Abstract
The sandy-savanna Mussununga is a little-known ecosystem that is already presenting signs of degradation due to effects of invasive species, anthropogenic disturbances, and land-use change. We investigated the effect of biological invasion by Acacia mangium and A. auriculiformis, disturbance promoted by fire and Eucalyptus management, as well as land-use change around Mussununga patches in this neglected ecosystem. Acacia occurrence is negatively related to abundance, basal area, and height of native woody species, and positively correlated with the proportion of dead woody plants. There is a trend of change in vegetation type from open savanna to woodland Mussununga promoted by Acacia. Mussununga patches disturbed by the planting of Eucalyptus species in the past have greater richness in the woody layer (large shrubs and trees), but lower abundance of species in the ground layer (small shrubs and herbs). The land-use change from forest to Eucalyptus plantation was associated with an increase of richness and abundance in the ground layer. Acacia tends to dominate the woody community in Mussununga by invading treeless patches, presenting a correlation with death of woody plants. Subsequently, Acacia promotes several changes that culminate in a height increment and change of the vegetation type. Different from Acacia, disturbance and land-use change associated with Eucalyptus, the main economic activity in the study area, affected both woody and ground layers. The conservation of Mussununga should include actions to preclude disturbance and land-use change, as well as monitoring and control of biological invasion.
Similar content being viewed by others
Introduction
The invasive genus Acacia, subgenus Phyllodineae, originally from Australia and surrounding islands (Murphy 2008), comprises some of the most invasive woody plants and is currently widespread around the world (Richardson and Rejmánek 2011), mainly due to human economic interest in some species for use in a range of different purposes. Many species of Acacia genus grow fast and have good adaptability to soils with low nutrient levels as well as to disturbed areas (National Research Council 1983; Griffin et al. 2011). The genus Acacia contains species that are able to thrive in a diversity of ecosystems such as dunes, understory of plantations, savanna vegetation, open forests, forest edges, and abandoned lands, and contributes to a large variety of ecosystem changes (Rascher et al. 2011; Delnatte and Meyer 2012; Aguiar et al. 2014).
Although a range of ecosystems can be invaded by different species of Acacia, the outcomes are usually similar, including changes to the native plant community and ecosystem functioning, mainly in light availability, nitrogen cycle dynamics, and soil characteristics (Marchante et al. 2008; Meira-Neto et al. 2017; Große-Stoltenberg et al. 2018; Koutika and Richardson 2019). For instance, Acacia longifolia (Andrews) Willd. reduced the growth of native seedlings, and thereby negatively affected the regeneration of native vegetation and promoted the growth of their own seedlings in pine forests (Rascher et al. 2011). In dune ecosystems A. longifolia may also increase the contents of macronutrients (N, K, and Mg) and organic matter in the soil (Hellmann et al. 2011), as well as promote litter accumulation (Marchante et al. 2008). Other species such as Acacia mangium Willd. can increase richness and leaf nitrogen content of native species, reduce light availability and promote an increase in height of the horizontal structure of the Mussununga vegetation (Lehmann et al. 2017; Meira-Neto et al. 2017). Such diversity and ecosystem changes are commonly reported when Acacia species colonize nutrient-poor and non-forest ecosystems, and degraded areas (e.g., Marchante et al. 2008; Rascher et al. 2011; Delnatte and Meyer 2012; Aguiar et al. 2014).
Some species of Acacia are favored by disturbance and can germinate and become dominant after fire occurrence (Le Maitre et al. 2011). Furthermore, the replacement of native forest by woody plantations can also affect Acacia species’ seed scattering. Invasion rates increase in areas with land that is susceptible to invasion and, in turn, this increases the propagule pressure due to an increase in the occurrence of exotic species (Donaldson et al. 2014a). Anthropogenic factors, such as disturbance and land-use change, may increase the rate of invasion due to the exclusion of native species (Kennedy et al. 2002), changes in disturbance regimes (Dillis et al. 2017) or an increase in the frequency of invasive species introduction events (Donaldson et al. 2014a). Highways and roads may benefit invasive plants either through increased disturbance intensity (Pollnac et al. 2012) or they may facilitate the spread of invasive species (Thiele et al. 2008; Delnatte and Meyer 2012). In turn, the larger the invaded area, the greater will be the propagule pressure in such a landscape (Donaldson et al. 2014a).
Anthropogenic factors and biological invasion can also directly impact diversity and ecosystem functioning (Vitousek et al. 1997; Miller et al. 2011). For instance, the increase in frequency or intensity of fire due to human activity may contribute to the shaping of a treeless savanna (Moreira 2000), mainly through the exclusion of fire-sensitive woody species (Hoffmann 1996). Habitat degradation decreases landscape connectivity affecting plant dispersal and reducing similarity between remaining habitats (Thiele et al. 2018), while benefitting the distribution of some invasive species (Thiele et al. 2008; Donaldson et al. 2014a). Finally, biological invasion affects diversity by means of competition and replacement of native species (Gaertner et al. 2009; Fischer et al. 2014) or due to changes in ecosystem processes (Fridley et al. 2007; Gaertner et al. 2014). Thus, investigating these sets of predictive variables allows for a better comprehension of how anthropogenic factors and biological invasion interfere in the loss of diversity and ecosystem structure.
