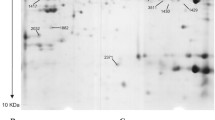
Abstract
Skeletal muscle ageing is characterized by a progressive and dramatic loss of muscle mass and strength leading to decreased muscular function resulting in muscle weakness which is often referred to as sarcopenia. Following the standardisation of “omics” approaches to study the genome (genomics) and the transcriptome (transcriptomics), the study of the proteins encoded by the genome, referred to as proteomics, is a tremendous challenge. Unlike the genome, the proteome varies in response to many physiological or pathological factors. In addition, the proteome is orders of magnitude more complex than the transcriptome due to post-translational modifications, protein oxidation and limited protein degradation. Proteomic studies, including the analysis of protein abundance as well as post-translational modified proteins have been shown to provide valuable information to unravel the key molecular pathways implicated in complex biological processes, such as tissue and organ ageing. In this article, we will describe proteomic approaches for the analysis of protein abundance as well as the specific protein targets for oxidative damage upon oxidative stress and/or during skeletal muscle ageing.



Similar content being viewed by others
References
Ahmed EK, Rogowska-Wrzesinska A, Roepstorff P, Bulteau AL, Friguet B (2010) Protein modification and replicative senescence of WI-38 human embryonic fibroblasts. Aging Cell 9(2):252–272. doi:10.1111/j.1474-9726.2010.00555.x
Andersson DC, Betzenhauser MJ, Reiken S, Meli AC, Umanskaya A, Xie W, Shiomi T, Zalk R, Lacampagne A, Marks AR (2011) Ryanodine receptor oxidation causes intracellular calcium leak and muscle weakness in aging. Cell Metab 14:196–207
Bandopadhyay R, Kingsbury AE et al (2004) The expression of DJ-1 (PARK7) in normal human CNS and idiopathic Parkinson’s disease. Brain 127:420–430
Baraibar MA, Friguet B (2012) Changes of the proteasomal system during the aging process. Prog Mol Biol Transl Sci 109:249–275. doi:10.1016/B978-0-12-397863-9.00007-9
Baraibar MA, Friguet B (2013) Oxidative proteome modifications target specific cellular pathways during oxidative stress, cellular senescence and aging. Exp Gerontol. doi: 10.1016/j.exger.2012.10.007
Baraibar MA, Hyzewicz J, Rogowska-Wrzesinska A, Ladouce R, Roepstorff P, Mouly V, Friguet B (2011) Oxidative stress-induced proteome alterations target different cellular pathways in human myoblasts. Free Radic Biol Med 51(8):1522–1532. doi:10.1016/j.freeradbiomed.2011.06.032
Baraibar MA, Liu L, Ahmed EK, Friguet B (2012a) Protein oxidative damage at the crossroads of cellular senescence, aging, and age-related diseases. Oxid Med Cell Longev 2012:919832. doi:10.1155/2012/919832
Baraibar MA, Barbeito AG, Muhoberac BB, Vidal R (2012b) A mutant light-chain ferritin that causes neurodegeneration has enhanced propensity toward oxidative damage. Free Radic Biol Med 52(9):1692–1697. doi:10.1016/j.freeradbiomed.2012.02.015
Baraibar MA, Ladouce R, Friguet B (2012c) A method for detecting and/or quantifying carbonylated proteins, PCT/EP2012/061749. http://patentscope.wipo.int/search/en/WO2012175519. Accessed 27 Dec 2012
Barreiro E, Hussain SN (2010) Protein carbonylation in skeletal muscles: impact on function. Antioxid Redox Signal 12(3):417–429. doi:10.1089/ars.2009.2808
Barreiro E, Coronell V, Laviña B et al (2006) Aging, sex differences, and oxidative stress in human respiratory and limb muscles. Free Radic Biol Med 41(5):797–809
