Abstract
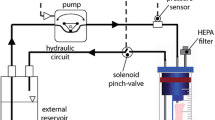
Arteries often endure axial twist due to body movement and surgical procedures, but how arteries remodel under axial twist remains unclear. The objective of this study was to investigate early stage arterial wall remodeling under axial twist. Porcine carotid arteries were twisted axially and maintained for three days in ex vivo organ culture systems while the pressure and flow remained the same as untwisted controls. Cell proliferation, internal elastic lamina (IEL) fenestrae shape and size, endothelial cell (EC) morphology and orientation, as well as the expression of matrix metalloproteinases (MMPs), MMP-2 and MMP-9, and tissue inhibitor of metalloproteinase-2 (TIMP-2) were quantified using immunohistochemistry staining and immunoblotting. Our results demonstrated that cell proliferation in both the intima and media were significantly higher in the twisted arteries compared to the controls. The cell proliferation in the intima increased from 1.33 ± 0.21% to 7.63 ± 1.89%, and in the media from 1.93 ± 0.84% to 8.27 ± 2.92% (p < 0.05). IEL fenestrae total area decreased from 26.07 ± 2.13% to 14.74 ± 0.61% and average size decreased from 169.03 ± 18.85 μm2 to 80.14 ± 1.96 μm2 (p < 0.01), but aspect ratio increased in the twist group from 2.39 ± 0.15 to 2.83 ± 0.29 (p < 0.05). MMP-2 expression significantly increased (p < 0.05) while MMP-9 and TIMP-2 showed no significant difference in the twist group. The ECs in the twisted arteries were significantly elongated compared to the controls after three days. The angle between the major axis of the ECs and blood flow direction under twist was 7.46 ± 2.44 degrees after 3 days organ culture, a decrease from the initial 15.58 ± 1.29 degrees. These results demonstrate that axial twist can stimulate artery remodeling. These findings complement our understanding of arterial wall remodeling under mechanical stress resulting from pressure and flow variations.






Similar content being viewed by others
References
Anwar, M. A., J. Shalhoub, C. S. Lim, M. S. Gohel, and A. H. Davies. The effect of pressure-induced mechanical stretch on vascular wall differential gene expression. J. Vasc. Res. 49(6):463–478, 2012.
Bilgin, S. S., M. Topalan, W. Y. Ip, and S. P. Chow. Effect of torsion on microvenous anastomotic patency in a rat model and early thrombolytic phenomenon. Microsurgery 23(4):381–386, 2003.
Cheng, C. P., N. M. Wilson, R. L. Hallett, R. J. Herfkens, and C. A. Taylor. In vivo MR angiographic quantification of axial and twisting deformations of the superficial femoral artery resulting from maximum hip and knee flexion. J. Vasc. Interv. Radiol. 17(6):979–987, 2006.
Chesler, N. C., D. N. Ku, and Z. S. Galis. Transmural pressure induces matrix-degrading activity in porcine arteries ex vivo. Am. J. Physiol. Heart Circ. Physiol. 277(5):H2002–H2009, 1999.
Chien, S., S. Li, and J. Y. J. Shyy. Effects of mechanical forces on signal transduction and gene expression in endothelial cells. Hypertension 31(1):162–169, 1998.
Chiu, J. J., and S. Chien. Effects of disturbed flow on vascular endothelium: pathophysiological basis and clinical perspectives. Physiol. Rev. 91(1):327–387, 2011.
Deng, S. X., J. Tomioka, J. C. Debes, and Y. C. Fung. New experiments on shear modulus of elasticity of arteries. Am. J. Physiol. 266(1 Pt 2):H1–H10, 1994.
Dobrin, P. B., D. Hodgett, T. Canfield, and R. Mrkvicka. Mechanical determinants of graft kinking. Ann. Vasc. Surg. 15(3):343–349, 2001.
Endean, E. D., S. DeJong, and P. B. Dobrin. Effect of twist on flow and patency of vein grafts. J. Vasc. Surg. 9(5):651–655, 1989.
Flaherty, J. T., J. E. Pierce, V. J. Ferrans, D. J. Patel, W. K. Tucker, and D. L. Fry. Endothelial nuclear patterns in the canine arterial tree with particular reference to hemodynamic events. Circ. Res. 30(1):23–33, 1972.
Fuster, J. J., P. Fernandez, H. Gonzalez-Navarro, C. Silvestre, Y. N. Nabah, and V. Andres. Control of cell proliferation in atherosclerosis: insights from animal models and human studies. Cardiovasc. Res. 86(2):254–264, 2010.
