Abstract
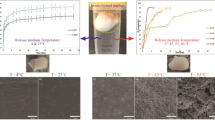
There is a growing number of protein drugs, yet their limited oral bioavailability requires that patients receive frequent, high dose injections. In situ forming implants (ISFIs) for controlled release of biotherapeutics have the potential to greatly reduce the injection frequency and improve patient compliance. However, protein release from ISFIs is a challenge due to their proclivity for instability. Specifically, factors such as the acidic microclimate within ISFIs can lead to protein aggregation and denaturation. Basic salts have been shown in PLGA microparticle and microcylinder formulations to significantly reduce protein instability by neutralizing this acidic environment. The overall objective of the study was to demonstrate that basic salts can be used with an ISFI system to neutralize the implant acidification. To this end, the basic salts MgCO3 and Mg(OH)2 were added to a protein-releasing ISFI and the effect on drug release, pH, implant swelling, implant diffusivity, and implant erosion were evaluated. Either salt added at 3 wt% neutralized the acidic environment surrounding the implants, keeping the pH at 6.64 ± 0.03 (MgCO3) and 6.46 ± 0.11 (Mg(OH)2) after 28 day compared to 3.72 ± 0.05 with no salts added. The salts initially increased solution uptake into the implants but delayed implant degradation and erosion. The 3 wt% Mg(OH)2 formulation also showed slightly improved drug release with a lower burst and increased slope. We showed that salt additives can be an effective way to modulate the pH in the ISFI environment, which can improve protein stability and ultimately improve the capacity of ISFIs for delivering pH-sensitive biomolecules. Such a platform represents a low-cost method of improving overall patient compliance and reducing the overall healthcare burden.





Similar content being viewed by others
References
Agarwal, P., and I. D. Rupenthal. Injectable implants for the sustained release of protein and peptide drugs. Drug Discov. Today. 18:337–349, 2013
Brodbeck, K., J. Desnoyer, and A. McHugh. Phase inversion dynamics of PLGA solutions related to drug delivery: part II. The role of solution thermodynamics and bath-side mass transfer. J. Control. Release. 62:333–344, 1999
Brown, L. R. Commercial challenges of protein drug delivery. Expert Opin. Drug Deliv. 2:29–42, 2005
Brunner, A., K. Mäder, and A. Göpferich. pH and osmotic pressure inside biodegradable microspheres during erosion. Pharm. Res. 16:847–853, 1999
Bruno, B. J., G. D. Miller, and C. S. Lim. Basics and recent advances in peptide and protein drug delivery. Ther. Deliv. 4:1443–1467, 2013
Cui, C., V. C. Stevens, and S. P. Schwendeman. Injectable polymer microspheres enhance immunogenicity of a contraceptive peptide vaccine. Vaccine. 25:500–509, 2007
Ding, A., and S. P. Schwendeman. Acidic microclimate ph distribution in PLGA microspheres monitored by confocal laser scanning microscopy. Pharm. Res. 25:2041–2052, 2008
Dunn, R., J. English, D. Cowsar and D. Vanderbilt. Biodegradable in-situ forming implants and methods of producing the same. U.S. Patent US4938763A, 1988 .
Eliaz, R. E., and J. Kost. Characterization of a polymeric PLGA-injectable implant delivery system for the controlled release of proteins. J. Biomed. Mater. Res. 50:388–396, 2000
Estey, T., J. Kang, S. P. Schwendeman, and J. F. Carpenter. BSA degradation under acidic conditions: a model for protein instability during release from PLGA delivery systems. J. Pharm. Sci. 95:1626–1639, 2006
Fu, K., A. M. Klibanov, and R. Langer. Protein stability in controlled-release systems. Nat. Biotechnol. 18:24–25, 2000
Fu, K., D. W. Pack, A. M. Klibanov, and R. Langer. Visual evidence of acidic environment within degrading poly(lactic-co-glycolic acid) (PLGA) microspheres. Pharm. Res. 17:100–106, 2000
Graham, P., K. Brodbeck, and A. McHugh. Phase inversion dynamics of PLGA solutions related to drug delivery. J. Control. Release. 58:233–245, 1999
Hopkins, K. A., N. Vike, X. Li, J. Kennedy, E. Simmons, J. Rispoli, and L. Solorio. Noninvasive characterization of in situ forming implant diffusivity using diffusion-weighted MRI. J. Control. Release. 309:289–301, 2019
Houchin, M. L., and E. M. Topp. Chemical degradation of peptides and proteins in PLGA: a review of reactions and mechanisms. J. Pharm. Sci. 97:2395–2404, 2008
Ibeanu, N., R. Egbu, L. Onyekuru, H. Javaheri, P. T. Khaw, G. R. Williams, S. Brocchini, and S. Awwad. Injectables and depots to prolong drug action of proteins and peptides. Pharmaceutics. 12:999, 2020
Jiang, W., and S. P. Schwendeman. Stabilization of tetanus toxoid encapsulated in plga microspheres. Mol. Pharm. 5:808–817, 2008
Kang, J., and S. P. Schwendeman. Comparison of the effects of Mg(OH)2 and sucrose on the stability of bovine serum albumin encapsulated in injectable poly(d, l-lactide-co-glycolide) implants. Biomaterials. 23:239–245, 2002
