Abstract
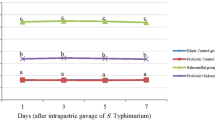
This study evaluated the hematological, biochemical, and histopathological responses of sorubim Pseudoplatystoma spp., experimentally infected with Lactococcus garvieae. Sorubim juveniles (n = 66; 21.3 ± 5.5 g; 17.1 ± 1.3 cm) were infected with L. garvieae by coelmatic inoculation (1.5 × 108 CFU mL−1) and sampling to 1, 3, 7, 14, and 21 days post-inoculation (dpi) for evaluation of hematological, biochemical parameters. Histopathological data of the spleen and kidney were estimated according to the histological alteration index (HAIn). Additionally, a decrease of hematocrit (1, 3, and 14 dpi), hemoglobin, and count of erythrocytes (3, 14, and 21 dpi), blood glucose (3 dpi), and proteinemy (14 dpi) were observed. Monocytes decreased at 7, 14, and 21 dpi; neutrophils at 3, 7, and 21 dpi; and lymphocytes at 21 dpi. Edema, hyperemia, hemorrhage, inflammatory infiltrate, and necrosis were the major histological changes observed in the spleen and kidney. HAIn was higher at 7 to 14 dpi in the spleen. Edema, hyperemia, and hyperplasia with hypertrophy of melanomacrophage centers (MMCs) associated with mild focal necrosis areas, particularly in white pulp, were observed. In the head kidney, it was observed that MMCs and melanomacrophages surrounded the capillaries and dispersed inside the sinusoidal capillaries network. Internalized bacteria by macrophages were commonly observed at 3 to 7 dpi. Tubular neogenesis characterized by nephrogenic bodies and rare MMCs at 21 dpi was observed. The experimental infection with L. garvieae caused significant hematological and biochemical responses. Pathological manifestations are closely related to the spleen and head kidneys mainly represented by hyperplasia and hypertrophy of melanomacrophage centers and mild necrosis areas.







Similar content being viewed by others
References
Adeyemi JA, Atere TG, Oyedara OO, Olabiyi KO (2013) Hematological assessment of health status of African catfish Clarias gariepinus (Burchell 1822) experimentally challenged with Escherichia coli and Vibrio fischeri. Comp Clin Pathol 23:1309–1313. https://doi.org/10.1007/s00580-013-1780-y
Agius C, Roberts RJ (2003) Melano-macrophage centers and their role in fish pathology. J Fish Dis 26:499–509. https://doi.org/10.1046/j.1365-2761.2003.00485.x
Aguado-Urda M, Rodríguez-Bertos A, Heras AI, Blanco MM, Acosta F, Cid R, Fernández-Garayzábal JF, Gibello A (2014) Experimental Lactococcus garvieae infection in zebrafish and first evidence of its ability to invade non-phagocytic cells. Vet Microbiol 171:248–254. https://doi.org/10.1016/j.vetmic.2014.03.029
Algöet M, Bayley AE, Feist SW, Wheeler RW, Verner-Jeffreys DW (2009) Susceptibility of selected freshwater fish species to a UK Lactococcus garvieae isolate. J Fish Dis 32:825–834. https://doi.org/10.1111/j.1365-2761.2009.01058.x
Alsaid M, Abuseliana AF, Daud HH, Mustapha NM, Bejo SK, Abdelhadi YM, Hamdam RH (2014) Haematological, biochemical and clinical signs changes following experimental infection of Streptococcus agalactiae in red hybrid tilapia (Oreochromis sp.). Aquac Indones 15:86–93. https://doi.org/10.21534/ai.v15i2.36
