Abstract
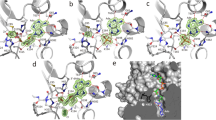
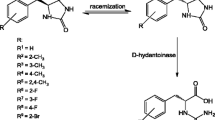
d-Hydantoinase (HDT) is a metal-dependent enzyme that is widely used in industrial bioconversion to d-amino acids as valuable intermediates in the fields of food, pharmaceutical industry and agriculture. In this report, we prepared apo-HDT (metal-removed HDT) and Zn2+-HDT (Zn2+-added HDT) in vitro from a recombinant HDT (re-HDT) expressed in E. coli. The Zn2+-HDT and re-HDT contain 2.17 and 0.95 mol Zn2+ per mol subunit, respectively, and they have comparable enzymatic activities. In contrast, the apo-HDT only retains 0.04 mol Zn2+ per mol subunit with less than 10% activity, compared with the re-HDT. When the apo-HDT was reconstituted with ZnCl2, the enzymatic activity recovery was about 75%. Moreover, the fluorescence intensity, circular dichroism spectra and thermo-stability of the apo-HDT and Zn2+-HDT are quite different from those of the re-HDT. These data suggest that the re-HDT may have two Zn2+-binding sites, one is an intrinsic or tight-binding site (zinc-α) essential for its activity and the other is a vacant or loose-binding site (zinc-β) possibly non-essential for the activity.







Similar content being viewed by others
References
Abendroth J, Niefind K, May O, Siemann M, Syldatk C, Schomburg D (2002) The structure of l-hydantoinase from Arthobacter aurescens leads to an understanding of dihydropyrimidinase substrate and enantio specificity. Biochemistry 41:8589–8597
Altenbuchner J, Siemann-Herzberg M, Syldatk C (2001) Hydantoinases and related enzymes as biocatalysts for the synthesis of unnatural chiral amino acids. Curr Opin Biotechnol 12:559–563
Armas A, Sonois V, Mothes E, Mazarguil H, Faller P (2006) Zinc(II) binds to the neuroprotective peptide humanin. J Inorg Biochem 100:1672–1678
Brooks KP, Jones EA, Kim BD, Sander EG (1983) Bovine liver dihydropyrimidine amidohydrolase: purification, properties, and characterization as a zinc metalloenzyme. Arch Biochem Biophys 226:469–483
Cheon YH, Kim HS, Han KH, Abendroth J, Niefind K, Schomburg D, Wang J, Kim Y (2002) Crystal structure of d-hydantoinase from Bacillus stearothermophilus: insight into the stereochemistry of enantioselectivity. Biochemistry 41:9410–9417
Cheon YH, Park HS, Lee SC, Lee DE, Kim HS (2003) Structure-based mutational analysis of the active site residues of d-hydantoinase. J Mol Catal B 26:217–222
Erk I, Huet JC, Duarte M, Duquerroy S, Rey F, Cohen J, Lepault J (2003) A zinc ion controls assembly and stability of the major capsid protein of rotavirus. Virol J 77:3595–3601
Gojkovic Z, Rislund L, Andersen B, Sandrini MPB, Cook PF, Schnackerz KD, Piškur J (2003) Dihydropyrimidine amidohydrolases and dihydroorotases share the same origin and several enzymatic properties. Nucleic Acids Res 31:1683–1692
Golynskiy MV, Davis TC, Helmann JD, Cohen SM (2005) Metal-induced structural organization and stabilization of the metalloregulatory protein MntR. Biochemistry 44:3380–3389
Huang DTC, Thomas MAW, Christopherson RI (1999) Divalent metal derivatives of the hamster dihydroorotase domain. Biochemistry 38:9964–9970
Jahnke K, Podschun B, Schnackerz KD, Kautz J, Cook PF (1993) Acid–base catalytic mechanism of dihydropyrimidinase from pH studies. Biochemistry 32:5160–5166