Baumgartner RN, Koehler KM, Gallagher D, Romero L, Heymsfield SB, Ross RR, Garry PJ, Lindeman RD (1998) Epidemiology of sarcopenia among the elderly in New Mexico. Am J Epidemiol 147(8):755–763
Berlett BS, Stadtman ER (1997) Protein oxidation in aging, disease, and oxidative stress. J Biol Chem 272(33):20313–20316
Biteau B, Labarre J, Toledano MB (2003) ATP-dependent reduction of cysteine-sulphinic acid by S. cerevisiae sulphiredoxin. Nature 425(6961):980–984
Breusing N, Grune T (2008) Regulation of proteasome-mediated protein degradation during oxidative stress and aging. Biol Chem 389:203–209
Capitanio D, Vasso M, Fania C, Moriggi M, Viganò A, Procacci P, Magnaghi V, Gelfi C (2009) Comparative proteomic profile of rat sciatic nerve and gastrocnemius muscle tissues in ageing by 2-D DIGE. Proteomics 9:2004–2020. doi:10.1002/pmic.200701162
Chan CY, McDermott JC, Siu KW (2011) Secretome analysis of skeletal myogenesis using SILAC and shotgun proteomics. Int J Proteomics 2011:329467. doi:10.1155/2011/329467
Choksi KB, Papaconstantinou J (2008) Age-related alterations in oxidatively damaged proteins of mouse heart mitochondrial electron transport chain complexes. Free Radic Biol Med 44(10):1795–1805. doi:10.1016/j.freeradbiomed.2008.01.032
Clark KA, McElhinny AS, Beckerle MC, Gregorio CC (2002) Striated muscle cytoarchitecture: an intricate web of form and function. Annu Rev Cell Dev Biol 18:637–706
Donoghue P, Staunton L, Mullen E, Manning G, Ohlendieck K (2010) DIGE analysis of rat skeletal muscle proteins using nonionic detergent phase extraction of young adult versus aged gastrocnemius tissue. J Proteomics 73(8):1441–1453. doi:10.1016/j.jprot.2010.01.014
Doran P, Gannon J, O’Connell K, Ohlendieck K (2007) Ageing skeletal muscle shows a drastic increase in the small heat shock proteins B-crystallin/HspB5 and cvHsp/HspB7. Eur J Cell Biol 86:629–640
Doran P, O’Connell K, Gannon J, Kavanagh M, Ohlendieck K (2008) Opposite pathobiochemical fate of pyruvate kinase and adenylate kinase in aged rat skeletal muscle as revealed by proteomic DIGE analysis. Proteomics 8:364–377
Droge W (2002) Free radicals in the physiological control of cell function. Physiol Rev 82:47–95
Duguez S, Duddy W, Johnston H, Laine J, Le Bihan MC, Brown KJ, Bigot A, Hathout Y, Butler-Browne G, Partridge T (2013) Dystrophin deficiency leads to disturbance of LAMP1-vesicle-associated protein secretion. Cell Mol Life Sci. doi:10.1007/s00018-012-1248-2
Engerson TD, McKelvey TG, Rhyne DB, Boggio EB, Snyder SJ, Jones HP (1987) Conversion of xanthine dehydrogenase to oxidase in ischemic rat tissues. J Clin Invest 79:1564–1570
Engler D (2007) Hypothesis: Musculin is a hormone secreted by skeletal muscle, the body’s largest endocrine organ. Evidence for actions on the endocrine pancreas to restrain the beta-cell mass and to inhibit insulin secretion and on the hypothalamus to co-ordinate the neuroendocrine and appetite responses to exercise. Acta Biomed 78(Suppl 1):156–206
Evans WJ (1995) What is sarcopenia? J Gerontol A Biol Sci Med Sci 50 Spec No, 5–8
Fedorova M, Kuleva N, Hoffmann R (2010) Identification, quantification, and functional aspects of skeletal muscle protein-carbonylation in vivo during acute oxidative stress. J Proteome Res 9(5):2516–2526. doi:10.1021/pr901182r
Ferreira LF, Reid MB (2008) Muscle-derived ROS and thiol regulation in muscle fatigue. J Appl Physiol 104:853–860
Friguet B (2002) Aging of proteins and the proteasome. Prog Mol Subcell Biol 29:17–33