In a recent paper, Meira-Neto et al. (2017) showed that early Acacia mangium invasion in Mussunungas increases shading as well as leaf nitrogen content in the neighborhood plants and concluded that it would be necessary to assess whether Acacia invasion could lead to diversity loss in subsequent years. Nevertheless, the above-mentioned authors did not investigate the role of anthropogenic factors in the Mussununga ecosystem as was observed some years earlier (Meira-Neto et al. 2005). Considering that anthropogenic habitat alteration and biological invasion may play a synergetic role in changes in diversity and ecosystem properties (Gurevitch and Padilla 2004; Didham et al. 2005; Le Maitre et al. 2011), in this paper we aimed to study the effects of invasion by A. mangium and A. auriculiformis Cunn. ex Benth., disturbance by fire and Eucalyptus management, and land-use change around Mussununga patches in an area of Mussununga vegetation. To do so, we investigated the hypothesis that (1) biological invasion by A. mangium and A. auriculiformis promotes loss of diversity and changes in the vegetation structure of the Mussununga ecosystem; (2) disturbance promoted by fire occurrence and Eucalyptus management inside Mussununga patches as well as land-use change from forest to Eucalyptus plantation around Mussununga promotes diversity loss and change in the vegetation structure; (3) the woody layer community and ground layer community are affected in different ways by biological invasion, disturbance, and land-use change.
Methodology
Study area
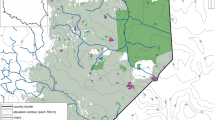
The study was conducted in eastern Brazil, in the states of Espírito Santo and Bahia. We sampled Mussununga patches located between Linhares, in northern Espírito Santo (19° 25′S, 40° 04′W), and Caravelas, in southern Bahia (17° 44′S, 39° 15′W) (Fig. 1). The climate in the region ranges from tropical with a dry winter to tropical monsoon and tropical with no dry season (respectively, Aw, Am, and Af in the Köppen climate classification system—Alvares et al. 2014). The original vegetation of the Atlantic Forest domain is highly fragmented, and the current landscape is dominated by Eucalyptus plantations, pastures, crop fields, forest remnants, mangrove, Restinga (coastal dunes originated in the Quaternary period and having a wide range of vegetation types composed of herbs, shrubs, and trees) and patches of Mussununga savannas.
The Mussununga patches vary widely in size and shape and are spread throughout a matrix of Lowland Atlantic Forest (IBGE 2004) and anthropogenic landscape (e.g., Eucalyptus plantations, pastures, and crop fields). There is a gradient of vegetation types in the Mussununga, from grasslands, which are dominated by a few monocot species, to savanna itself, composed of one ground layer vegetation and another layer of scattered woody plants, and to woodlands with closed canopies (Saporetti-Junior et al. 2012). The occurrence of Mussununga is strictly associated with acid, nutrient-poor, sandy soils, which are formed by podzolization due to high humidity and by the hydromorphism caused by an impermeable Ortstein layer (Saporetti-Junior et al. 2012; Ferreira et al. 2014). The Mussununga ecosystem is still highly neglected in the scientific literature (Eisenlohr et al. 2015) despite the fact that it is already threatened by many factors, such as fire, logging, road construction, and biological invasion.
Study species
The exotic invasive species Acacia auriculiformis and A. mangium are native to Australia, Papua New Guinea and Indonesia (National Research Council 1983), and both are the most widespread Acacia species in the world for human use, such as for solid wood, pulpwood, fuelwood production, and for recovery of degraded lands (Griffin et al. 2011; Koutika and Richardson 2019). In Brazil, these species are reported to occur in several states along the entire coastline (Santa Catarina, Rio de Janeiro, Espírito Santo, Bahia, Pernambuco, Maranhão, Amapá, Amazonas, and Roraima) (I3N Brazil 2017). Both species were introduced in the Lowland Atlantic Forest region at around the 1970s as an alternative for the recovery of degraded lands. Since then, they have spread and invaded not only degraded lands but also pastures, Eucalyptus plantations and the Mussunungas in the region (Lehmann et al. 2017), having been first recorded in the sampling patches less than 10 years ago. Both species are well adapted to regions with a short dry period, high temperatures, high average annual rainfall and nutrient-poor soil conditions (National Research Council 1983; Delnatte and Meyer 2012).
Sampling design
Fieldwork was conducted between September 2015 and March 2016 in 13 Mussununga patches (Fig. 1). We sampled thirteen Mussununga patches by allocating 10 to 15 plots in each patch, distributed at least 10 m apart from each other. In total we sampled 144 plots, of which 35 plots were invaded by A. mangium or A. auriculiformis and 109 were not invaded. With respect to plot management history, a total of 51 had signs of fire, 30 had Eucalyptus management in the past, 10 had both fire and Eucalyptus, and 53 did not present signs of disturbance. A total of 94 plots were sampled in Mussunungas predominantly surrounded by Eucalyptus plantations, 30 were sampled in Mussunungas predominantly surrounded by forest, and 20 were sampled in Mussunungas surrounded by both these vegetation types (see Selected variables below).
