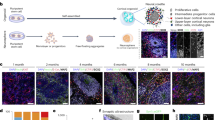
The proliferative properties of tanycyte subpopulations during postnatal development and aging were studied. Using immunohistochemical markers, we described the distribution of proliferative markers and markers of neural stem cells (NSC) in 4 tanycyte subpopulations (α1-, α2-, β1-, and β2-tanycytes). During the first postnatal week, all tanycyte subpopulations exhibit proliferative activity. During aging, β-tanycytes lose their proliferative activity and retain a limited set of NSC markers, whereas α-tanycytes maintain both the ability to proliferate and the properties of NSC throughout the entire postnatal development including aging. The data obtained significantly improve modern understanding of the proliferative potential of tanycytes and their subpopulation differences in early postnatal period and during aging.
Similar content being viewed by others
References
Aleksandrova MA, Marey MV. Stem Cells in the Brain of Mammals and Human: Fundamental and Applied Aspects. Zh. Vyssh. Nervn. Deyat. 2015;65(3):271-305. Russian. https://doi.org/10.7868/S004446771503003X
Toda T, Gage FH. Review: adult neurogenesis contributes to hippocampal plasticity. Cell Tissue Res. 2018;373(3):693-709. https://doi.org/10.1007/s00441-017-2735-4
La Rosa C, Parolisi R, Bonfanti L. Brain structural plasticity: from adult neurogenesis to immature neurons. Front. Neurosci. 2020;14:75. https://doi.org/10.3389/fnins.2020.00075
Recabal A, Caprile T, García-Robles MLA. Hypothalamic neurogenesis as an adaptive metabolic mechanism. Front. Neurosci. 2017;11:190. https://doi.org/10.3389/fnins.2017.00190
Cheng MF. Hypothalamic neurogenesis in the adult brain. Front. Neuroendocrinol. 2013;34(3):167-178. https://doi.org/10.1016/j.yfrne.2013.05.001
Rodríguez E, Guerra M, Peruzzo B, Blázquez JL. Tanycytes: A rich morphological history to underpin future molecular and physiological investigations. J. Neuroendocrinol. 2019;31(3):e12690. https://doi.org/10.1111/jne.12690
Rodríguez EM, Blázquez JL, Pastor FE, Peláez B, Peña P, Peruzzo B, Amat P. Hypothalamic tanycytes: a key component of brain-endocrine interaction. Int. Rev. Cytol. 2005;247:89-164. https://doi.org/10.1016/S0074-7696(05)47003-5
Prevot V, Dehouck B, Sharif A, Ciofi P, Giacobini P, Clasadonte J. The versatile tanycyte: a hypothalamic integrator of reproduction and energy metabolism. Endocr. Rev. 2018;39(3):333-368. https://doi.org/10.1210/er.2017-00235
Robins SC, Stewart I, McNay DE, Taylor V, Giachino C, Goetz M, Ninkovic J, Briancon N, Maratos-Flier E, Flier JS, Kokoeva MV, Placzek M. α-Tanycytes of the adult hypothalamic third ventricle include distinct populations of FGF-responsive neural progenitors. Nat. Commun. 2013;4:2049. https://doi.org/10.1038/ncomms3049
Kim JG, Suyama S, Koch M, Jin S, Argente-Arizon P, Argente J, Liu ZW, Zimmer MR, Jeong JK, Szigeti-Buck K, Gao Y, Garcia-Caceres C, Yi CX, Salmaso N, Vaccarino FM, Chowen J, Diano S, Dietrich MO, Tschöp MH, Horvath TL. Leptin signaling in astrocytes regulates hypothalamic neuronal circuits and feeding. Nat. Neurosci. 2014;17(7):908-910. https://doi.org/10.1038/nn.3725
Sharif A, Prevot V. When size matters: how astrocytic processes shape metabolism. Cell Metab. 2017;25(5):995-996. https://doi.org/10.1016/j.cmet.2017.04.016
Maggi R, Zasso J, Conti L. Neurodevelopmental origin and adult neurogenesis of the neuroendocrine hypothalamus. Front. Cell Neurosci. 2015;8:440. https://doi.org/10.3389/fncel.2014.00440
Korzhevskii DE, Sukhorukova EG, Kirik OV, Grigorev IP. Immunohistochemical demonstration of specific antigens in the human brain fixed in zinc-ethanol-formaldehyde. Eur. J. Histochem. 2015;59(3):2530. https://doi.org/10.4081/ejh.2015.2530
Paxinos G, Watson C. The Rat Brain in Stereotaxic Coordinates. New York, 2007.
