Abstract
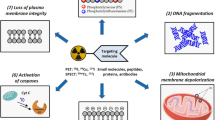
The exposition of phosphatidylserine (PS) from the cell membrane is associated with most cell death programs (apoptosis, necrosis, autophagy, mitotic catastrophe, etc.), which makes PS an attractive target for overall cell death imaging. To this end, zinc(II) macrocycle coordination complexes with cyclic polyamine units as low-molecular-weight annexin mimics have a selective affinity for biomembrane surfaces enriched with PS, and are therefore useful for detection of cell death. In the present study, a 11C-labeled zinc(II)–bis(cyclen) complex (11C-CyclenZn2) was prepared and evaluated as a new positron emission tomography (PET) probe for cell death imaging. 11C-CyclenZn2 was synthesized by methylation of its precursor, 4-methoxy-2,5-di-[10-methyl-1,4,7,10-tetraazacyclododecane-1,4,7-tricarboxylic acid tri-tert-butyl ester] phenol (Boc-Cyclen2) with 11C-methyl triflate as a prosthetic group in acetone, deprotection by hydrolysis in aqueous HCl solution, and chelation with zinc nitrate. The cell death imaging capability of 11C-CyclenZn2 was evaluated using in vitro cell uptake assays with camptothecin-treated PC-3 cells, biodistribution studies, and in vivo PET imaging in Kunming mice bearing S-180 fibrosarcoma. Starting from 11C-methyl triflate, the total preparation time for 11C-CyclenZn2 was ~40 min, with an uncorrected radiochemical yield of 12 ± 3% (based on 11C-CH3OTf, n = 10), a radiochemical purity of greater than 95%, and the specific activity of 0.75–1.01 GBq/μmol. The cell death binding specificity of 11C-CyclenZn2 was demonstrated by significantly different uptake rates in camptothecin-treated and control PC-3 cells in vitro. Inhibition experiments for 18F-radiofluorinated Annexin V binding to apoptotic/necrotic cells illustrated the necessity of zinc ions for zinc(II)–bis(cyclen) complexation in binding cell death, and zinc(II)–bis(cyclen) complexe and Annexin V had not identical binding pattern with apoptosis/necrosis cells. Biodistribution studies of 11C-CyclenZn2 revealed a fast clearance from blood, low uptake rates in brain and muscle tissue, and high uptake rates in liver and kidney, which provide the main metabolic route. PET imaging using 11C-CyclenZn2 revealed that cyclophosphamide-treated mice (CP-treated group) exhibited a significant increase of uptake rate in the tumor at 60 min postinjection, compared with control mice (Control group). The results indicate that the ability of 11C-CyclenZn2 to detect cell death is comparable to Annexin V, and it has potential as a PET tracer for noninvasive evaluation and monitoring of anti-tumor chemotherapy.






Similar content being viewed by others
References
Corsten MF, Hofstra L, Narula J et al (2006) Counting heads in the war against cancer: defining the role of annexin A5 imaging in cancer treatment and surveillance. Cancer Res 66:1255–1260
Tait JF, Gibson D, Fujikawa K (1989) Phospholipid binding properties of human placental anticoagulant protein-I, a member of the lipocortin family. J Biol Chem 264:7944–7949
Mochizuki T, Kuge Y, Zhao S et al (2003) Detection of apoptotic tumor response in vivo after a single dose of chemotherapy with 99mTc-annexin V. J Nucl Med 44:92–97
Narula J, Acio ER, Narula N et al (2001) Annexin-V imaging for noninvasive detection of cardiac allograft rejection. Nat Med 7:1347–1352
Laufer EM, Reutelingsperger CP, Narula J et al (2008) Annexin A5: an imaging biomarker of cardiovascular risk. Basic Res Cardiol 103:95–104
Hu S, Kiesewetter DO, Zhu L et al (2012) Longitudinal PET imaging of doxorubicin-induced cell death with 18F-Annexin V. Mol Imaging Biol 14:762–770
Wei X, Li Y, Zhang S et al (2014) Ultrasound targeted apoptosis imaging in monitoring early tumor response of trastuzumab in a murine tumor xenograft model of her-2-positive breast cancer. Transl Oncol 7:284–291