Friguet B (2006) Oxidized protein degradation and repair in ageing and oxidative stress. FEBS Lett 580(12):2910–2916. doi:10.1016/j.febslet.2006.03.028
Friguet B, Bulteau AL, Chondrogianni N, Conconi M, Petropoulos I (2000) Protein degradation by the proteasome and its implications in aging. Ann N Y Acad Sci 908:143–154
Fugere NA, Ferrington DA, Thompson LV (2006) Protein nitration with aging in the rat semimembranosus and soleus muscles. J Gerontol Ser A Biol Sci Med Sci 61(8):806–812
Gannon J, Staunton L, O’Connell K, Doran P, Ohlendieck K (2008) Phosphoproteomic analysis of aged skeletal muscle. Int J Mol Med 22:33–42
Gannon J, Doran P, Kirwan A, Ohlendieck K (2009) Drastic increase of myosin light chain MLC-2 in senescent skeletal muscle indicates fast-to-slow fibre transition in sarcopenia of old age. Eur J Cell Biol 88:685–700. doi:10.1016/j.ejcb.2009.06.004
Gelfi C, Vigano A, Ripamonti M, Pontoglio A, Begum S, Pellegrino MA, Grassi B, Bottinelli R, Wait R, Cerretelli P (2006) The human muscle proteome in aging. J Proteome Res 5:1344–1353
Gianni P, Jan KJ, Douglas MJ, Stuart PM, Tarnopolsky MA (2004) Oxidative stress and the mitochondrial theory of aging in human skeletal muscle. Exp Gerontol 39(9):1391–1400
Gilmore JM, Washburn MP (2010) Advances in shotgun proteomics and the analysis of membrane proteomes. J Proteomics 73(11):2078–2091. doi:10.1016/j.jprot.2010.08.005
Giulivi C, Traaseth NJ, Davies KJ (2003) Tyrosine oxidation products: analysis and biological relevance. Amino Acids 25(3–4):227–232
Goldfarb LG, Dalakas MC (2009) Tragedy in a heartbeat: malfunctioning desmin causes skeletal and cardiac muscle disease. J Clin Invest 119:1806–1813
Gorg A, Drews O, Luck C, Weiland F, Weiss W (2009) 2-DE with IPGs. Electrophoresis 30(Suppl 1):S122–S132. doi:10.1002/elps.200900051
Görg A, Weiss W, Dunn MJ (2004) Current two-dimensional electrophoresis technology for proteomics. Proteomics 4:3665–3685
Henningsen J, Rigbolt KT, Blagoev B, Pedersen BK, Kratchmarova I (2010) Dynamics of the skeletal muscle secretome during myoblast differentiation. Mol Cell Proteomics 9(11):2482–2496. doi:10.1074/mcp.M110.002113
Hojlund K, Yi Z, Hwang H, Bowen B, Lefort N, Flynn CR, Langlais P, Weintraub ST, Mandarino LJ (2008) Characterization of the human skeletal muscle proteome by one-dimensional gel electrophoresis and HPLC-ESI-MS/MS. Mol Cell Proteomics 7:257–267
Horiuchi S, Araki N (1994) Advanced glycation end products of the Maillard reaction and their relation to aging. Gerontology 40(Suppl 2):10–15
Jackson MJ, Papa S, Bolanos J et al (2002) Antioxidants, reactive oxygen and nitrogen species, gene induction and mitochondrial function. Mol Aspects Med 23:209–285
Kanski J, Hong SJ, Schöneich C (2005) Proteomic analysis of protein nitration in ageing skeletal muscle and identification of nitrotyrosine-containing sequences in vivo by nanoelectrospray ionization tandem mass spectrometry. J Biol Chem 280:24261–24266
Kondo H, Miura M, Itokawa Y (1991) Oxidative stress in skeletal muscle atrophied by immobilization. Acta Physiol Scand 142:527–528
Kondo H, Nishino K, Itokawa Y (1994) Hydroxyl radical generation in skeletal muscle atrophied by immobilization. FEBS Lett 349:169–172
Le Bihan MC, Bigot A, Jensen SS, Dennis JL, Rogowska-Wrzesinska A, Lainé J, Gache V, Furling D, Jensen ON, Voit T, Mouly V, Coulton GR, Butler-Browne G (2012) In-depth analysis of the secretome identifies three major independent secretory pathways in differentiating human myoblasts. J Proteomics 77:344–356. doi:10.1016/j.jprot.2012.09.008