Similar to the Cerrado sensu lato described by Oliveira-Filho and Ratter (2002), the sandy-savanna Mussununga is characterized by two layers of plant communities. The first one is composed of trees and large shrubs (hereafter called the woody layer), and the other is composed by small shrubs and herbs (hereafter called ground layer). We therefore used 10 × 10 m plots to survey the woody layer and added a 2 × 2 m subplot in the southeast corner of each plot to sample the ground layer. In the woody layer, we sampled all individuals of plants rooted in the plot and having a diameter at ground line ≥ 3.2 cm, height ≥ 0.9 cm, recording the species name and height. To sample the ground layer community, we examined all individuals in the subplot and recorded their species name, the absolute percentage cover per species, and mean of cover per species according to Braun-Blanquet (1979). Saplings or shrubs that did not meet the inclusion criteria for the woody layer were included in the ground layer sampling provided they were within the subplots. The mean of cover was calculated using transformed values from the cover-abundance scale of Braun-Blanquet (1979), and native species were classified following Brazilian Flora 2020 (2017).
Selected variables
For the woody layer, we used the presence/absence, basal area (measured as the cross-section of the stem at ground line), and abundance of A. mangium and A. auriculiformis per plot as independent variables. Additionally, we included two categorical variables related to anthropogenic activity as independent variables: disturbance and land-use change. Disturbance refers to anthropogenic factors that directly affect a Mussununga patch and was categorized per plot: no disturbance, presence of Eucalyptus, fire, and Eucalyptus plus fire. Although Eucalyptus invasion has not been seen in the studied Mussunungas, the few isolated eucalypt trees that remained from the management practices used in the past could affect native vegetation. Several companies have tried to establish Eucalyptus plantations inside these Mussununga patches, but they were not successful. Thus, after the patches had been abandoned, the Mussununga vegetation regenerated and were then no longer targets of management.
Land-use change, on the other hand, refers to activities conducted around the Mussununga patch and therefore was categorized for each patch as native forest, Eucalyptus plantation, and a mix of native forest and Eucalyptus plantation. Both disturbance and land-use change were categorized based exclusively on clear evidence found during fieldwork and could not be scaled due to the absence of historical records. More details about the independent variables and their measurements are presented in Table 1.
In the woody layer, we investigated the following dependent variables per plot: species richness, Pielou’s evenness, and Shannon’s index as diversity predictors, and abundance, proportion of dead individuals, geometric mean of height, basal area, and the two ordination axes of a non-metric multidimensional scaling (NMDS) as predictors of native vegetation structure. Moreover, we also tested the basal area and the geometric mean of height, including Acacia species in the sum as dependent variables (henceforth referred to as “total basal area” and “total geometric mean of height”, respectively), aiming to evaluate the effect of biological invasion, anthropogenic disturbance and land-use change on the vegetation type.
In the case of the ground layer, we tested the same aforementioned independent variables and added the presence and absolute percentage cover of A. mangium and A. auriculiformis in the subplots as an additional independent variable, aiming to test the immediate effect of the species. As dependent variables, we used the following dependent variables per plot: species richness, Pielou’s evenness, Shannon’s index (diversity variables), absolute percentage cover, abundance, and the first two ordination axes of an NMDS (structure variables). Neither the mean of cover (sensu Braun-Blanquet 1979) nor abundance of Acacia in the subplots was used in our model, as they both showed a high Spearman’s correlation coefficient with absolute percentage cover of Acacia (rs = 1.0 and 1.0, respectively), and the native mean of cover (sensu Braun-Blanquet 1979) was also not used because it showed a high Spearman’s correlation coefficient with native absolute percentage cover (rs = 0.96).
Statistical analyses
First, we constructed a matrix with all the above-mentioned variables and then used the software R version 3.4.1 (R Development Core Team 2016) to calculate the NMDS axis using the Bray–Curtis distance, species abundance (stress = 0.13 and 0.18 for the woody and ground layer samplings, respectively) and Shannon’s index, with the “vegan” package (Oksanen et al. 2016). Pielou’s index was obtained as the ratio between Shannon’s index and richness (natural logarithm). Subsequently we tested Spearman’s correlation between variables using the “PerformanceAnalytics” package (Peterson and Carl 2014) and excluded all variables having a Spearman’s correlation coefficient ≥ 0.70. In addition, we also verified the effect of multicollinearity using the GVIF(1/2*df) implemented in the “car” package (Fox and Monette 1992; Fox and Weisberg 2011).
We tested the effect of Acacia invasion and anthropogenic factors on the diversity, structure and vegetation type variables using generalized linear mixed models in the “lme4” package (Bates et al. 2015) for each plot or subplot (the latter in ground layer analysis). We added the identity of each Mussununga patch as a random effect in our model to account for the nested sampling design. To choose the best model and proceed with necessary transformations we tested linearity using the “gof” package (Holst 2014), based on the generalized linear model, and distribution was tested using the “MASS” package (Venables and Ripley 2002). All models were built using transformed data of the basal area and abundance of Acacia, which were divided by the standard deviation (except for total geometric mean of height, in which case we used a square root transformation). For the significant relationships between anthropogenic factors and dependent variables with more than two classes (disturbance and land-use change) we conducted a contrast analysis to test the differences between classes. To test the correlation between Acacia invasion and the anthropogenic factors disturbance and land-use change we used a Chi square in the “stats” package (R Development Core Team 2016). A probability level of α = 0.05 was used for all statistical tests.