Austin B, Austin DA (2012) Bacterial fish pathogens- disease of farmed and wild fish. Springer, Dordrecht
Avci H, Birincioglu SS, Tanrikul TT, Epikmen ET, Metin N, Avsever ML (2013) Experimental Lactococcus garvieae infection in rainbow trout, Oncorhynchus mykiss, Walbaum 1792: a comparative histopathological and immunohistochemical study. J Fish Dis 37:481–495. https://doi.org/10.1111/jfd.12132
Avsever ML, Tanrikul TT, Gürsoy D, Metin S, Akşit H, Tunahgil S (2014) Investigation of certain blood parameters in rainbow trout (Oncorhynchus mykiss Walbaum, 1792) naturally infected with Lactococcus garvieae. J Fish Sci 8:114–120. https://doi.org/10.3153/jfscom.201415
Barbosa ON, Raizer J, Gonda MF, Silva JM (2011) Desempenho e coeficiente intestinal de alevinos puros e híbridos de pintados em condicionamento alimentar. R Bras Zootec 40:2621–2627. https://doi.org/10.1590/S1516-35982011001200001
Barnes A, Guyot C, Hansen BG, Mackenzie K, Horne MT, Ellis AE (2002) Resistance to serum killing may contribute to differences in the abilities of capsulate and non-capsulated isolates of Lactococcus garvieae to cause disease in rainbow trout (Oncorhynchus mykiss L.). Fish Shellfish Immunol 12:155–168. https://doi.org/10.1006/fsim.2001.0361
Bercovier H, Ghittino C, Eldar A (1997) Immunization with bacterial antigens: infection with streptococci and related organisms. Dev Biol Stand 90:153–160
Bernet D, Schmidt H, Meier W, Burkhardt-Holm P, Wahli T (1999) Histopathology in fish: proposal for a protocol to assess aquatic pollution. J Fish Dis 22:25–34. https://doi.org/10.1046/j.1365-2761.1999.00134.x
Bruno D, Noguera PA, Poppe TT (2013) A color atlas of salmonid diseases. Springer, London
Campos JL (2013) O cultivo do pintado (Pseudoplatystoma corruscans Spix & Agassiz, 1829) e outras espécies do gênero Pseudoplatystoma e seus híbridos. In: Baldisserotto B, Gomes LC (ed) Espécies nativas para a piscicultura no Brasil, 2nd. Editora da UFSM, Santa Maria, pp 335–3610
Castro R, Tafalla C (2015) Overview of fish immunity. In: Beck BH, Peatman E (eds) Mucosal health in aquaculture. Academic Press, Cambridge, pp 3–54
Castro R, Reguera-Brito M, López-Campos GH, Blanco MM, Aguado-Urda M, Fernández-Garayzábal JF, Gibello A (2017) How does temperature influences the development of lactococcosis? Transcriptomic and immunoproteomic in vitro approaches. J Fish Dis 40:1285–1297. https://doi.org/10.1111/jfd.12601
Chang PH, Lin CW, Lee YC (2002) Lactococcus garvieae infection of cultured rainbow trout, Oncorhynchus mykiss, in Taiwan and associated biophysical characteristics and histopathology. Bull Eur Assoc Fish Pathol 22:319–326
Chen SC, Lin LD, Liaw LL, Wang PC (2001) Lactococcus garvieae infection in the giant freshwater prawn Macrobrachium rosenbergii confirmed by polymerase chain reaction and 16S rDNA sequencing. Dis Aquat Org 45:45–52. https://doi.org/10.3354/dao045045
Chen SC, Liaw LL, Su HY, Co SC, Wu CY, Chaung HC, Tsai YH, Yang KL, Chen YC, Chen TH, Lin GR, Cheng SY, Lin YD, Lee JL, Lai CC, Weng YJ, Chu SY (2002) Lactococcus garvieae, a cause of disease in grey mullet, Mugil cephalus L., in Taiwan. J Fish Dis 25:727–732. https://doi.org/10.1046/j.1365-2761.2002.00415.x
Clauss TM, Dove ADM, Arnold JE (2008) Hematologic disorders of fish. Vet Clin North Am Exot Anim Pract 11:445–462. https://doi.org/10.1016/j.cvex.2008.03.007
