Abstract
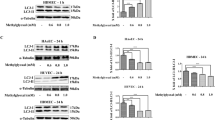
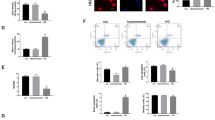
Cardiac microvascular endothelial cells (CMECs) dysfunction is an important pathophysiological event in the cardiovascular complications induced by diabetes. However, the underlying mechanism is not fully clarified. Autophagy is involved in programmed cell death. Here we investigated the potential role of autophagy on the CMECs injury induced by high glucose. CMECs were cultured in normal or high glucose medium for 6, 12 and 24 h respectively. The autophagy of CMECs was measured by green fluorescence protein (GFP)-LC3 plasmid transfection. Moreover, the apoptosis of CMEC was determined by flow cytometry. Furthermore, 3-Methyladenine (3MA), ATG7 siRNA and rapamycin were administrated to regulate the autophagy state. Moreover, Western blotting assay was performed to measure the expressions of Akt, mTOR, LC3 and p62. High glucose stress decreased the autophagy, whereas increased the apoptosis in CMECs time dependently. Meanwhile, high glucose stress activated the Akt/mTOR signal pathway. Furthermore, autophagy inhibitor, 3-MA and ATG7 siRNA impaired the autophagy and increased the apoptosis in CMECs induced by high glucose stress. Conversely, rapamycin up-regulated the autophagy and decreased the apoptosis in CMECs under high glucose condition. Our data provide evidence that high glucose directly inhibits autophagy, as a beneficial adaptive response to protect CMECs against apoptosis. Furthermore, the autophagy was mediated, at least in part, by mTOR signaling.







Similar content being viewed by others
References
Brownlee M (2001) Biochemistry and molecular cell biology of diabetic complications. Nature 414:813–820
Mazzone T (2010) Intensive glucose lowering and cardiovascular disease prevention in diabetes: reconciling the recent clinical trial data. Circulation 122:2201–2211
Angeja BG, de Lemos J, Murphy SA, Marble SJ, Antman EM, Cannon CP et al (2002) Impact of diabetes mellitus on epicardial and microvascular flow after fibrinolytic therapy. Am Heart J 144:649–656
Liu Y, Ma Y, Wang R, Xia C, Zhang R, Lian K et al (2011) Advanced glycation end products accelerate ischemia/reperfusion injury through receptor of advanced end product/nitrative thioredoxin inactivation in cardiac microvascular endothelial cells. Antioxid Redox Signal 15:1769–1778
Zhang Z, Li W, Sun D, Zhao L, Zhang R, Wang Y et al (2011) Toll-like receptor 4 signaling in dysfunction of cardiac microvascular endothelial cells under hypoxia/reoxygenation. Inflamm Res 60:37–45
Peng C, Ma J, Gao X, Tian P, Li W, Zhang L (2013) High glucose induced oxidative stress and apoptosis in cardiac microvascular endothelial cells are regulated by FoxO3a. PLoS ONE 8:e79739
Takagi H, Matsui Y, Sadoshima J (2007) The role of autophagy in mediating cell survival and death during ischemia and reperfusion in the heart. Antioxid Redox Signal 9:1373–1381
Terman A, Brunk UT (2005) Autophagy in cardiac myocyte homeostasis, aging, and pathology. Cardiovasc Res 68:355–365
Castino R, Isidoro C, Murphy D (2005) Autophagy-dependent cell survival and cell death in an autosomal dominant familial neurohypophyseal diabetes insipidus in vitro model. FASEB J 19:1024–1026
Mizushima N, Levine B (2010) Autophagy in mammalian development and differentiation. Nat Cell Biol 12:823–830
Kim KH, Lee MS. (2013). Autophagy as a crosstalk mediator of metabolic organs in regulation of energy metabolism. Rev Endocr Metab Disord
Liu Y, Chen Y, Wen L, Cui G (2012) Molecular mechanisms underlying the time-dependent autophagy and apoptosis induced by nutrient depletion in multiple myeloma: a pilot study. J Huazhong Univ Sci Technol Med Sci 32:1–8
Maiuri MC, Zalckvar E, Kimchi A, Kroemer G (2007) Self-eating and self-killing: crosstalk between autophagy and apoptosis. Nat Rev Mol Cell Biol 8:741–752
Shintani T, Klionsky DJ (2004) Autophagy in health and disease: a double-edged sword. Science 306:990–995
Wu YT, Tan HL, Huang Q, Ong CN, Shen HM (2009) Activation of the PI3K-Akt-mTOR signaling pathway promotes necrotic cell death via suppression of autophagy. Autophagy 5:824–834
Oh SY, Choi SJ, Kim KH, Cho EY, Kim JH, Roh CR (2008) Autophagy-related proteins, LC3 and Beclin-1, in placentas from pregnancies complicated by preeclampsia. Reprod Sci 15:912–920
Tanida I, Ueno T, Kominami E (2008) LC3 and Autophagy. Methods Mol Biol 445:77–88
Tanida I, Ueno T, Kominami E (2004) LC3 conjugation system in mammalian autophagy. Int J Biochem Cell Biol 36:2503–2518
Meijer WH, van der Klei IJ, Veenhuis M, Kiel JA (2007) ATG genes involved in non-selective autophagy are conserved from yeast to man, but the selective Cvt and pexophagy pathways also require organism-specific genes. Autophagy 3:106–116