Results
During this study, we recorded a total of 5539 individuals, 90 species, 74 genera, and 40 families. In the woody layer, 1361 individuals, 39 species, 35 genera, and 23 families were found, while in the ground layer we sampled 4178 individuals, 79 species, 66 genera and 36 families. In the woody layer plots, the abundance of native species ranged from 0 to 31 individuals (8.7 ± 6.3 SD) and richness from 0 to 8 species (2.6 ± 1.5), while in the ground subplots the abundance of native species ranged from 4 to 116 individuals (28.9 ± 19.9), richness from 2 to 13 species (6.0 ± 2.1) and absolute percentage cover from 16 to 221 (104.3 ± 41.7). Acacia occurrence presented a correlation with disturbance and land-use change (respectively, X2 = 38.019 and 24.595; p < 0.001; Supplementary material, Fig. S1a, b). All independent variables tested had GVIF(1/2*df) ≤ 2, therefore we did not consider collinearity as an issue in our analysis (Supplementary material, Table S2).
Woody layer community
Acacia affected the woody layer but not the ground layer. The presence of Acacia was associated with lower values of abundance, basal area, and the geometric mean of height of woody plants, as well as with a higher proportion of dead woody individuals (Fig. 2a, b, c; Table 2). The abundance of Acacia positively affected Pielou’s evenness and the proportion of dead woody individuals (Fig. 2f; Table 2). Basal area of Acacia, in turn, had a positive effect on abundance and basal area of woody variables when we did not exclude the outlier from the analysis (Fig. 2d, e; Table 2; Supplementary material, Table S1). When we tested how Acacia variables affected the total basal area and the total geometric mean of height, in order to assess the effects on vegetation type, we found positive effects of the basal area of Acacia on both dependent variables (Table 2).
The relationship between A. mangium and A. auriculiformis invasion (presence, basal area, and abundance) and woody community variables. Each dot corresponds to the value found in one plot; dots are darker according to the number of plots with the same value. Black bars correspond to the median; grey boxes correspond to the range between the first and third quartiles; and the lower-case letters over the boxes correspond to a probability level of α = 0.05 in GLMM (a–c). Solid curves represent the estimated means (d–f)
Additionally, disturbance due to Eucalyptus management was positively associated with native species richness and negatively associated with NMDS axis 1 in the woody layer. Native forest land-use had a negative effect on NMDS axis 2 (Fig. 3a, b, c; Table 2).
The relationship of disturbance and land-use change (referred as land-use) with woody community variables (a–c) and ground community variables (d–f). Each dot corresponds to the value found in one plot; dots are darker according to the number of plots with the same value. Black bars correspond to the median; grey boxes correspond to the range between the first and third quartiles; and the lower-case letters over the boxes correspond to a probability level of α = 0.05 in contrast analysis. Euc. = Eucalyptus
Ground layer community
Acacia species had no effect on the ground layer, even when their presence, absolute percentage cover, and abundance in the subplot were tested (Supplementary material, Table S2). Nevertheless, anthropogenic factors affected the abundance and richness of the ground layer. Although the disturbance promoted by Eucalyptus management was negatively associated with native species abundance, the Eucalyptus plantation around Mussununga patches had a positive effect on native species abundance and richness (Fig. 3d, e, f; Table 2). Neither disturbance nor land-use change affected the total basal area or total geometric mean of height (Supplementary material, Table S2).
Discussion
Acacia invasion, disturbance by Eucalyptus management, and land-use change from forest to Eucalyptus plantation are causing changes in the diversity, structure, and vegetation type of the little-known Mussununga ecosystem. With respect to the hypotheses described at the beginning of this study, we found that (1) Acacia has a negative effect on woody species composition, causing death of plants, and on increment in total height and total basal area of the invaded Mussununga; (2) disturbance and land-use change affect diversity and structure of the vegetation; and (3) the ground layer community was not affected by Acacia, while biological invasion seems to be the main driver in the woody layer.
Acacia invasion
The negative relation of A. mangium and A. auriculiformis on the woody layer community in Mussununga is in agreement with other studies that evaluated different species of acacias (e.g., Costello et al. 2000; Marchante et al. 2003; Rascher et al. 2011) and corroborates that Acacia dominance is a key factor in the destruction of the Mussununga ecosystem (Meira-Neto et al. 2017). The effect of Acacia invasion on the woody community suggests a replacement of species, caused mainly by Acacia and the death of native woody plants (see Fig. 1). That is a concern since the plant species in Mussununga are adapted to very specific environmental conditions with a combination of flooding and drought and nutrient-poor sandy soil (Meira-Neto et al. 2005; Saporetti-Junior et al. 2012; Gastauer et al. 2017).
