Abstract
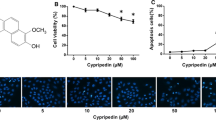
Phenazine-1-carboxamide (PCN), a naturally occurring simple phenazine derivative isolated from Pseudomonas sp. strain PUP6, exhibited selective cytotoxic activity against lung (A549) and breast (MDA-MB-231) cancer cell lines in differential and dose-dependent manner compared to normal peripheral blood mononuclear cells. PCN-treated cancer cells showed the induction of apoptosis as evidenced by the release of low level of LDH, morphological characteristics, production of reactive oxygen species, loss of mitochondrial membrane potential (ΔΨm) and induction of caspase-3. At molecular level, PCN instigates apoptosis by mitochondrial intrinsic apoptotic pathway via the overexpression of p53, Bax, cytochrome C release and activation of caspase-3 with the inhibition of oncogenic anti-apoptotic proteins such as PARP and Bcl-2 family proteins (Bcl-2, Bcl-w and Bcl-xL). The in silico docking studies of PCN targeted against the anti-apoptotic members of Bcl-2 family proteins revealed the interaction of PCN with the BH3 domain, which might lead to the induction of apoptosis due to the inhibition of antiapoptotic proteins. Due to its innate inhibition potential of antiapoptotic Bcl-2 family proteins, PCN may be used as potent anticancer agent against both lung and breast cancer.







Similar content being viewed by others
References
Giddens SR, Feng Y, Mahanty HK (2002) Characterization of a novel phenazine antibiotic gene cluster in Erwinia herbicola Eh1087. Mol Microbiol 45:769–783
Mavrodi DV, Blankenfeldt W, Thomashow LS (2006) Phenazine compounds in fluorescent Pseudomonas spp. biosynthesis and regulation. Annu Rev Phytopathol 44:417–445
Abken HJ, Tietze M, Brodersen J, Bäumer S, Beifuss U, Deppenmeier U (1998) Isolation and characterization of methanophenazine and function of phenazines in membrane-bound electron transport of Methanosarcina mazei Gö1. J Bacteriol 180:2027–2032
Kerr JR, Taylor GW, Rutman A, Hoiby N, Cole PJ, Wilson R (1999) Pseudomonas aeruginosa pyocyanin and 1-hydroxyphenazine inhibit fungal growth. J Clin Pathol 52:385–387
Hussain H, Specht S, Sarite SR, Saeftel M, Hoerauf A, Schulz B, Krohn K (2011) A new class of phenazines with activity against a chloroquine resistant Plasmodium falciparum strain and antimicrobial activity. J Med Chem 54:4913–4917
Neves-Pinto C, Malta VRS, Pinto MDCFR, Santos RHA, de Castro SL, Pinto AV (2002) A trypanocidal phenazine derived from b-lapachone. J Med Chem 45:2112–2115
Wang W, Préville P, Morin N, Mounir S, Cai W, Siddiqui MA (2000) Hepatitis C viral IRES inhibition by phenazine and phenazine-like molecules. Bioorg Med Chem Lett 10:1151–1154
Abdelfattah MS, Kazufumi T, Ishibashi M (2011) Isolation and structure elucidation of izuminosides AeC: a rare phenazine glycosides from Streptomyces sp. IFM 11260. J Antibiot 64:271–275
Rewcastle GW, Denny WA, Baguley BC (1987) Potential antitumor agents. 51. Synthesis and antitumor activity of substituted phenazine-1-carboxamides. J Med Chem 30:843–851
Cimmino A, Evidente A, Mathieu V, Andolfi A, Lefranc F, Kornienko A, Kiss R (2012) Phenazines and cancer. Nat Prod Rep 29:487–501
Cimmino A, Andolfi A, Evidente A (2013) Microbion phenazines. In: Chincholkar, Thomashow (eds) Phenazine as an anticancer agent. Springer, Heidelberg, pp 217–243
Gao X, Lu Y, Xing Y, Ma Y, Lu J, Bao W, Wang Y, Xi T (2012) A novel anticancer and antifungus phenazine derivative from a marine actinomycete BM-17. Microbiol Res 167:616–622
Gao X, Lu Y, Fang L, Fang X, Xing Y, Gou S, Xi T (2013) Synthesis and anticancer activity of some novel 2-phenazinamine derivatives. Eur J Med Chem 69:1–9
Pierson LS III, Pierson EA (2010) Metabolism and function of phenazines in bacteria: impacts on the behaviour of bacteria in the environment and biotechnological processes. Appl Microbiol Biotechnol 86:1659–1670
Sorensen RU, Klinger JD, Cash HA, Chase PA, Dearborn DG (1983) In vitro inhibition of lymphocyte proliferation by Pseudomonas aeruginosa phenazine pigments. Infect Immun 41:321–330
Cerecetto H, González M, Lavaggi ML, Azqueta A, de Cerain AL, Monge A (2005) Phenazine 5,10-dioxide derivatives as hypoxic selective cytotoxins. J Med Chem 48:21–23
Laursen JB, Nielsen J (2004) Phenazine natural products: biosynthesis, synthetic analogues and biological activity. Chem Rev 104:1663–1685
Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM (2010) Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer 127:2893–2917
Kornblau SM, Qiu YH, Bekele BN, Cade JS, Zhou X, Harris D, Andreeff M (2006) Studying the right cell in acute myelogenous leukemia: dynamic changes of apoptosis and signal transduction pathway protein expression in chemotherapy resistant ex vivo selected “survivor cells”. Cell Cycle 5:2769–2777