Davies LC, Jenkins SJ, Allen JE, Taylor PR (2013) Tissue-resident macrophages. Nat Immun 14:986–995. https://doi.org/10.1038/ni.2705
Didinen BI, Yardimci B, Onuk EE, Metin S, Yildirim P (2014) Naturally Lactococcus garvieae infection in rainbow trout (Oncorhynchus mykiss Walbaum, 1972): new histopathological observations, phenotypic and molecular identification. Rev Med Vet 165:12–19
Eldar A, Ghittino C (1999) Lactococcus garvieae and Streptococcus iniae infections in rainbow trout Oncorhynchus mykiss: similar, but different diseases. Dis Aquat Org 36:227–231. https://doi.org/10.3354/dao036227
Eraclio G, Ricci G, Quattrini M, Moroni P, Fortina MG (2018) Detection of virulence-related genes in Lactococcus garvieae and their expression in response to different conditions. Folia Microbiol 63:291–298. https://doi.org/10.1007/s12223-017-0566-z
Evans JJ, Klesius PH, Shoemaker CA (2009) First isolation and characterization of Lactococcus garvieae from Brazilian Nile tilapia, Oreochromis niloticus (L.), and Pintado, Pseudoplatystoma corruscans (Spix & Agassiz). J Fish Dis 32:943–951. https://doi.org/10.1111/j.1365-2761.2009.01075.x
Fagundes M, Urbinati EC (2008) Stress in pintado (Pseudoplatystoma corruscans) during farming procedures. Aquaculture 276:112–119. https://doi.org/10.1016/j.aquaculture.2008.02.006
Fantini LE, Rodrigues RA, Nunes AL, Sanches MSS, Ushizima TT, Campos CM (2013) Rendimento de carcaça de surubins Pseudoplatystoma spp. produzidos em tanque rede e viveiro. Rev Bras Saúde Prod Anim 14:538–545. https://doi.org/10.1590/S1519-99402013000300019
Ferguson HW (2006) Systemic pathology of fish. In: A text and Atlas of normal tissues in teleosts and their responses in diseases. Scotian Press, Leicester
Food and Agriculture Organization (2019) Fisheries and aquaculture information and statistics branch. FAO, Roma
Fukushima HCS, Leal CAG, Cavalcante RB, Figueiredo HCP, Arijo S, Moriñigo MA, Ishikawa M, Borra RC, Ranzani-Paiva MJT (2017) Lactococcus garvieae outbreaks in Brazilian farms Lactococosis in Pseudoplatystoma sp.- development of an autogenous vaccine as a control strategy. J Fish Dis 40:263–272. https://doi.org/10.1111/jfd.12509
Garcia F, Moraes FR (2009) Hematologia e sinais clínicos de Piaractus mesopotamicus infectados experimentalmente com Aeromonas hydrophila. Acta Sci Biol Sci 31:17–21. https://doi.org/10.4025/actascibiolsci.v31i1.308
Kang SH, Shin GW, ShinYW PKJ, Kim YR, Lee EY, Lee EG, Huh NE, Ju OM, Jung HH (2004) Experimental evaluation of pathogenicity of Lactococcus garvieae in black rockfish (Sebastes schlegeli). J Vet Sci 5:387–390
Kav K, Erganis O (2008) Antibiotic susceptibility of Lactococcus garvieae in rainbow trout (Oncorhynchus mykiss) farms. Bull Vet Inst Pulawy 52:223–226
Kawanishi M, Yoshida T, Yagashiro S, Kijima M, Yagyu K, Nakai T, Murakami M, Morita H, Suzuki S (2006) Differences between Lactococcus garvieae isolated from the genus Seriola in Japan and those isolated from other animals (trout, terrestrial animals from Europe) with regard to pathogenicity, phage susceptibility and genetic characterization. J Appl Microbiol 101:496–504. https://doi.org/10.1111/j.1365-2672.2006.02951.x