Sawicka A, Seiser C. Histone H3 phosphorylation — a versatile chromatin modification for different occasions. Biochimie. 2012;94(11):2193-2201. https://doi.org/10.1016/j.biochi.2012.04.018
Komar D, Juszczynski P. Rebelled epigenome: histone H3S10 phosphorylation and H3S10 kinases in cancer biology and therapy. Clin. Epigenetics. 2020;12(1):147. https://doi.org/10.1186/s13148-020-00941-2
Juríková M, Danihel Ľ, Polák Š, Varga I. Ki67, PCNA, and MCM proteins: Markers of proliferation in the diagnosis of breast cancer. Acta Histochem. 2016;118(5):544-552. https://doi.org/10.1016/j.acthis.2016.05.002
Kirik OV, Beznin GV, Korzhevskiy DE. Proliferation markers used in histological studies. Morfologiya. 2009;136(6):95-100. Russian.
Haan N, Goodman T, Najdi-Samiei A, Stratford CM, Rice R, El Agha E, Bellusci S, Hajihosseini MK. Fgf10-expressing tanycytes add new neurons to the appetite/energy-balance regulating centers of the postnatal and adult hypothalamus. J. Neurosci. 2013;33(14):6170-6180. https://doi.org/10.1523/JNEUROSCI.2437-12.2013
Zhang S, Cui W. Sox2, a key factor in the regulation of pluripotency and neural differentiation. World J. Stem Cells. 2014;6(3):305-311. https://doi.org/10.4252/wjsc.v6.i3.305
Forouzanfar M, Lachinani L, Dormiani K, Nasr-Esfahani MH, Gure AO, Ghaedi K. Intracellular functions of RNA-binding protein, Musashi1, in stem and cancer cells. Stem Cell Res. Ther. 2020;11(1):193. https://doi.org/10.1186/s13287-020-01703-w
Zoli M, Ferraguti F, Frasoldati A, Biagini G, Agnati LF. Age-related alterations in tanycytes of the mediobasal hypothalamus of the male rat. Neurobiol. Aging. 1995;16(1):77-83. https://doi.org/10.1016/0197-4580(95)80010-o
Kostin A, Alam MA, McGinty D, Alam MN. Adult hypothalamic neurogenesis and sleep-wake dysfunction in aging. Sleep. 2021;44(2):zsaa173. https://doi.org/10.1093/sleep/zsaa173
Chaker Z, George C, Petrovska M, Caron JB, Lacube P, Caillé I, Holzenberger M. Hypothalamic neurogenesis persists in the aging brain and is controlled by energy-sensing IGF-I pathway. Neurobiol. Aging. 2016;41:64-72. https://doi.org/10.1016/j.neurobiolaging.2016.02.008
Zhang Y, Kim MS, Jia B, Yan J, Zuniga-Hertz JP, Han C, Cai D. Hypothalamic stem cells control ageing speed partly through exosomal miRNAs. Nature. 2017;548:52-57. https://doi.org/10.1038/nature23282
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Kletochnye Tekhnologii v Biologii i Meditsine, No. 4, pp. 254-260, December, 2022
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sufieva, D.A., Korzhevskii, D.E. Proliferative Markers and Neural Stem Cells Markers in Tanycytes of the Third Cerebral Ventricle in Rats. Bull Exp Biol Med 174, 564–570 (2023). https://doi.org/10.1007/s10517-023-05748-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-023-05748-8