Lefort N, Yi Z, Bowen B, Glancy B, De Filippis EA, Mapes R, Hwang H, Flynn CR, Willis WT, Civitarese A, Højlund K, Mandarino LJ (2009) Proteome profile of functional mitochondria from human skeletal muscle using one-dimensional gel electrophoresis and HPLC ESI-MS/MS. J Proteomics 72:1046–1060. doi:10.1016/j.jprot.2009.06.011
Levine RL, Stadtman ER (2001) Oxidative modification of proteins during aging. Exp Gerontol 36(9):1495–1502
Levine RL, Wehr N, Williams JA, Stadtman ER, Shacter E (2000) Determination of carbonyl groups in oxidized proteins. Methods Mol Biol 99:15–24
Lombardi A, Silvestri E, Cioffi F, Senese R, Lanni A, Goglia F, de Lange P, Moreno M (2009) Defining the transcriptomic and proteomic profiles of rat ageing skeletal muscle by the use of a cDNA array, 2D- and Blue native-PAGE approach. J Proteomics 72:708–721. doi:10.1016/j.jprot.2009.02.007
Madian AG, Regnier FE (2010) Proteomic identification of carbonylated proteins and their oxidation sites. J Proteome Res 9(8):3766–3780. doi:10.1021/pr1002609
Marzani B, Felzani G, Bellomo RG, Vecchiet J, Marzatico F (2005) Human muscle ageing: ROS-mediated alterations in rectus abdominis and vastus lateralis muscles. Exp Gerontol 40:959–965
Meunier B, Dumas E, Piec I, Béchet D, Hébraud M, Hocquette JF (2007) Assessment of hierarchical clustering methodologies for proteomic data mining. J Proteome Res 6:358–366
Moreau R, Heath SH, Doneanu CE, Lindsay JG, Hagen TM (2003) Age-related increase in 4-hydroxynonenal adduction to rat heart alpha-ketoglutarate dehydrogenase does not cause loss of its catalytic activity. Antioxid Redox Signal 5(5):517–527
O’Connell K, Ohlendieck K (2009) Proteomic DIGE analysis of the mitochondria-enriched fraction from aged rat skeletal muscle. Proteomics 9:5509–5524
Pansarasa O, Castagna B, Colombi J, Vecchiet J, Felzani C, Marzatico F (2000) Age and sex differences in human skeletal muscle: role of reactive oxygen species. Free Radic Res 33(3):287–293
Petropoulos I, Friguet B (2006) Maintenance of proteins and aging: the role of oxidized protein repair. Free Radic Res 40(12):1269–1276
Piec I, Listrat A, Alliot J, Chambon C, Taylor RG, Bechet D (2005) Differential proteome analysis of aging in rat skeletal muscle. FASEB J 19(9):1143–1145. doi:10.1096/fj.04-3084fje
Powers SK, Jackson MJ (2008) Exercise-induced oxidative stress: cellular mechanisms and impact on muscle force production. Physiol Rev 88:1243–1276
Rabilloud T (2002) Two-dimensional gel electrophoresis in proteomics: old, old fashioned, but it still climbs up the mountains. Proteomics 2:3–10
Rabilloud T, Lelong C (2011) Two-dimensional gel electrophoresis in proteomics: a tutorial. J Proteomics 74(10):1829–1841. doi:10.1016/j.jprot.2011.05.040
Rabilloud T, Heller M, Gasnier F, Luche S, Rey C, Aebersold R, Benahmed M, Louisot P, Lunardi J (2002) Proteomics analysis of cellular response to oxidative stress: evidence for in vivo overoxidation of peroxiredoxins at their active site. J Biol Chem 277:19396–19401
Reid MB (2001) Nitric oxide, reactive oxygen species, and skeletal muscle contraction. Med Sci Sports Exerc 33:371–376
Roca-Rivada A, Al-Massadi O, Castelao C, Senin LL, Alonso J, Seoane LM, Garcia-Caballero T, Casanueva FF, Pardo M (2012) Muscle tissue as an endocrine organ: comparative secretome profiling of slow-oxidative and fast-glycolytic rat muscle explants and its variation with exercise. J Proteomics 75(17):5414–5425. doi:10.1016/j.jprot.2012.06.037