Han MS, Kim DH (2002) Naked-eye detection of phosphate ions in water at physiological pH: a remarkably selective and easy-to-assemble colorimetric phosphate-sensing probe. Angew Chem Int Ed Engl 41:3809–3811
Lee DH, Im JH, Son SU et al (2003) An azophenol-based chromogenic pyrophosphate sensor in water. J Am Chem Soc 125:7752–7753
Ojida A, Mito-Oka Y, Inoue MA et al (2002) First artificial receptors and chemosensors toward phosphorylated peptide in aqueous solution. J Am Chem Soc 124:6256–6258
Smith BA, Xiao S, Wolter W et al (2011) In vivo targeting of cell death using a synthetic fluorescent molecular probe. Apoptosis 16:722–731
Wang Z, Zhang L, Guo Z et al (2012) A unique feature of iron loss via close adhesion of Helicobacter pylori to host erythrocytes. PLoS ONE 7:e50314
Wyffels L, Gray BD, Barber C et al (2011) Synthesis and preliminary evaluation of radiolabeled bis(zinc(II)-dipicolylamine) coordination complexes as cell death imaging agents. Bioorg Med Chem 19:3425–3433
Liu L, Liu S, Chen X et al (2013) Switching molecular orientation of individual fullerene at room temperature. Sci Rep 3:3062
Kimura E, Aoki S, Kikuta E et al (2003) A macrocyclic zinc(II) fluorophore as a detector of apoptosis. PNAS 100:3731–3736
Oltmanns D, Zitzmann-Kolbe S, Mueller A et al (2011) Zn(II)-bis(cyclen) complexes and the imaging of apoptosis/necrosis. Bioconjug Chem 22:2611–2624
Zulkefeli M, Sogon T, Takeda K et al (2009) Design and synthesis of a stable supramolecular trigonal prism formed by the self-assembly of a linear tetrakis(Zn2+-cyclen) complex and trianionic trithiocyanuric acid in aqueous solution and its complexation with DNA (cyclen = 1,4,7,10-tetraazacyclododecane). Inorg Chem 48:9567–9578
Aghanejad A, Jalilian AR, Fazaeli Y et al (2014) Synthesis and evaluation of [(67)Ga]-AMD3100: a novel imaging agent for targeting the chemokine receptor CXCR4. Sci Pharm 82:29–42
Wang H, Tang X, Tang G et al (2013) Noninvasive positron emission tomography imaging of cell death using a novel small-molecule probe, (18)F labeled bis(zinc(II)-dipicolylamine) complex. Apoptosis 18:1017–1027
Hanshaw RG, Lakshmi C, Lambert TN et al (2005) Fluorescent detection of apoptotic cells by using zinc coordination complexes with a selective affinity for membrane surfaces enriched with phosphatidylserine. Chembiochem 6:2214–2220
Swairjo MA, Concha NO, Kaetzel MA et al (1995) Ca(2+)-bridging mechanism and phospholipid head group recognition in the membrane-binding protein annexin V. Nat Struct Biol 2:968–974
Hanshaw RG, O`Neil EJ, Foley M et al (2005) Indicator displacement assays that detect bilayer membranes enriched in phosphatidylserine. J Mater Chem 15:2707–2713
Hinz JM, Helleday T, Meuth M (2003) Reduced apoptotic response to camptothecin in CHO cells deficient in XRCC3. Carcinogenesis 24:249–253
Wang HL, Tang GH, Hu KZ et al (2014) PET imaging of sterile inflammation with a 18F-labeled bis(zinc(II)-dipicolylamine) complex. J Radioanal Nucl Chem 302:273–280
Li W, Nie S, Chen Y et al (2011) Enhancement of cyclophosphamide-induced antitumor effect by a novel polysaccharide from Ganoderma atrum in sarcoma 180-bearing mice. J Agric Food Chem 59:3707–3716
Acknowledgements
This work was supported by the National Natural Science Foundation (No. 81471695, 81101076, 30970856, 81571716), Postdoctoral Science Foundation of China (20110490964), The Doctoral Starting up Foundation of Shanxi Medical University (No. 03201316), Scientific and Technologial Innovation Programs of Higher Education Institutions in Shanxi (No. 2014134), Molecular imaging Precision Medicine collaborative innovation Center of Shanxi Medical University.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, H., Wu, Z., Li, S. et al. Synthesis and evaluation of a radiolabeled bis-zinc(II)–cyclen complex as a potential probe for in vivo imaging of cell death. Apoptosis 22, 585–595 (2017). https://doi.org/10.1007/s10495-017-1344-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-017-1344-8