Kempe, S., and K. Mader. In situ forming implants - an attractive formulation principle for parenteral depot formulations. J. Control. Release. 161:668–679, 2012
Lambert, W. J., and K. D. Peck. Development of an in situ forming biodegradable poly-lactide-co-glycolide system for the controlled release of proteins. J. Control. Release. 33:189–195, 1995
Lewis, A. L., and L. Illum. Formulation strategies for sustained release of proteins. Ther. Deliv. 1:457–479, 2010
Lewis, A. L., and J. Richard. Challenges in the delivery of peptide drugs: an industry perspective. Ther. Deliv. 6:49–163, 2015
Liu, Y., and S. P. Schwendeman. Mapping microclimate ph distribution inside protein-encapsulated plga microspheres using confocal laser scanning microscopy. Mol. Pharm. 9:1342–1350, 2012
Packhaeuser, C., J. Schnieders, C. Oster, and T. Kissel. In situ forming parenteral drug delivery systems: an overview. Eur. J. Pharm. Biopharm. 58:445–455, 2004
Parent, M., C. Nouvel, M. Koerber, A. Sapin, P. Maincent, and A. Boudier. PLGA in situ implants formed by phase inversion: critical physiochemical parameters to modulate drug release. J. Control. Release. 172:292–304, 2013
Patel, R. B., L. Solorio, H. Wu, T. Krupka, and A. A. Exner. Effect of injection site on in situ forming implant formation and drug release in vivo. J. Control. Release. 147(3):350–358, 2010
Pauletti, G. M., S. Gangwar, T. J. Siahaan, J. Aubé, and R. T. Borchardt. Improvement of oral peptide bioavailability: peptidomimetics and prodrug strategies. Adv. Drug. Deliv. Rev. 27:235–256, 1997
Putney, S. D., and P. A. Burke. Improving protein therapeutics with sustained-release formulations. Nat. Biotechnol. 16:153–157, 1998
Renukuntla, J., A. D. Vadlapudi, A. Patel, S. H. Boddu, and A. K. Mitra. Approaches for enhancing oral bioavailability of peptides and proteins. Int. J. Pharm. 447:75–93, 2013
Schädlich, A., S. Kempe, and K. Mäder. Noninvasive in vivo characterization of microclimate pH inside in situ forming PLGA implants using multispectral fluorescence imaging. J. Control. Release. 179:52–62, 2014
Schwendeman, S. P. Recent advances in the stabilization of proteins encapsulated in injectable plga delivery systems. Crit. Rev. Ther. Drug Carrier Syst. 19:73–98, 2002
Schwendeman, S. P., R. B. Shah, B. A. Bailey, and A. S. Schwendeman. Injectable controlled release depots for large molecules. J. Control. Release. 190:240–253, 2014
Shenderova, A., A. G. Ding, and S. P. Schwendeman. Potentiometric method for determination of microclimate pH in poly(lactic-co-glycolic acid) films. Macromolecules. 37:10052–10058, 2004
Shire, S. J., Z. Shahrokh, and J. Liu. Challenges in the development of high protein concentration formulations. J. Pharm. Sci. 93:1390–1402, 2004
Solorio, L., and A. A. Exner. Effect of the subcutaneous environment on phase-sensitive in situ-forming implant drug release, degradation, and microstructure. J. Pharm. Sci. 104(12):4322–4328, 2015
Stejskal, E. O., and J. E. Tanner. Spin diffusion measurements: spin echoes in the presence of a time-dependent field gradient. J. Chem. Phys. 42:288–292, 1965
Stevenson, C. L., C. A. Rhodes and S. J. Prestrelski. Delivery of peptides and proteins via long acting injections and implants. In: Long Acting Injections and Implants. Boston: Springer, 2012 .
Vaishya, R., V. Khurana, S. Patel, and A. K. Mitra. Long-term delivery of protein therapeutics. Expert Opin. Drug Deliv. 12:415–440, 2015
van de Weert, M., W. E. Hennink, and W. Jiskoot. Protein instability in poly(lactic-co-glycolic acid) microparticles. Pharm. Res. 17:1159–1167, 2000
Ye, M., S. Kim, and K. Park. Issues in long-term protein delivery using biodegradable microparticles. J. Control. Release. 146:241–260, 2010
Zhu, G., S. R. Mallery, and S. P. Schwendeman. Stabilization of proteins encapsulated in injectable poly(lactide-co-glycolide). Nat. Biotechnol. 18:52–57, 2000
Zhu, G., and S. P. Schwendeman. Stabilization of proteins encapsulated in cylindrical poly(lactide-co-glycolide) implants: mechanism of stabilization by basic additives. Pharm. Res. 17:351–357, 2000
Acknowledgments
We would like to thank the Purdue Electron Microscopy Facility and the Purdue MRI Facility for their assistance with SEM and DWI, respectively. We would also like to acknowledge the Summer Undergraduate Research Fellowship (SURF) at Purdue University for E.W.
Funding
None.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Stefan M. Duma oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hopkins, K., Wakelin, E., Romick, N. et al. Basic Salt Additives Modulate the Acidic Microenvironment Around In Situ Forming Implants. Ann Biomed Eng 51, 966–976 (2023). https://doi.org/10.1007/s10439-022-03109-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-022-03109-6