Rogowska-Wrzesinska A, Le Bihan MC, Thaysen-Andersen M, Roepstorff P (2013) 2D gels still have a niche in proteomics. J Proteomics. doi:10.1016/j.jprot.2013.01.010
Schiaffino S (2010) Fibre types in skeletal muscle: a personal account. Acta Physiol 199(4):451–463. doi:10.1111/j.1748-1716.2010.02130
Shenton D, Smirnova JB, Selley JN, Carroll K, Hubbard SJ, Pavitt GD, Ashe MP, Grant CM (2006) Global translational responses to oxidative stress impact upon multiple levels of protein synthesis. J Biol Chem 281:29011–29021
Shindoh C, DiMarco A, Nethery D, Supinski G (1992) Effect of PEG-superoxide dismutase on the diaphragmatic response to endotoxin. Am Rev Respir Dis 145:1350–1354
Short KR, Bigelow ML, Kahl J, Singh R, Coenen-Schimke J, Raghavakaimal S, Nair KS (2005) Decline in skeletal muscle mitochondrial function with ageing in humans. Proc Natl Acad Sci USA 102:5618–5623
Snijders T, Verdijk LB, van Loon LJ (2009) The impact of sarcopenia and exercise training on skeletal muscle satellite cells. Ageing Res Rev 8(4):328–338. doi:10.1016/j.arr.2009.05.003
Snow LM, Fugere NA, Thompson LV (2007) Advanced glycation end-product accumulation and associated protein modification in type II skeletal muscle with ageing. J Gerontol Ser A Biol Sci Med Sci 62(11):1204–1210
Stadtman ER, Levine RL (2003) Free radical-mediated oxidation of free amino acids and amino acid residues in proteins. Amino Acids 25(3–4):207–218
Staunton L, Zweyer M, Swandulla D, Ohlendieck K (2012) Mass spectrometry-based proteomic analysis of middle-aged vs. aged vastus lateralis reveals increased levels of carbonic anhydrase isoform 3 in senescent human skeletal muscle. Int J Mol Med 30:723–733. doi:10.3892/ijmm.2012.1056
Thompson LV, Durand D, Fugere NA, Ferrington DA (2006) Myosin and actin expression and oxidation in aging muscle. J Appl Physiol 101(6):1581–1587. doi:10.1152/japplphysiol.00426.2006
Ünlü M, Minden JS (2002) Difference gel electrophoresis. In: Walker JM (ed) The protein protocols handbook. Humana Press, Totowa, pp 185–196
Whidden MA, McClung JM, Falk DJ, Hudson MB, Smuder AJ, Nelson WB, Powers SK (2009) Xanthine oxidase contributes to mechanical ventilation-induced diaphragmatic oxidative stress and contractile dysfunction. J Appl Physiol 106:385–394
Yan LJ, Forster MJ (2011) Chemical probes for analysis of carbonylated proteins: a review. J Chromatogr B Anal Technol Biomed Life Sci 879(17–18):1308–1315. doi:10.1016/j.jchromb.2010.08.004
Yoon JH, Kim J, Song P, Lee TG, Suh PG, Ryu SH (2012) Secretomics for skeletal muscle cells: a discovery of novel regulators? Adv Biol Regul 52(2):340–350. doi:10.1016/j.jbior.2012.03.001
Acknowledgments
The authors are very thankful to the FP7 EU-funded project MyoAge (No. 223576), Inserm, UPMC, AFLD and the Association Française contre les Myopathies (AFM). M.A.B has received a post-doctoral fellowship from MyoAge and is currently the recipient of a post-doctoral fellowship from the Association Française contre les Myopathies (AFM). S.D. has received a post-doctoral fellowship from MyoAge. M.G. is supported by a PhD fellowship from the Conseil Régional Auvergne and Fonds Européens de Développement Régional (FEDER). The authors are grateful to Romain Ladouce for his expert assistance with the Oxi-DIGE.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Baraibar, M.A., Gueugneau, M., Duguez, S. et al. Expression and modification proteomics during skeletal muscle ageing. Biogerontology 14, 339–352 (2013). https://doi.org/10.1007/s10522-013-9426-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10522-013-9426-7