However, the positive relationship of the basal area of Acacia with abundance and basal area of native woody species challenges our previous expectations. Although the process of (re-)cover after disturbance is a parsimonious interpretation (see below “Threats to the Mussununga ecosystem”; Fig. 4), Acacia acts as nurse plant in the early invasion stage in the Mussununga ecosystem, as supported by the higher native species richness in Acacia-focused plots than non-invaded ones (Meira-Neto et al. 2017). When at low density, Acacia possibly can benefit native species by ameliorating the stressed conditions in Mussununga, providing shade and increasing soil nitrogen and carbon contents (Marchante et al. 2009; Hellmann et al. 2011; Meira-Neto et al. 2017), thereby, facilitating the establishment of plants not adapted to stress conditions. For instance, Yang et al. (2009) found better physiological status and lower stress in shaded plants under A. mangium and A. auriculiformis canopies in China.
We hypothesize that the positive effect of nitrogen addition and shading in the Mussununga should only occur up to a threshold of Acacia abundance. After becoming dominant, Acacia would outcompete the light-demanding species (Meira-Neto et al. 2017) and possibly favor their own seedlings (Rascher et al. 2011). This point of view is corroborated here by the positive relationship of the basal area of Acacia on the woody layer community and, contrarily, the statistical effect of the abundance of Acacia on the proportion of dead plants (Fig. 2d, e, f) and by the fact that the negative effects of invasion of Acacia species frequently take decades to appear (Koutika and Richardson 2019). The increase of biomass promoted by Acacia measured by total basal area and the total geometric mean of height (that is, including Acacia in the measurement) indicates a change in the vegetation structure of the Mussununga. Therefore, the gradient of vegetation types from grassland to woodland, limited by flooding, nutrient content in soil, and fire (Saporetti-Junior et al. 2012; Gastauer et al. 2017), is unbalanced by Acacia occurrence. Despite the negative effect on the native woody community, following Acacia invasion, the sandy-savanna Mussununga tends to become taller and more closed with consequent reduction of light availability and increase of nitrogen content. This trend is clearly related to the capacity of the Acacia species studied here to grow faster and taller than native plants from Mussununga, and even in the early stages of invasion this can already be detected using aerial images (Lehmann et al. 2017). Under this new environmental condition, the typical light-demanding species of Mussununga could be replaced by shade-tolerant ones or, in worse scenarios, by Acacia itself becoming a monodominant forest.
Anthropogenic factors
Anthropogenic factors affected the diversity and structure of both woody and ground layers, but the effect of each category varied (Fig. 3). We expected disturbance caused by Eucalyptus management would negatively affect the whole community due to suppression of native vegetation and movement of machinery during management. Nevertheless, we observed a positive effect on the woody layer richness and a negative one on the abundance of the ground layer community. Nutrient addition may promote biomass reduction in herbs, as shown in the Brazilian Cerrado by (Lannes et al. 2016), thus alleviating soil conditions in the Mussununga and allowing the establishment of woody species less adapted to nutrient-poor soils. Furthermore, we cannot exclude the possibility of an association, as the negative effect of Eucalyptus disturbance on ground abundance could reduce the dominance of grasses and benefit the establishment and growth of woody species (Spearman’s correlation between ground abundance and woody richness: rs = − 0.27; p = 0.001). As a matter of fact, the Eucalyptus plantation nearby the sampled Mussunungas are rich in native woody species that established spontaneously in the understory. We also expected that the occurrence of fire would have a role in the balance between woody and ground layer communities, since fire tends to affect the woody layer more severely than the ground layer in savanna-type ecosystems (Moreira 2000) and is one of the most common environmental filtering factors in open Mussunungas (Gastauer et al. 2017). Considering the role that intensity, frequency, and time since the last disturbance play in the diversity-disturbance relationship (Miller et al. 2011), we argue that future research should include those variables in order to properly assess the role of fire.
Mussununga patches surrounded by Eucalyptus plantations showed higher richness and abundance in ground samples (Fig. 3e, f). The ground layer community in Mussunungas are mostly composed of light-demanding species, which themselves could not cross the dense forest matrix of Lowland Rainforest. We speculate that human-aided long-distance dispersal, described for some invasive Acacia species (Donaldson et al. 2014a), plays a key role in increasing richness and abundance in Mussununga patches because this would enhance the dispersal rates of light-demanding species among Mussununga patches. Furthermore, the set of roads and highways around patches surrounded by Eucalyptus plantations could facilitate dispersal, as observed with invasive species (Thiele et al. 2008; Pollnac et al. 2012). The land-use occupied by Eucalyptus plantations includes frequent movement of vehicles to transport timber or manage the plantations and could help in the spread of native species, analogous to observations of several Acacia species in South Africa (Donaldson et al. 2014b).
