Abstract
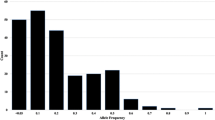
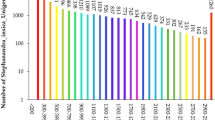
Dendrocalamus hamiltonii is a giant bamboo species native to Indian subcontinent with high economic importance. Nevertheless, highly outcross nature and flowering once in decades impose severe limitation in its propagation. Identification and mixed cultivation of genetically diverse genotypes may assist successful breeding and natural recombination of desirable traits. Characterization of existing genetic diversity and population structure are indispensable for efficient implementation of such strategies, which is facing a major challenge due to non-availability of sequence-based markers for the species. In this study, 8121 EST-SSR markers were mined from D. hamiltonii transcriptome data. Among all, tri-repeats were most represented (52%), with the abundance of CCG/CGG repeat motif. A set of 114 polymorphic markers encompassing epigenetic regulators, transcription factors, cell cycle regulators, signaling, and cell wall biogenesis, detected polymorphism and interaction (in silico) with important genes, that might have role in bamboo growth and development. Genetic diversity and population structure of the three D. hamiltonii populations (72 individuals) revealed moderate to high-level genetic diversity (mean alleles per locus: 5.8; mean PIC: 0.44) using neutral EST-SSR markers. AMOVA analysis suggests maximum diversity (59%) exists within population. High genetic differentiation (Gst = 0.338) and low gene flow (Nm = 0.49) were evident among populations. Further, PCoA, dendrogram, and Bayesian STRUCTURE analysis clustered three populations into two major groups based on geographical separations. In future, SSR marker resources created can be used for systematic breeding and implementation of conservation plans for sustainable utilization of bamboo complex.




Similar content being viewed by others
References
Abdelkrim J, Robertson BC, Stanton J-AL, Gemmell NJ (2009) Fast, cost-effective development of species-specific microsatellite markers by genomic sequencing. Biotechniques 46:185
Ambreen H, Kumar S, Variath MT, et al (2015) Development of genomic microsatellite markers in Carthamus tinctorius L.(safflower) using next generation sequencing and assessment of their cross-species transferability and utility for diversity analysis. PLoS ONE 10:e0135443
Anderson JA, Churchill GA, Autrique JE, et al (1993) Optimizing parental selection for genetic linkage maps. Genome 36:181–186
Bhandawat A, Sharma V, Sharma H et al (2013) Development and crosstransferability of functionally relevant microsatellite markers in Dendrocalamus latiflorus and related bamboo species. TC 5:5
Bhandawat A, Singh G, Raina AS et al (2016) Development of genic SSR marker resource from RNA-Seq data in Dendrocalamus latiflorus. J Plant Biochem Biotechnol 25:179–190
Bhandawat A, Singh G, Seth R et al (2017) Genome-wide transcriptional profiling to elucidate key candidates involved in bud burst and rattling growth in a subtropical bamboo (Dendrocalamus hamiltonii). Front Plant Sci 7:2038
Bhatt BP, Singha LB, Singh K, Sachan MS (2003) Some commercial edible bamboo species of North East India: production, indigenous uses, cost-benefit and management strategies. Bamboo Sci Cult 17:4–20
Bystriakova N, Kapos V, Lysenko I, Stapleton CMA (2003) Distribution and conservation status of forest bamboo biodiversity in the Asia-Pacific Region. Biodivers Conserv 12:1833–1841
Das P, Rout GR (1994) Analysis of current methods and approaches on the micropropagation of bamboo. Proc Natl Acad Sci India Sect B 64:235
Doyle JJ, Doyle JL (1990) Isolation of DNA from small amounts of plant tissues. BRL Focus 12:13–15
Earl DA (2012) Structure Harvester: a website and program for visualizing STRUCTURE output and implementing the Evanno method. Conserv Genet Resour 4:359–361
Epplen C, Melmer G, Siedlaczck I et al (1993) On the essence of “meaningless” simple repetitive DNA in eukaryote genomes. In: Pena SDJ, Chakraborty R, Epplen JT, Jeffreys AJ (eds) DNA fingerprinting: state of the Science. Springer, Dordrecht, pp 29–45
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software structure: a simulation study. Mol Ecol 14:2611–2620
Falush D, Stephens M, Pritchard JK (2007) Inference of population structure using multilocus genotype data: dominant markers and null alleles. Mol Ecol Notes 7:574–578
Foll M, Gaggiotti O (2008) A genome-scan method to identify selected loci appropriate for both dominant and codominant markers: a Bayesian perspective. Genetics 180:977–993
Ghorai A, Sharma A (1980) Bambuseae—a review. Feddes Repert 91:281–299
Hancock JM, Simon M (2005) Simple sequence repeats in proteins and their significance for network evolution. Gene 345:113–118
He C, Cui K, Zhang J et al (2013) Next-generation sequencing-based mRNA and microRNA expression profiling analysis revealed pathways involved in the rapid growth of developing culms in Moso bamboo. BMC Plant Biol 13:119. https://doi.org/10.1186/1471-2229-13-119
Jakobsson M, Rosenberg NA (2007) CLUMPP: a cluster matching and permutation program for dealing with label switching and multimodality in analysis of population structure. Bioinformatics 23:1801–1806
Janes JK, Miller JM, Dupuis JR et al (2017) The K = 2 conundrum. Mol Ecol 26(14):3594–3602
Lessard G, Chouinard A (1980) Bamboo research in Asia: proceedings of a workshop held in Singapore, 28–30 May 1980. In: Bamboo research in Asia: proceedings of a workshop held in Singapore, 28–30 May 1980. International Development Research Centre.