Kotob MH, Menanteau-Ledouble S, Kumar G, Abdelzaher M, El-Matbouli M (2016) The impact of co-infections on fish: a review. Vet Res. https://doi.org/10.1186/s13567-016-0383-4
Kusuda K, Hamaguchi M (1988) Extracellular and intracellular toxins of Streptococcus sp. isolated from yellowtail. Bull Eur Assoc Fish Pathol 8:9–10
Magnadóttir B (2006) Innate immunity of fish (overview). Fish Shellfish Immunol 20:137–151. https://doi.org/10.1016/j.fsi.2004.09.006
Martins ML, Mouriño JLP, Amaral GV, Vieira FN, Dotta G, Jatobá AMB, Pedrotti FS, Jerônimo GT, Buglione-Neto CC, Pereira G Jr (2008) Haematological changes in Nile tilapia experimentally infected with Enterococcus sp. Braz J Biol 68:657–661. https://doi.org/10.1590/S1519-69842008000300025
Neely MN, Pfeifer JD, Caparon M (2002) Streptococcus zebrafish model of bacterial pathogenesis. Infect Immun 70:3904–3914. https://doi.org/10.1128/IAI.70.7.3904-3914.2002
Pereira F, Ravelo C, Toranzo AE, Romalde JL (2004) Lactococcus garvieae, and emerging pathogen for the Portuguese trout culture. Bull Eur Assoc Fish Pathol 24:274–279
Ranzani-Paiva MJT, Ishikawa CM, Eiras AC, Silveira VR (2004) Effects of an experimental challenge with Mycobacteriu marinum on the blood parameters of Nile Tilapia, Oreochromis niloticus (Linnaeus, 1757). Braz Arch Biol Technol 46:945–953. https://doi.org/10.1590/S1516-89132004000600014
Ranzani-Paiva MJT, Pádua SB, Tavares-Dias M, Egami MI (2013) Métodos para análise hematológica em peixes. Eduem, Maringá
Řehulka J (2002) Aeromonas causes severe skin lesions in rainbow trout (Oncorhynchus mykiss): clinical pathology, Haemathology and biochemistry. Acta Vet BRNO 71:351–360. https://doi.org/10.2754/avb200271030351
Řehulka J, Minařic B (2007) Blood parameters in brook trout Salvelinus fontinalis (Mitchill, 1815), affected by columnaris disease. Aquac Res 38:1182–1197. https://doi.org/10.1111/j.1365-2109.2007.01786.x
Reimschuessel R (2001) A fish model of renal regeneration and development. ILAR J 42:285–291. https://doi.org/10.1093/ilar.42.4.285
Sebastião FA, Furlan LR, Hashimoto DT, Pilarski F (2015) Identification of bacterial fish pathogens in Brazil by direct colony PCR and 16S rRNA gene sequencing. Adv Microbiol 5:409–424. https://doi.org/10.4236/aim.2015.56042
Secombes CJ (1996) The nonspecific immune system: celular defenses. In: Iwama G, Nakanishi T (eds) The fish immune system. Academic Press, London, pp 63–105
Shahi N, Mallik SN, Sahoo M, Chandra S, Singh AK (2018) First report on characterization and pathogenicity study of emerging Lactococcus garvieae infection in farmed rainbow trout, Oncorhynchus mykiss (Walbaum), from India. Transboud Emerg Dis 65:1039–1048. https://doi.org/10.1111/tbed.12843
Silva BC, Mouriño JLP, Vieira FN, Jatobá A, Seiffert WQ, Martins ML (2012) Haemorrhagic septicemia in the hybrid surubim (Pseudoplatystoma corruscans X Pseudoplatystoma fasciatum) caused by Aeromonas hydrophila. Aquac Res 42:908–916. https://doi.org/10.1111/j.1365-2109.2011.02905.x
Steinel NC, Bolnick DI (2017) Melanomacrophage centers as a histological indicator of immune function in fish and other poikilotherms. Front Immunol. https://doi.org/10.3389/fimmu.2017.00827