Yordy B, Tal MC, Hayashi K, Arojo O, Iwasaki A (2013) Autophagy and selective deployment of Atg proteins in antiviral defense. Int Immunol 25:1–10
Jaakkola PM, Pursiheimo JP (2009) p62 degradation by autophagy: another way for cancer cells to survive under hypoxia. Autophagy 5:410–412
Rusten TE, Stenmark H (2010) p62, an autophagy hero or culprit? Nat Cell Biol 12:207–209
Xu X, Hueckstaedt LK, Ren J (2013) Deficiency of insulin-like growth factor 1 attenuates aging-induced changes in hepatic function: role of autophagy. J Hepatol 59:308–317
Kobayashi S, Xu X, Chen K, Liang Q (2012) Suppression of autophagy is protective in high glucose-induced cardiomyocyte injury. Autophagy 8:577–592
Zhang Z, Li S, Cui M, Gao X, Sun D, Qin X et al (2013) Rosuvastatin enhances the therapeutic efficacy of adipose-derived mesenchymal stem cells for myocardial infarction via PI3K/Akt and MEK/ERK pathways. Basic Res Cardiol 108:333
Feng Q, Song W, Lu X, Hamilton JA, Lei M, Peng T et al (2002) Development of heart failure and congenital septal defects in mice lacking endothelial nitric oxide synthase. Circulation 106:873–879
Levine B, Kroemer G (2008) Autophagy in the pathogenesis of disease. Cell 132:27–42
Casado P, Bilanges B, Rajeeve V, Vanhaesebroeck B, Cutillas PR (2014) Environmental stress affects the activity of metabolic and growth factor signaling networks and induces autophagy markers in MCF7 breast cancer cells. Mol Cell Proteomics 13(3):836–848
Levine B, Yuan J (2005) Autophagy in cell death: an innocent convict? J Clin Invest 115:2679–2688
Shi R, Weng J, Zhao L, XM Li, Gao TM, Kong J (2012) Excessive autophagy contributes to neuron death in cerebral ischemia. CNS Neurosci Ther 18:250–260
Wilson CM, Magnaudeix A, Yardin C, Terro F (2013) Autophagy dysfunction and its link to Alzheimer’s disease and type II diabetes mellitus. CNS Neurol Disord Drug Targets 13(2):226–246
Yamamoto S, Kazama JJ, Fukagawa M (2013) Autophagy: a two-edged sword in diabetes mellitus. Biochem J 456:e1–e3
Torisu T, Torisu K, Lee IH, Liu J, Malide D, Combs CA et al (2013) Autophagy regulates endothelial cell processing, maturation and secretion of von Willebrand factor. Nat Med 19:1281–1287
Mellor KM, Bell JR, Young MJ, Ritchie RH, Delbridge LM (2011) Myocardial autophagy activation and suppressed survival signaling is associated with insulin resistance in fructose-fed mice. J Mol Cell Cardiol 50:1035–1043.
Polager S, Ofir M, Ginsberg D (2008) E2F1 regulates autophagy and the transcription of autophagy genes. Oncogene 27:4860–4864
Wang B, Ling S, Lin WC (2010) 14-3-3t regulates beclin 1 and is required for autophagy. PLoS ONE 5(4):e10409
Wang X, Proud CG (2006) The mTOR pathway in the control of protein synthesis. Physiology 21:362–369
Pene F, Claessens YE, Muller O, Viguie F, Mayeux P, Dreyfus F et al (2002) Role of the phosphatidylinositol 3-kinase/Akt and mTOR/P70S6-kinase pathways in the proliferation and apoptosis in multiple myeloma. Oncogene 21:6587–6597
Sarkar S, Ravikumar B, Floto RA, Rubinsztein DC (2009) Rapamycin and mTOR-independent autophagy inducers ameliorate toxicity of polyglutamine-expanded huntingtin and related proteinopathies. Cell Death Differ 16:46–56
Xu N, Lao Y, Zhang Y, Gillespie DA (2012) Akt: a double-edged sword in cell proliferation and genome stability. J Oncol 2012:951724
Zhang Y, Xu X, Ren J (2013) MTOR overactivation and interrupted autophagy flux in obese hearts: a dicey assembly? Autophagy 9:939–941
He C, Zhu H, Li H, Zou MH, Xie Z (2013) Dissociation of Bcl-2-Beclin1 complex by activated AMPK enhances cardiac autophagy and protects against cardiomyocyte apoptosis in diabetes. Diabetes 62:1270–1281
Acknowledgements
This work was supported by National Nature Science Foundation of China (No. 81400274).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted.
Additional information
Zheng Zhang, Shenwei Zhang, Yong Wang, and Ming Yang have contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10495_2017_1398_MOESM1_ESM.tif
Supplementary material 1 HG induced apoptosis via caspase way. A: Representative TUNEL staining imaging of CMECs in each group. B: Quantification of the apoptotic CMECs (n=3, *p < 0.05). C: Representative Western blots of cleaved caspase-3 in each group. D: Quantitative analysis of the expressions of cleaved caspase-3/β actin. (TIF 1619 KB)
Rights and permissions
About this article
Cite this article
Zhang, Z., Zhang, S., Wang, Y. et al. Autophagy inhibits high glucose induced cardiac microvascular endothelial cells apoptosis by mTOR signal pathway. Apoptosis 22, 1510–1523 (2017). https://doi.org/10.1007/s10495-017-1398-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-017-1398-7