Galis, Z. S., and J. J. Khatri. Matrix metalloproteinases in vascular remodeling and atherogenesis: the good, the bad, and the ugly. Circ. Res. 90(3):251–262, 2002.
Garcia, J. R., S. D. Lamm, and H. C. Han. Twist buckling behavior of arteries. Biomech. Model. Mechanobiol. 12(5):915–927, 2013.
Gleason, R. L., E. Wilson, and J. D. Humphrey. Biaxial biomechanical adaptations of mouse carotid arteries cultured at altered axial extension. J. Biomech. 40(4):766–776, 2007.
Godin, D., E. Ivan, C. Johnson, R. Magid, and Z. S. Galis. Remodeling of carotid artery is associated with increased expression of matrix metalloproteinases in mouse blood flow cessation model. Circulation 102(23):2861–2866, 2000.
Guo, Z. Y., Z. Q. Yan, L. Bai, M. L. Zhang, and Z. L. Jiang. Flow shear stress affects macromolecular accumulation through modulation of internal elastic lamina fenestrae. Clin. Biomech. 23:S104–S111, 2008.
Han, H.-C., S. Marita, and D. N. Ku. Changes of opening angle in hypertensive and hypotensive arteries in 3-day organ culture. J. Biomech. 39(13):2410–2418, 2006.
Han, H. C., and D. N. Ku. Contractile responses in arteries subjected to hypertensive pressure in seven-day organ culture. Ann. Biomed. Eng. 29(6):467–475, 2001.
Han, H. C., D. N. Ku, and R. P. Vito. Arterial wall adaptation under elevated longitudinal stretch in organ culture. Ann. Biomed. Eng. 31(4):403–411, 2003.
Han, H. C. Twisted blood vessels: symptoms, etiology and biomechanical mechanisms. J. Vasc. Res. 49(3):185–197, 2012.
Han, H. C., J. K. Chesnutt, J. R. Garcia, Q. Liu, and Q. Wen. Artery buckling: new phenotypes, models, and applications. Ann. Biomed. Eng. 41(7):1399–1410, 2013.
Hayman, D. M., Y. Xiao, Q. Yao, Z. Jiang, M. L. Lindsey, and H.-C. Han. Alterations in pulse pressure affect artery function. Cell. Mol. Bioeng. 5(4):474–487, 2012.
Hirata, A., P. Baluk, T. Fujiwara, and D. M. Mcdonald. Location of focal silver staining at endothelial gaps in inflamed venules examined by scanning electron-microscopy. Am. J. Physiol. Lung. Cell Mol. Physiol. 269(3):L403–L418, 1995.
Izquierdo, R., P. B. Dobrin, K. Fu, F. Park, and G. Galante. The effect of twist on microvascular anastomotic patency and angiographic luminal dimensions. J. Surg. Res. 78(1):60–63, 1998.
Jackson, Z. S., A. I. Gotlieb, and B. L. Langille. Wall tissue remodeling regulates longitudinal tension in arteries. Circ. Res. 90(8):918–925, 2002.
Kandalam, V., R. Basu, L. Moore, D. Fan, X. Wang, D. M. Jaworski, G. Y. Oudit, and Z. Kassiri. Lack of tissue inhibitor of metalloproteinases 2 leads to exacerbated left ventricular dysfunction and adverse extracellular matrix remodeling in response to biomechanical stress. Circulation 124(19):2094–2105, 2011.
Kassiri, Z., and R. Khokha. Myocardial extra-cellular matrix and its regulation by metalloproteinases and their inhibitors. Thromb. Haemost. 93(2):212–219, 2005.
Klein, A. J., S. J. Chen, J. C. Messenger, A. R. Hansgen, M. E. Plomondon, J. D. Carroll, and I. P. Casserly. Quantitative assessment of the conformational change in the femoropopliteal artery with leg movement. Catheter. Cardiovasc. Interv. 74(5):787–798, 2009.
Langille, B. L., J. J. Graham, D. Kim, and A. I. Gotlieb. Dynamics of shear-induced redistribution of F-actin in endothelial cells in vivo. Arterioscler. Thromb. 11(6):1814–1820, 1991.
Langille, B. L. Remodeling of developing and mature arteries: endothelium, smooth muscle, and matrix. J. Cardiovasc. Pharmacol. 21(Suppl 1):S11–S17, 1993.