Suzuki Y, Iida T (1992) Fish granulocytes in the process of inflammation. Annu Rev Fish Dis 2:149–160. https://doi.org/10.1016/0959-8030(92)90061-2
Tsai MA, Wang PC, Liaw LL, Yoshida T, Chen SC (2012) Comparison of genetic characteristics and pathogenicity of Lactococcus garvieae isolated from aquatic animals in Taiwan. Dis Aquat Org 102:43–51. https://doi.org/10.3354/dao02516
Türe M, Haliloğlu HI, Altuntas C, Boran H, Kutju I (2014) Comparison of experimental susceptibility of rainbow trout (Oncorhynchus mykiss), turbot (Psetta maxima), black sea trout (Salmo trutta labrax) and sea bass (Dicentrarchus labrax) to Lactococcus garvieae. Turk J Fish Aquat Sci 14:507–513. https://doi.org/10.4194/1303-2712-v14_2_22
Vela AI, Vázquez J, Gibello A, Blanco MM, Moreno MA, Liébana P, Albendea C, Alcalá B, Mendez A, Domínguez L, Fernández-Garayzábal JF (2000) Phenotypic and genetic characterization of Lactococcus garvieae isolated in Spain from Lactococcosis outbreaks and comparison with isolates of other countries and sources. J Clin Microbiol 38:3791–3795
Vendrell D, Balcázar JL, Ruiz-Zarzuela I, Blas I, Gironés O, Múzquiz JL (2006) Lactococcus garvieae in fish: a review. Comp Immunol Microbiol Infect Dis 29:177–198. https://doi.org/10.1016/j.cimid.2006.06.003
Wang CYC, Shie HS, Chen SC, Huang JP, Hsieh IC, Wen MS, Lin FC, Wu D (2007) Lactococcus garvieae infections in humans: possible association with aquaculture outbreaks. Int J Clin Pract 61:60–73. https://doi.org/10.1111/j.1742-1241.2006.00855.x
Watanabe Y, Naito T, Kikuchi K, Amari Y, Uehara Y, Isonuma H, Hisaoka T, Yoshida T, Yaginuma K, Takaya N, Daida H, Hiramatsu K (2011) Infective endocarditis with Lactococcus garvieae in Japan: a case report. J Med Case Rep 5:356–360. https://doi.org/10.1186/1752-1947-5-356
Yoshida T, Endo M, Sakai M, Inglis V (1997) A cell capsule with possible involvement in resistance to opsonophagocytosis in Enterococcus seriolicida isolated from yellowtail Seriola quinqueradiata. Dis Aquat Org 29:233–235
Yu JH, Han JJ, Park SW (2010) Haematological and biochemical alterations in Korean catfish, Silurus asotus, experimentally infected with Edwardsiella tarda. Aquac Res 41:295–302. https://doi.org/10.1111/j.1365-2109.2009.02331.x
Acknowledgments
The authors thank Ph.D. Fernanda de Alexandre Sebastião for the support in the molecular identification of strains.
Funding
This study was financially supported by the Fundação de Apoio ao Desenvolvimento do Ensino, Ciência e Tecnologia do Estado de Mato Grosso do Sul (FUNDECT) (Process Number 23/200.233/2014).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical principles
All experimental procedures were conducted according to the National Council of Animal experimentation/CONCEA and approved by the Animal Ethics Committee of the Federal University of Mato Grosso do Sul (protocol no. 601/2014).
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rodrigues, R.A., do Nascimento Silva, A.L., Siqueira, M.S. et al. Hematological, biochemical, and histopathological responses in sorubim Pseudoplatystoma spp. experimentally infected with Lactococcus garvieae. Aquacult Int 28, 1907–1923 (2020). https://doi.org/10.1007/s10499-020-00566-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-020-00566-5