Kikugawa M, Kaneko M, Fujimoto-Sakata S, Maeda M, Kawasaki K, Takagi T, Tamaki N (1994) Purification, characterization and inhibition of dihydropyrimidinase from rat liver. Eur J Biochem 219:393–399
Lohkamp B, Andersen B, Piškur J, Dobritzsch D (2006) The crystal structures of dihydropyrimidinases reaffirm the close relationship between cyclic amidohydrolases and explain their substrate specificity. J Biol Chem 281:13762–13776
Marti-Renom MA, Stuart AC, Fiser A, Sanchez R, Melo F, Sali A (2000) Comparative protein structure modeling of genes and genomes. Annu Rev Biophys Biomol Struct 29:291–325
May O, Siemann M, Siemann MG, Syldatk C (1998a) The hydantoin amidohydrolase from Arthrobacter aurescens DSM 3745 is a zinc metalloenzyme. J Mol Catal B 5:367–370
May O, Siemann M, Siemann MG, Syldatk C (1998b) Catalytic and structural function of zinc for the hydantoinase from Arthrobacter aurescens DSM 3745. J Mol Catal B 4:211–218
Mishima T, Ohkuri T, Monji A, Imoto T, Ueda T (2006) Amyloid formation in denatured single-mutant lysozymes where residual structures are modulated. Protein Sci 15:2448–2452
Niu LX, Zhang XY, Shi YW, Yuan JM (2007) Subunit dissociation and stability alteration of d-hydantoinase deleted at the terminal amino acid residue. Biotechnol Lett 29:303–308
Pozo C, Rodelas B, de la Escalera S, González-López J (2002) d, l-Hydantoinase activity of an Ochrobactrum anthropi strain. J Appl Microbiol 92:1028–1034
Shaw CF III, Savas MM, Petering DH (1991) Ligand substitution and sulfhydryl reactivity of metallothionein. Methods Enzymol 205:401–414
Shi YW, Niu LX, Feng X, Yuan JM (2006) Purification, enzymatic properties of a recombinant d-hydantoinase and its dissociation by zinc ion. World J Microbiol Biotechnol 22:675–680
Shi YW, Zhang L, Yuan JM, Xiao H, Yang XQ, Niu LX (2008) Zinc binding site in PICK1 is dominantly located at the CPC motif of its PDZ domain. J Neurochem 106:1027–1034
Vieille C, Zeikus GJ (2001) Hyperthermophilic enzymes: sources, uses, and molecular mechanisms for thermostability. Microbiol Mol Biol Rev 65:1–43
Vlasova TN, Ugarova NN (2007) Quenching of the fluorescence of Tyr and Trp residues of firefly luciferase from Luciola mingrelica by the substrates. Biochemistry (Moscow) 72:962–967
Xu Z, Liu Y, Yang Y, Jiang W, Arnold E, Ding J (2003) Crystal structure of d-hydantoinase from Burkholderia pickettii at a resolution of 2.7 Angstroms: insights into the molecular basis of enzyme thermostability. J Bacteriol 185:4038–4049
Zhang XY, Niu LX, Shi YW, Yuan JM (2008) The flexibility of the non-conservative region at the C terminus of d-hydantoinase from Pseudomonas putida YZ-26 is extremely limited. Appl Biochem Biotechnol 144:237–247
Zhao GH, Pu P, Hu XS, Zhao L (2004) Effect of Zn(II) on the structure and biological activity of natural β-NGF. Acta Biochim Biophys Sin 36:99–104
Acknowledgment
We acknowledge that this project is supported by the Natural Science Foundation of Shanxi province in China (No. 031042).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, X., Yuan, J., Niu, L. et al. Quantitative analysis and functional evaluation of zinc ion in the d-hydantoinase from Pseudomonas putida YZ-26. Biometals 23, 71–81 (2010). https://doi.org/10.1007/s10534-009-9267-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-009-9267-7