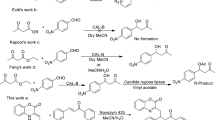
Abstract
Acylation of (hydroxyalkyl)phenols with vinyl esters by lipase B from Candida antarctica proceeded smoothly in a highly chemoselective manner, affording their alkyl esters exclusively or at least predominantly. The enzyme therefore discriminates between an alcoholic hydroxyl from a phenolic hydroxyl in addition to having versatile catalytic abilities for organic synthesis.


Similar content being viewed by others
References
Allevi P, Ciuffreda P, Longo A, Anastasia M (1998) Lipase-catalysed chemoselective monoacetylation of hydroxyalkylphenols and chemoselective removal of a single acetyl group from their diacetates. Tetrahedron Asymmetry 9:2915–2924
Anderson EM, Larsson KM, Kirk O (1998) One biocatalyst—many applications: the use of Candida antarctica B-lipase in organic synthesis. Biocatal Biotransformation 16:181–204
Bornscheuer UT, Kazlauskas RJ (2006a) Hydrolases in organic synthesis: regio- and stereoselective biotransformations, 2nd edn. Wiley-VCH, Weinheim, pp 84–140
Bornscheuer UT, Kazlauskas RJ (2006b) Hydrolases in organic synthesis: regio- and stereoselective biotransformations, 2nd edn. Wiley-VCH, Weinheim, pp 141–162
Breton GW (1997) Selective monoacetylation of unsymmetrical diols catalyzed by silica gel-supported sodium hydrogen sulfate. J Org Chem 62:8952–8954
de los Ríos AP, van Rantwijk F, Sheldon RA (2012) Effective resolution of 1-phenyl ethanol by Candida antarctica lipase B catalysed acylation with vinyl acetate in protic ionic liquids (PILs). Green Chem 14:1584–1588
Kirk O, Christensen MW (2002) Lipases from Candida Antarctica: unique biocatalysts from a unique origin. Org Process Res Dev 6:446–451
Lambusta D, Nicolosi G, Patti A, Piattelli M (1993) Enzyme-mediated regioprotection–deprotection of hydroxyl groups in (+)-catechin. Synthesis 11:1155–1158
Miyazawa T, Onishi K, Murashima T, Yamada T, Tsai S-W (2005) Resolution of non-protein amino acids via Carica papaya lipase-catalyzed enantioselective transesterification. Tetrahedron Asymmetry 16:2569–2573
Miyazawa T, Hamada M, Morimoto R, Murashima T, Yamada T (2007) Secondary alcohols act as better nucleophiles than primary alcohols in the lipase-catalyzed regioselective deacylation of dihydroxybenzenes acylated at both phenolic hydroxyls. Tetrahedron Lett 48:8334–8337
Miyazawa T, Hamada M, Morimoto R, Murashima T, Yamada T (2008) Highly regioselective propanoylation of dihydroxybenzenes mediated by Candida antarctica lipase B in organic solvents. Tetrahedron Lett 49:175–178
Natoli M, Nicolosi G, Piattelli M (1992) Regioselective alcoholysis of flavonoid acetates with lipase in an organic solvent. J Org Chem 57:5776–5778
Orita A, Mitsutome A, Otera J (1998) Distannoxane-catalyzed highly selective acylation of alcohols. J Org Chem 63:2420–2421
Paravidino M, Böhm P, Gröger H, Hanefeld U (2012) Hydrolysis and formation of carboxylic acid esters. In: Drauz K, Gröger H, May O (eds) Enzyme catalysis in organic synthesis, chap 8, 3rd edn. Wiley-VCH, Weinheim
Parmar VS, Prasad AK, Sharma NK, Singh SK, Pati HN, Gupta S (1992) Regioselective deacylation of polyacetoxy aryl-methyl ketones by lipases in organic solvents. Tetrahedron 48:6495–6498
Parmar VS, Prasad AK, Sharma NK, Varan A, Pati HN, Sharma SK, Bisht KS (1993) Lipase-catalyzed selective deacylation of peracetylated benzopyranones. J Chem Soc Chem Commun 27–29
Yamada S, Sugaki T, Matsuzaki K (1996) Twisted amides as selective acylating agents for hydroxyl groups under neutral conditions: models for activated peptides during enzymatic acyl transfer reaction. J Org Chem 61:5932–5938
Yoon KR, Hong S-P, Kong B, Choi IS (2012) Polycondensation of sebacic acid with primary and secondary hydroxyl groups containing diols catalyzed by Candida antarctica lipase B. Synth Commun 42:3504–3512
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Miyazawa, T., Yamamoto, M. & Danjo, H. Chemoselective acylation of (hydroxyalkyl)phenols catalyzed by Candida antarctica lipase B. Biotechnol Lett 35, 625–630 (2013). https://doi.org/10.1007/s10529-012-1124-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-012-1124-z