Threats to the Mussununga ecosystem
The results shown here reveal a synergetic effect of anthropogenic factors and biological invasion on Mussununga, as well as a role of disturbance and land-use change in the biological invasion. Based on the framework developed by Le Maitre et al. (2011) and our field observations, we propose an explanation of the ecosystem changes observed in Mussununga. The occurrence of disturbance, such as severe drought, flooding, fire, or logging, decreases the density of woody species (Gastauer et al. 2017) and favors the establishment of Acacia. Subsequently, Acacia and native woody species would (re-)cover the ecosystem without causing negative effects to one another. Nevertheless, due to the fast growth rates, Acacia would become dominant quickly and then displace native vegetation by increasing shadow and causing changes in soil nutrient content (Meira-Neto et al. 2017) (Fig. 4 summarizes this hypothesis). The similar process shown in Portuguese dunes corroborates this, as described by Le Maitre et al. (2011). In this study the authors showed that during the post-disturbance recovery stage, both the density of A. longifolia and richness of native species increased until a threshold, from which the ecosystem had high contents of nitrogen, soil carbon, and increased reinvasion potential, and native species richness decreased (Marchante et al. 2009; Le Maitre et al. 2011).
Our results support those from previous studies that demonstrated that Mussununga is suffering from a set of drivers that affect this still little-known ecosystem. Thus, we could expect that over time Acacia invasion would drastically reduce the native woody community while promoting an increase of biomass in the ecosystem due to the rapid covering by Acacia plants. At the same time, Mussununga patches that remained after Eucalyptus plantations were abandoned and those surrounded by Eucalyptus plantations would present an imbalance between the woody and ground layers. Long-term studies incorporating precise recordings of fire would help to understand how fire affects Mussununga vegetation and how it promotes biological invasion by Acacia. Considering the changes shown here and the “Precautionary Principle”, based on the results of introduction of A. mangium and A. auriculiformis around the world (see Koutika and Richardson 2019), we emphasize that adopting actions such as discouraging or precluding human disturbances, monitoring Mussununga regeneration after disturbance events and controlling Acacia invasion before it crosses the threshold of dominance may be the keys to preservation of the characteristics of this peculiar ecosystem.
References
Aguiar A Jr, Barbosa RI, Barbosa JBF, Mourao M Jr (2014) Invasion of Acacia mangium in Amazonian savannas following planting for forestry. Plant Ecol Divers 7:359–369. https://doi.org/10.1080/17550874.2013.771714
Alvares CA, Stape JL, Sentelhas PC et al (2014) Köppen’s climate classification map for Brazil. Meteorol Zeitschrift 22:711–728. https://doi.org/10.1127/0941-2948/2013/0507
Bates D, Maechler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48. https://doi.org/10.18637/jss.v067.i01
Braun-Blanquet J (1979) Fitosociología: bases para el estudio de las comunidades vegetales. H. Blume, Madrid
Brazilian Flora 2020 (2017) Jardim Botânico do Rio de Janeiro, Rio de Janeiro, Brazil. Available at: http://floradobrasil.jbrj.gov.br/. Accessed June 29 2017
Costello DA, Lunt ID, Williams JE (2000) Effects of invasion by the indigenous shrub Acacia sophorae on plant composition of coastal grasslands in south-eastern Australia. Biol Conserv 96:113–121. https://doi.org/10.1016/S0006-3207(00)00058-6
Delnatte C, Meyer J-Y (2012) Plant introduction, naturalization, and invasion in French Guiana (South America). Biol Invasions 14:915–927. https://doi.org/10.1007/s10530-011-0129-1
Didham RK, Tylianakis JM, Hutchison MA et al (2005) Are invasive species the drivers of ecological change? Trends Ecol Evol 20:470–474. https://doi.org/10.1016/j.tree.2005.07.006
Dillis C, Marshall AJ, Rejmánek M (2017) Change in disturbance regime facilitates invasion by Bellucia pentamera Naudin (Melastomataceae) at Gunung Palung National Park, Indonesia. Biol Invasions 19:1329–1337. https://doi.org/10.1007/s10530-016-1345-5
Donaldson JE, Hui C, Richardson DM et al (2014a) Invasion trajectory of alien trees: the role of introduction pathway and planting history. Glob Change Biol 20:1527–1537. https://doi.org/10.1111/gcb.12486
Donaldson JE, Richardson DM, Wilson JRU (2014b) The seed ecology of an ornamental wattle in South Africa: why has Acacia elata not invaded a greater area? S Afr J Bot 94:40–45. https://doi.org/10.1016/j.sajb.2014.05.004
Eisenlohr PV, Oliveira-Filho AT, Prado J (2015) The Brazilian Atlantic Forest: new findings, challenges and prospects in a shrinking hotspot. Biodivers Conserv 24:2129–2133. https://doi.org/10.1007/s10531-015-0995-4