Li Y, Korol AB, Fahima T et al (2002) Microsatellites: genomic distribution, putative functions and mutational mechanisms: a review. Mol Ecol 11:2453–2465
Li Y-C, Korol AB, Fahima T, Nevo E (2004) Microsatellites within genes: structure, function, and evolution. Mol Biol Evol 21:991–1007
Liu M, Qiao G, Jiang J et al (2012) Transcriptome sequencing and de novo analysis for ma bamboo (Dendrocalamus latiflorus Munro) using the Illumina platform. PLoS ONE 7:e46766
Liu T, Zhu S, Fu L, et al (2013) Development and characterization of 1827 expressed sequence tag-derived simple sequence repeat markers for ramie (Boehmeria nivea L. Gaud). PLoS ONE 8:e60346
Loveless MD, Hamrick JL (1984) Ecological determinants of genetic structure in plant populations. Annu Rev Ecol Syst 15:65–95
Madlung A (2013) Polyploidy and its effect on evolutionary success: old questions revisited with new tools. Heredity (Edinb) 110:99–104
Manel S, Conord C, Després L (2009) Genome scan to assess the respective role of host-plant and environmental constraints on the adaptation of a widespread insect. BMC Evol Biol 9:288
Metzgar D, Bytof J, Wills C (2000) Selection against frameshift mutations limits microsatellite expansion in coding DNA. Genome Res 10:72–80
Parida SK, Pandit A, Gaikwad K et al (2010) Functionally relevant microsatellites in sugarcane unigenes. BMC Plant Biol 10(1):2511
Parisod C, Joost S (2010) Divergent selection in trailing-versus leading-edge populations of Biscutella laevigata. Ann Bot 105:655–660
Patel RK, Jain M (2012) NGS QC Toolkit: a toolkit for quality control of next generation sequencing data. PLoS ONE 7:e30619
Peakall ROD, Smouse PE (2006) GENALEX 6: genetic analysis in Excel. Population genetic software for teaching and research. Mol Ecol Notes 6:288–295
Perrier X, Jacquemoud-Collet JP (2006) DARwin software: dissimilarity analysis and representation for windows. https://darwin.cirad.fr/õ [accessed 1 March 2013]
Pfeiffer T, Roschanski AM, Pannell JR et al (2011) Characterization of microsatellite loci and reliable genotyping in a polyploid plant, Mercurialis perennis (Euphorbiaceae). J Hered 102(4):479–488
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure using multilocus genotype data. Genetics 155:945–959
Riechmann JL, Heard J, Martin G et al (2000) Arabidopsis transcription factors: genome-wide comparative analysis among eukaryotes. Science 290:2105–2110
Rosenberg NA (2004) DISTRUCT: a program for the graphical display of population structure. Mol Ecol Notes 4:137–138
Sampson JF, Byrne M (2012) Genetic diversity and multiple origins of polyploid Atriplex nummularia Lindl. (Chenopodiaceae). Biol J Linn Soc 105:218–230
Shukla U, Das D (1981) On the occurrence of some medicinal grasses in Eastern India. Indian J For
Slatkin M, Barton NH (1989) A comparison of three indirect methods for estimating average levels of gene flow. Evolution 43(7):1349–1368
Smoot ME, Ono K, Ruscheinski J et al (2011) Cytoscape 2.8: new features for data integration and network visualization. Bioinformatics 27:431–432
Tatusov RL, Fedorova ND, Jackson JD et al (2003) The COG database: an updated version includes eukaryotes. BMC Bioinform 4(1):41
Tautz D, Renz M (1984) Simple sequences are ubiquitous repetitive components of eukaryotic genomes. Nucleic Acids Res 12:4127–4138
te Beest M, Le Roux JJ, Richardson DM et al (2011) The more the better? The role of polyploidy in facilitating plant invasions. Ann Bot 109(1):19–45
Tewari DN (1992) A monograph on teak (Tectona grandis Linn. f.). International book distributors
Thiel T, Michalek W, Varshney R, Graner A (2003) Exploiting EST databases for the development and characterization of gene-derived SSR-markers in barley (Hordeum vulgare L.). Theor Appl Genet 106:411–422
Unamba CIN, Nag A, Sharma RK (2015) Next generation sequencing technologies: the doorway to the unexplored genomics of non-model plants. Front Plant Sci 6:1074
