Abstract
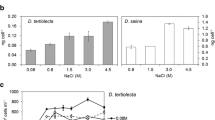
An isolate of Dunaliella salina (DUNS-1) and other two isolates (DUNS-2 and DUNS-3), collected from coastal lagoons with 14 and 30% (w/v) of NaCl, respectively, were analyzed under different saline conditions. Glycerol (380 mg l−1) and carotene (5.9 mg l−1) contents for DUNS-2 were 0.3 and 10 times higher than DUNS-3, even though both isolates were collected from the same lagoon and share a similar ribosomal DNA sequence.




Similar content being viewed by others
References
Ben-Amotz A, Shaish A, Avron M (1991) The biotechnology of cultivating Dunaliella for production of β-carotene rich algae. Bioresour Technol 38:233–235
Borowitzka MA, Siva CJ (2007) The taxonomy of the genus Dunaliella (Chlorophyta, Dunaliellales) with emphasis on the marine and halophilic species. J Appl Phycol 19:567–590
Díaz-Ordaz DP, Castrejon L, Band-Schmidt CJ, Mendoza-Maravillas A (1997) Bioecología de la Artemia en la laguna La Colorada. Ciencia y Mar 1:3–21
García F, Freile-Pelegrín Y, Robledo D (2007) Physiological characterization of Dunaliella sp. (Chlorophyta, Volvocales) from Yucatan, Mexico. Bioresour Technol 98:1359–1365
González M, Coleman AW, Gómez P, Montoya R (2001) Phylogenetic relationship among various strains of Dunaliella (Chlorophyceae) based on nuclear ITS rDNA sequences. J Phycol 37:604–611
Hoshaw RW, Rosowski JR (1973) Methods for microscopic algae. In: Stein JR (ed) 220 Handbook of phycological methods: culture methods and growth measurements. 221. Cambridge University Press, London, UK, pp 53–68
Kaçka A, Dönmez G (2008) Isolation of Dunaliella spp. from a hypersaline lake and their ability to accumulate glycerol. Bioresour Technol 99:8348–8352
Loeblich LA (1982) Photosynthesis and pigments influenced by light intensity and salinity in the halophile Dunaliella salina (Chlorophyta). J Mar Biol Ass UK 62:493–508
Logares R, Rengefors K, Kremp A, Shalchian-Tabrizi K, Boltovskoy A, Tengs T, Shurtleff A, Klaveness D (2007) Phenotypically different microalgal morphospecies with identical ribosomal DNA: a case of rapid adaptive evolution? Microb Ecol 53:549–561
Logares R, Brate J, Bertilsson S, Clasen JL, Shalchian-Tabrizi K, Rengefors K (2009) Infrequent marine–freshwater transitions in the microbial world. Trends Microbiol 17:414–422
Loret P, Tengs T, Villareal TA, Singler H, Richardson B, McGuire P, Morton S, Busman M, Campbell L (2002) No difference found in ribosomal DNA sequences from physiologically diverse clones of Karenia brevis (Dinophyceae) from the Gulf of Mexico. J Plankton Res 24:735–739
McFeeters RF (1993) Single-Injection HPLC analysis of acids, sugars, and alcohols in cucumber fermentations. J Agric Food Chem 41:1439–1443
Melis A, Neidhardt J, Benemann JR (1999) Dunaliella salina (Chlorophyta) with small chlorophyll antenna sizes exhibit higher photosynthetic productivities and photon use efficiencies than normally pigmented cells. J Appl Phycol 10:515–525
Montaño-Moctezuma G, Bückle-Ramirez LF (1996) Variations in some physico-chemical parameters in a hypersaline coastel lagoon of Baja California, Mexico. Int J Salt Lake Res 4:265–280
Olmos J, Paniagua J, Contreras R, Trujillo L (2002) Molecular identification of β-carotene hyper-producing strains of Dunaliella from saline environments using species-specific oligonucleotides. Biotech Letters 24:365–369
Olmos J, Ochoa L, Paniagua J, Contreras R (2009) DNA fingerprinting differentiation between β-carotene hyperproducer strains of Dunaliella from around the world. Saline Systems 5:5
Ramos A, Coesel S, Marques A, Rodriguez M, Baumgartner A, Noronha J, Rauter A, Brenig B, Varela J (2008) Isolation and characterization of a stress-inducible Dunaliella salina Lcy-β gene encoding a functional lycopene β-cyclase. Appl Microbiol Biotechnol 79:819–828
Rojas-Herrera R, Narváez-Zapata JA, Zamudio-Maya M, Mena-Martínez ME (2008) A simple silica-based method for metagenomic DNA extraction from soil and sediments. Mol Biotech 40:13–17
Sánchez-Estudillo L, Freile-Pelegrin Y, Rivera-Madrid R, Robledo D, Narváez-Zapata JA (2006) Regulation of two photosynthetic pigments-related genes during stress-induced pigments formation in the green alga Dunaliella salina. Biotech Letters 28:787–791
Stonynova-Bakalova E, Toncheva-Panova T (2003) Subcellular adaptation to salinity in Dunaliella salina. Biol Plantarum 47:233–236
Acknowledgments
This research was financed by the CONACyT (Grant J46435-Z) and by the Instituto Politécnico Nacional (Grant GPI-20100700).
Author information
Authors and Affiliations
Corresponding author
Additional information
An erratum to this article can be found at http://dx.doi.org/10.1007/s10529-011-0599-3
Rights and permissions
About this article
Cite this article
Narváez-Zapata, J.A., Rojas-Herrera, R., López-Uc, Y. et al. Different physiological responses influenced by salinity in genetically related Dunaliella salina isolates. Biotechnol Lett 33, 1021–1026 (2011). https://doi.org/10.1007/s10529-011-0523-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-011-0523-x