Ferreira VBR, Nascimento MT, de Menezes LFT (2014) Floristic and phytogeographic pattern of native field in southeastern Brazil. Acta Bot Bras 28:465–475. https://doi.org/10.1590/0102-33062014abb3556
Fischer FM, Oliveira JM, Dresseno ALP, Pillar VD (2014) The role of invasive pine on changes of plant composition and functional traits in a coastal dune ecosystem. Nat Conserv 12:19–23. https://doi.org/10.4322/natcon.2014.004
Fox J, Monette G (1992) Generalized collinearity diagnostics. J Am Stat Assoc 87:178–183
Fox J, Weisberg S (2011) An R companion to applied regression, 2nd edn. Sage, Thousands Oaks
Fridley JD, Stachowicz JJ, Naeem S et al (2007) The invasion paradox: reconciling pattern and process in species invasions. Ecology 88:3–17. https://doi.org/10.1890/0012-9658(2007)88%5b3:Tiprpa%5d2.0.Co;2
Gaertner M, Den Breeyen A, Hui C et al (2009) Impacts of alien plant invasions on species richness in Mediterranean-type ecosystems: a meta-analysis. Prog Phys Geogr 33:319–338. https://doi.org/10.1177/0309133309341607
Gaertner M, Biggs R, Te Beest M et al (2014) Invasive plants as drivers of regime shifts: identifying high-priority invaders that alter feedback relationships. Divers Distrib 20:733–744. https://doi.org/10.1111/ddi.12182
Gastauer M, Saporetti-Junior AW, Valladares F, Meira-Neto JAA (2017) Phylogenetic community structure reveals differences in plant community assembly of an oligotrophic white-sand ecosystem from the Brazilian Atlantic Forest. Acta Bot Bras 31:531–538. https://doi.org/10.1590/0102-33062016abb0442
Griffin AR, Midgley SJ, Bush D et al (2011) Global uses of Australian acacias - recent trends and future prospects. Divers Distrib 17:837–847. https://doi.org/10.1111/j.1472-4642.2011.00814.x
Große-Stoltenberg A, Hellmann C, Thiele J et al (2018) Invasive acacias differ from native dune species in the hyperspectral/biochemical trait space. J Veg Sci. https://doi.org/10.1111/jvs.12608
Gurevitch J, Padilla DK (2004) Are invasive species a major cause of extinctions? Trends Ecol Evol 19:470–474. https://doi.org/10.1016/j.tree.2004.07.005
Hellmann C, Sutter R, Rascher KG et al (2011) Impact of an exotic N2-fixing Acacia on composition and N status of a native Mediterranean community. Acta Oecol 37:43–50. https://doi.org/10.1016/j.actao.2010.11.005
Hoffmann WA (1996) The effects of fire and cover on seedling establishment in a Neotropical Savanna.pdf. J Ecol 84:383–393
Holst KK (2014) gof: Model-diagnostics based on cumulative residuals. R package version 0.9.1
I3 N Brazil – National Database of Invasive Alien Species Brazil (2017) Instituto Hórus de Desenvolvimento e Conservação Ambiental, Florianópolis, Brazil. Available at: http://i3n.institutohorus.org.br/www. Accessed June 30 2017
IBGE – Instituto Brasileiro de Geografia e Estatística (2004) Mapa da Vegetação do Brasil. Rio de Janeiro, Brasil. Available at: ftp://ftp.ibge.gov.br/Cartas_e_Mapas/Mapas_Murais/. Accessed June 29 2017
Kennedy TA, Naeem S, Howe KM et al (2002) Biodiversity as a barrier to ecological invasion. Nature 417:636–638. https://doi.org/10.1038/nature00776
Koutika L-S, Richardson DM (2019) Acacia mangium Willd: benefits and threats associated with its increasing use around the world. For Ecosyst 6. https://doi.org/10.1186/s40663-019-0159-1
Lannes LS, Bustamente MMC, Edwards PJ, Venterink HO (2016) Native and alien herbaceous plants in the Brazilian Cerrado are (co-)limited by different nutrients. Plant Soil 400:231–243. https://doi.org/10.1007/s11104-015-2725-9
Le Maitre DC, Gaertner M, Marchante E et al (2011) Impacts of invasive Australian acacias: implications for management and restoration. Divers Distrib 17:1015–1029. https://doi.org/10.1111/j.1472-4642.2011.00816.x
Lehmann JR, Prinz T, Ziller SR et al (2017) Open-source processing and analysis of aerial imagery acquired with a low-cost unmanned aerial system to support invasive plant management. Environ Sci. https://doi.org/10.3389/fenvs.2017.00044
Marchante H, Marchante E, Freitas H (2003) Invasion of the Portuguese dune ecosystems by the exotic species Acacia longifolia (Andrews) Willd.: effects at the community level. In: Child LE, Brock JH, Brundu G et al (eds) Plant invasions: ecological threats and management solutions. Backhuys, Leiden, pp 75–85
Marchante E, Kjøller A, Struwe S, Freitas H (2008) Short- and long-term impacts of Acacia longifolia invasion on the below ground processes of a Mediterranean coastal dune ecosystem. Appl Soil Ecol 40:210–217. https://doi.org/10.1016/j.apsoil.2008.04.004
Marchante E, Kjøller A, Struwe S, Freitas H (2009) Soil recovery after removal of the N2-fixing invasive Acacia longifolia: consequences for ecosystem restoration. Biol Invasions 11:813–823. https://doi.org/10.1007/s10530-008-9295-1