Varshney RK, Graner A, Sorrells ME (2005) Genic microsatellite markers in plants: features and applications. Trends Biotechnol 23:48–55
Xiao Y, Zhou L, Xia W et al (2014) Exploiting transcriptome data for the development and characterization of gene-based SSR markers related to cold tolerance in oil palm (Elaeis guineensis). BMC Plant Biol 14(1):384
Yang H-Q, An M-Y, Gu Z-J, Tian B (2012) Genetic diversity and differentiation of Dendrocalamus membranaceus (Poaceae: Bambusoideae), a declining bamboo species in Yunnan, China, as based on inter-simple sequence repeat (ISSR) analysis. Int J Mol Sci 13:4446–4457
Ye J, Fang L, Zheng H et al (2006) WEGO: a web tool for plotting GO annotations. Nucleic Acids Res 34:W293–W297
You FM, Huo N, Gu YQ et al (2008) BatchPrimer3: a high throughput web application for PCR and sequencing primer design. BMC Bioinform 9(1):253
Zalapa JE, Cuevas H, Zhu H et al (2012) Using next-generation sequencing approaches to isolate simple sequence repeat (SSR) loci in the plant sciences. Am J Bot 99:193–208
Zhang H, Zhuang S, Sun B, et al (2014). Estimation of biomass and carbon storage of moso bamboo (Phyllostachys pubescens Mazel ex Houz.) in southern China using a diameter–age bivariate distribution model. Forestry 87(5):674–82.
Acknowledgements
We acknowledge funding from CSIR research Grant MLP071. This is IHBT communication No. 4126.
Author information
Authors and Affiliations
Contributions
AB, RKS: Conceived and designed the experiments; AB, V.S., RS: Performed the experiments; AB, AN, PS, RS: Analyzed the data; AB, RKS: wrote the paper; JK: Helped in data interpretations; RKS: Editing and approval of final version of manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10528_2019_9914_MOESM1_ESM.tif
Fig. S1 Classification of Simple Sequence Repeats (SSRs) derived from D. hamiltonii transcriptome. Supplementary file1 (TIFF 2007 kb)
10528_2019_9914_MOESM2_ESM.tif
Fig. S2 Kyoto Encyclopedia of Gene and Genomes (KEGG) classification of SSR transcripts under four major categories A: cellular process, B: Environmental Information Processing, C: Genetic Information Processing and D: Metabolism. Supplementary file2 (DOCX 1565 kb)
10528_2019_9914_MOESM3_ESM.tif
Fig. S3 Eukaryotic Cluster of Orthologous Groups (KOG) classification of SSR transcripts. Supplementary file3 (DOCX 1361 kb)
10528_2019_9914_MOESM5_ESM.tif
Fig. S5 Gene ontology enrichment of SSR transcripts under biological process category. Supplementary file5 (TIFF 263 kb)
10528_2019_9914_MOESM6_ESM.tif
Fig. S6 Gene ontology enrichment of SSR transcripts under molecular function category. Supplementary file6 (TIFF 156 kb)
10528_2019_9914_MOESM7_ESM.tif
Fig. S7 Gene ontology enrichment of SSR transcripts under cellular component category. Supplementary file7 (TIFF 256 kb)
10528_2019_9914_MOESM8_ESM.tif
Fig. S8 BayeScan 2.0 plot of 231 alleles representing 40 SSR locus across 72 individuals from 3 populations from India. FST is plotted against the log10 of the posterior odds (PO). The vertical line shows the critical PO used for identifying outlier markers. Two markers on the right side of the vertical line are candidates for being under positive selection. Supplementary file8 (TIFF 14487 kb)
10528_2019_9914_MOESM9_ESM.tif
Fig. S9 Multiple sequence alignment of allelic variants of SSR locus: (a) DHTMS-45878; GAG-repeat, (b) DHTMS-1233; TCA-repeat, (c) DHTMS-22356; CTT-repeat. Region highlighted in green represents SSR locus, while dark region represents indels/point mutations. Supplementary file9 (TIFF 803 kb)
Rights and permissions
About this article
Cite this article
Bhandawat, A., Sharma, V., Singh, P. et al. Discovery and Utilization of EST-SSR Marker Resource for Genetic Diversity and Population Structure Analyses of a Subtropical Bamboo, Dendrocalamus hamiltonii. Biochem Genet 57, 652–672 (2019). https://doi.org/10.1007/s10528-019-09914-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10528-019-09914-4