant properties, and α-amylase and α-glucosidase inhibition effects of triterpene saponins from Piper auritum. Food Sci Biotechnol 25(1):229–239. https://doi.org/10.1007/s10068-016-0034-6
Riss TL, Moravec RA, Niles AL, Duellman S, Benink HA, Worzella TJ, Minor L (2016) Cell viability assays. Assay guidance manual. Eli Lilly & Company and the National Center for Advancing Translational Sciences, Bethesda, pp 1–23
Romero-García DM, Velázquez-Carriles CA, Gomez C, Velázquez-Juárez G, Silva-Jara JM (2023) Tannic acid-layered hydroxide salt hybrid: assessment of antibiofilm formation and foodborne pathogen growth inhibition. J Food Sci Technol 60(10):2659–2669. https://doi.org/10.1007/s13197-023-05790-4
Salleh WMNHW (2021) A systematic review of botany, phytochemicals and pharmacological properties of Hoja Santa (Piper auritum Kunth). Z Naturforsch C J Biosci 76(3–4):93–102. https://doi.org/10.1515/znc-2020-0116
Shehabeldine AM, Elbahnasawy MA, Hasaballah AI (2021) Green phytosynthesis of silver nanoparticles using Echinochloa stagnina extract with reference to their antibacterial, cytotoxic, and larvicidal activities. BioNanoScience 11:526–538. https://doi.org/10.1007/s12668-021-00846-1
Silva-Jara J, Angulo C, Macías ME, Velazquez C, Guluarte C, Reyes-Becerril M (2020) First screening report of immune and protective effect of non-toxic Jatropha vernicosa stem bark against Vibrio parahaemolyticus in Longfin Yellowtail Seriola rivoliana leukocytes. Fish Shellfish Immunol 101:106–114. https://doi.org/10.1016/j.fsi.2020.03.048
Singleton VL, Orthofer R, Lamuela-Raventós RM (1999) Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Meth Enzymol 299:152–178. https://doi.org/10.1016/S0076-6879(99)99017-1
Sneha T, Krishna MG, Sandhya RM (2017) Plant-mediated synthesis of silver nanoparticles—a critical review. Int J Pharmacogn Phytochem 9(7):947–956. https://doi.org/10.25258/phyto.v9i07.11161
Tang KWK, Millar BC, Moore JE (2023) Antimicrobial resistance (AMR). Br J Biomed Sci 80:11387. https://doi.org/10.3389/bjbs.2023.11387
Wang M, Zhu P, Zhao S, Nie C, Wang N, Du X, Zhou Y (2017) Characterization, antioxidant activity and immunomodulatory activity of polysaccharides from the swollen culms of Zizania latifolia. Int J Biol Macromol 95:809–817. https://doi.org/10.1016/j.ijbiomac.2016.12.010
Acknowledgements
Thanks to Alma Mercado Gutiérrez for technical support and Sergio Oliva León for X-ray and SEM support.
Funding
Authors thank the Consejo Nacional de Humanidades, Ciencia y Tecnología (CONAHCyT) for the post-doctoral economic support for three authors (Aguilar-Ávila, Hinojosa-Ventura and Velázquez-Carriles).
Author information
Authors and Affiliations
Contributions
Aguilar-Avila: Writing and editing, data analysis. Reyes-Becerril: Immune experiments, writing and editing, resource provide, data analysis. Velázquez-Carriles: Experimental research, first draft writing, and editing, data analysis. Hinojosa-Ventura: Writing and editing, spelling checking, data analysis. Macías-Rodríguez: Resource provide, review. Angulo: Resource provide, review. Silva-Jara: First draft writing, experimental design, resource provide, writing editing, data analysis.
Corresponding author
Ethics declarations
Conflict of interest
Authors declare no conflict interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Aguilar-Ávila, D.S., Reyes-Becerril, M., Velázquez-Carriles, C.A. et al. Biogenic Ag2O nanoparticles with “Hoja Santa” (Piper auritum) extract: characterization and biological capabilities. Biometals (2024). https://doi.org/10.1007/s10534-024-00589-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10534-024-00589-y