Naik PR, Sakthivel N (2006) Functional characterization of a novel hydrocarbonoclastic Pseudomonas sp. strain PUP6 with plant-growth-promoting traits and antifungal potential. Res Microbiol 157:538–546
Gurusiddaiah S, Weller DM, Sarkar A, Cook RJ (1986) Characterization of an antibiotic produced by a strain of Pseudomonas fluorescens inhibitory to Gaeumannomyces graminis var. tritici and Pythium spp. Antimicrob Agents Chemother 29:488–495
Chan FK, Moriwaki K, De Rosa MJ (2013) Detection of necrosis by release of lactate dehydrogenase (LDH) activity. Methods Mol Biol 979:6–70
Zilla MK, Nayak D, Amin H, Nalli Yedukondalu N, Kitchlu S, Goswami A, Ali A (2014) 4′-Demethyl-deoxypodophyllotoxin glucoside isolated from Podophyllum hexandrum exhibits potential anticancer activities by altering Chk-2 signaling pathway in MCF-7 breast cancer cells. Chem Biol Interact. doi:10.1016/j.cbi.2014.09.022
Subramaniya BR, Srinivasan G, Saduallah SSM, Davis N, Subhadara LBR, Halagowder D, Sivasitambaram ND (2011) Apoptosis inducing effect of plumbagin on colonic cancer cells depends on expression of COX-2. PLOs One 6:e18695
Shanmugaraj S, Selvaraj S, Murugan VP, Rajaganapathy BR, Shanmuganathan MV, Devaraj NS, Subhadra LB (2010) 3β-Hydroxylup-20(29)-ene-27,28-dioic acid dimethyl ester, a novel natural product from Plumbago zeylanica inhibits the proliferation and migration of MDA MB-231 cells. Chem Biol Interact 188:412–420
Chin-A-Woeng TFC, Bloemberg GV, Van der Bij AJ, Van der Drift KMGM, Schripse-ma J, Kroon B, Scheffer RJ, Keel C, Bakker PAHM, Tichy H, de Bruijn FJ, Thomas-Oates JE, Lugtenberg BJJ (1998) Biocontrol by phenazine-1-carboxamide producing Pseudomonas chlororaphis PCL1391 of tomato root rot caused by Fusarium oxysporum f. sp. radicis-lycopersici. Mol Plant Microbe Interact 11:1069–1077
Shanmugaiah V, Mathivanan N, Varghese B (2010) Purification, crystal structure and antimicrobial activity of phenazine-1-carboxamide produced by a growth-promoting biocontrol bacterium, Pseudomonas aeruginosa MML2212. J Appl Microbiol 108:703–711
Balachandran C, Sangeetha B, Duraipandiyan V, Raj MK, Ignacimuthu S, Al-Dhabi NA, Balakrishna K, Parthasarathy K, Arulmozhi NM, Arasu MV (2014) A flavonoid isolated from Streptomyces sp. (ERINLG-4) induces apoptosis in human lung cancer A549 cells through p53 and cytochrome c release caspase dependant pathway. Chem Biol Interact. doi:10.1016/j.cbi.2014.09.019
Ramsey MR, Sharpless NE (2006) ROS as a tumour suppressor? Nat Cell Biol 8:1213–1215
Takahashi A, Ohtani N, Yamakoshi K, Shin-ichi I, Tahara H, Nakayama K, Nakayama KI, Ide T, Saya H, Hara E (2006) Mitogenic signalling and the p16INK4a–Rb pathway cooperate to enforce irreversible cellular senescence. Nat Cell Biol 8:1291–1297
Nogueira V, Hay N (2013) Molecular pathways: reactive oxygen species homeostasis in cancer cells and implications for cancer therapy. Clin Cancer Res 19:4309–4314
Skulachev VP (2006) Bioenergetic aspects of apoptosis, necrosis and mitoptosis. Apoptosis 11:473–485
Tsujimoto Y, Shimizu S (2007) Role of the mitochondrial membrane permeability transition in cell death. Apoptosis 12:835–840
Circu ML, Aw TA (2010) Reactive oxygen species, cellular redox systems, and apoptosis. Free Radic Biol Med 48:749–762
Chen Q, Lesnefsky EJ (2006) Depletion of cardiolipin and cytochrome c during ischemia increases hydrogen peroxide production from the electron transport chain. Free Radic Biol Med 40:976–982
Renschler MF (2004) The emerging role of reactive oxygen species in cancer therapy. Eur J Cancer 40:1934–1940
Toler SM, Noe D, Sharma A (2006) Selective enhancement of cellular oxidative stress by chloroquine: implications for the treatment of glioblastoma multiforme. Neurosurg Focus 21:1–4
Wang C, Youle RJ (2009) The role of mitochondria in apoptosis. Annu Rev Genet 43:95–118
Brenner D, Mak TW (2009) Mitochondrial cell death effectors. Curr Opin Cell Biol 21:871–877
Wong RSY (2011) Apoptosis in cancer: from pathogenesis to treatment. J Exp Clin Cancer Res 30:1–14
Thornberry NA, Lazebnik Y (1998) Caspases: enemies within. Science 281:1312–1316
Clarke PG (1990) Developmental cell death: morphology diversity and multiple mechanisms. Anat Embryol (Berl) 181:195–213
Acknowledgments
The financial support from the Ministry of Science and Technology and Department of Biotechnology-Boost to University Interdisciplinary Life science Departments for Education and Research (DBT-BUILDER) programme, New Delhi, Government of India is gratefully acknowledged.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kennedy, R.K., Veena, V., Naik, P.R. et al. Phenazine-1-carboxamide (PCN) from Pseudomonas sp. strain PUP6 selectively induced apoptosis in lung (A549) and breast (MDA MB-231) cancer cells by inhibition of antiapoptotic Bcl-2 family proteins. Apoptosis 20, 858–868 (2015). https://doi.org/10.1007/s10495-015-1118-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-015-1118-0