Meira-Neto JAA, de Souza AL, de Lana JM, Valente GE (2005) Composição florística, espectro biológico e fitofisionomia da vegetação de muçununga nos municípios de Caravelas e Mucuri, Bahia. Rev Árvore 29:139–150. https://doi.org/10.1590/S0100-67622005000100015
Meira-Neto JAA, Silva MCNA, Tolentino GS et al (2017) Early Acacia invasion in a sandy ecosystem enables shading mediated by soil, leaf nitrogen, and facilitation. Biol Invasions. https://doi.org/10.1007/s10530-017-1647-2
Miller AD, Roxburgh SH, Shea K (2011) How frequency and intensity shape diversity–disturbance relationships. Proc Natl Acad Sci 108:5643–5648. https://doi.org/10.1073/pnas.1018594108
Moreira AG (2000) Effects of fire protection on savanna structure in Central Brazil. J Biogeogr 27:1021–1029. https://doi.org/10.1046/j.1365-2699.2000.00422.x
Murphy DJ (2008) A review of the classification of the Acacia (Leguminosae, Mimosoideae). Muelleria 26:10–26
National Research Council (1983) Mangium and other fast-growing Acacias for the humid tropics. National Academy Press, Washington
Oksanen J, Blanchet FG, Friendly M, et al (2016) vegan: Community ecology package. R package version 2.4-0. https://CRAN.R-project.org/package=vegan
Oliveira-Filho AT, Ratter JA (2002) Vegetation physiognomies and woody flora of the Cerrado biome. In: Oliveira PS, Marquis RJ (eds) The cerrados of Brazil: ecology and natural history of a neotropical savanna. Columbia University Press, New York, pp 91–120
Peterson BG, Carl P (2014) Performance analytics: econometric tools for performance and risk analysis. R package version 1.4.3541. https://CRAN.R-project.org/package=PerformanceAnalytics
Pollnac F, Seipel T, Repath C, Rew LJ (2012) Plant invasion at landscape and local scales along roadways in the mountainous region of the Greater Yellowstone Ecosystem. Biol Invasions 14:1753–1763. https://doi.org/10.1007/s10530-012-0188-y
R Development Core Team (2016) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. ISBN 3-900051-07-0, Available at: http://www.R-project.org/. Accessed June 29 2017
Rascher KG, Große-Stoltenberg A, Maguas C et al (2011) Acacia longifolia invasion impacts vegetation structure and regeneration dynamics in open dunes and pine forests. Biol Invasions 13:1099–1113. https://doi.org/10.1007/s10530-011-9949-2
Richardson DM, Rejmánek M (2011) Trees and shrubs as invasive alien species: a global review. Divers Distrib 17:788–809. https://doi.org/10.1111/j.1472-4642.2011.00782.x
Saporetti-Junior AW, Schaefer CEGR, de Souza AL et al (2012) Influence of soil physical properties on plants of the Mussununga ecosystem, Brazil. Folia Geobot 47:29–39. https://doi.org/10.1007/s12224-011-9106-9
Thiele J, Schuckert U, Otte A (2008) Cultural landscapes of Germany are patch-corridor-matrix mosaics for an invasive megaforb. Landsc Ecol 23:453–465. https://doi.org/10.1007/s10980-008-9202-2
Thiele J, Buchholz S, Schirmel J (2018) Using resistance distance from circuit theory to model dispersal through habitat corridors. J Plant Ecol 11:385–393. https://doi.org/10.1093/jpe/rtx004
Venables WN, Ripley BD (2002) Modern applied statistics with S, 4th edn. Springer, New York
Vitousek PM, Mooney HA, Lubchenco J, Melillo JM (1997) Human domination of earth’s ecosystems. Science 277:494–499. https://doi.org/10.1126/science.277.5325.494
Yang L, Liu N, Ren H, Wang J (2009) Facilitation by two exotic Acacia: Acacia auriculiformis and Acacia mangium as nurse plants in South China. For Ecol Manag 257:1786–1793. https://doi.org/10.1016/j.foreco.2009.01.033
Acknowledgements
We thank Coordination for the Improvement of Higher Education Personnel (CAPES) and the European Union’s Seventh Framework Programme FP7-PEOPLE-2010-IRSES (“INSPECTED.NET” Project – Proposal No. 269206) for the fellowships granted to GH during his PhD; CAPES and the Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG) for the postdoctoral fellowship to GH; Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for the productivity fellowship to JAAMN (No. 307591/2016-6); Geovane S. Siqueira, Eric K.O. Hattori, Pedro L. Viana and Mariana O. Bünger for plant identification; Lívia C. de Siqueira, Hugo G. Cândido, Nathália V.H. Safar, Eric K.O. Hattori, Leonardo R.M. Palmeira, Alex J. P. Coelho, Gabriel R. Silva and Rafael D. Marques for the assistance provided during field work; Luiz F.S. Magnago, Fabio A.R. Mato, and Maria Carolina N.A. da Silva for the helpful discussions during the development of this study; the company Vale S.A. for granting us permission to perform the study at the Vale Natural Reserve; and Dave Richardson and two anonymous reviewers for the very helpful improvements that were suggested.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Heringer, G., Thiele, J., Meira-Neto, J.A.A. et al. Biological invasion threatens the sandy-savanna Mussununga ecosystem in the Brazilian Atlantic Forest. Biol Invasions 21, 2045–2057 (2019). https://doi.org/10.1007/s10530-019-01955-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-019-01955-5