Lee, A. Y., B. Y. Han, S. D. Lamm, C. A. Fierro, and H. C. Han. Effects of elastin degradation and surrounding matrix support on artery stability. Am. J. Physiol. Heart Circ. Physiol. 302(4):H873–H884, 2012.
Lee, Y. U., D. Drury-Stewart, R. P. Vito, and H. C. Han. Morphologic adaptation of arterial endothelial cells to longitudinal stretch in organ culture. J. Biomech. 41(15):3274–3277, 2008.
Lu, X., J. Yang, J. B. Zhao, H. Gregersen, and G. S. Kassab. Shear modulus of porcine coronary artery: contributions of media and adventitia. Am. J. Physiol. Heart Circ. Physiol. 285(5):H1966–H1975, 2003.
Macchiarelli, Arterial repair after microvascular anastomosis. Acta Anat (Basel) 1991. 140.
Nerem, R. M. Tissue engineering a blood vessel substitute: the role of biomechanics. Yonsei Med. J. 41(6):735–739, 2000.
Nichol, J. W., M. Petko, R. J. Myung, J. W. Gaynor, and K. J. Gooch. Hemodynamic conditions alter axial and circumferential remodeling of arteries engineered ex vivo. Ann. Biomed. Eng. 33(6):721–732, 2005.
Ramaiah, V. G., C. S. Thompson, S. Shafique, J. A. Rodriguez, R. Ravi, L. DiMugno, and E. B. Diethrich. Crossing the limbs: a useful adjunct for successful deployment of the AneuRx stent-graft. J. Endovasc. Ther. 9(5):583–586, 2002.
Rizzoni, D., M. L. Muiesan, E. Porteri, C. De Ciuceis, G. E. Boari, M. Salvetti, A. Paini, and E. A. Rosei. Vascular remodeling, macro- and microvessels: therapeutic implications. Blood Press. 18(5):242–246, 2009.
Salgarello, M., P. Lahoud, G. Selvaggi, S. Gentileschi, M. Sturla, and E. Farallo. The effect of twisting on microanastomotic patency of arteries and veins in a rat model. Ann. Plast. Surg. 47(6):643–646, 2001.
Sarkar, S., H. J. Salacinski, G. Hamilton, and A. M. Seifalian. The mechanical properties of infrainguinal vascular bypass grafts: their role in influencing patency. Eur. J. Vasc. Endovasc. Surg. 31(6):627–636, 2006.
Selvaggi, G., M. Salgarello, E. Farallo, S. Anicic, and L. Formaggia. Effect of torsion on microvenous anastomotic patency in rat model and early thrombolytic phenomenon. Microsurgery 24(5):416–417, 2004.
Strongin, A. Y., I. Collier, G. Bannikov, B. L. Marmer, G. A. Grant, and G. I. Goldberg. Mechanism of cell surface activation of 72-kDa type IV collagenase. Isolation of the activated form of the membrane metalloprotease. J. Biol. Chem. 270(10):5331–5338, 1995.
Tada, S., and J. M. Tarbell. Internal elastic lamina affects the distribution of macromolecules in the arterial wall: a computational study. Am. J. Physiol. Heart Circ. Physiol. 287(2):H905–H913, 2004.
Van Epps, J. S., and D. A. Vorp. A new three-dimensional exponential material model of the coronary arterial wall to include shear stress due to torsion. J. Biomech. Eng. Trans. Asme 130(5):051001–051008, 2008.
Voorhees, A. P., and H. C. Han. A model to determine the effect of collagen fiber alignment on heart function post myocardial infarction. Theor. Biol. Med. Model 11:6, 2014.
Vos, A. W. F., M. A. M. Linsen, J. T. Marcus, J. C. van den Berg, J. A. Vos, J. A. Rauwerda, and W. Wisselink. Carotid artery dynamics during head movements: A reason for concern with regard to carotid stenting. J. Endovasc. Ther. 10(5):862–869, 2003.
Wang, J. H. C., P. Goldschmidt-Clermont, J. Wille, and F. C. P. Yin. Specificity of endothelial cell reorientation in response to cyclic mechanical stretching. J. Biomech. 34(12):1563–1572, 2001.
Wong, C. H., F. Cui, B. K. Tan, Z. Liu, H. P. Lee, C. Lu, C. L. Foo, and C. Song. Nonlinear finite element simulations to elucidate the determinants of perforator patency in propeller flaps. Ann. Plast. Surg. 59(6):672–678, 2007.
Xiao, Y., D. Hayman, S. S. Khalafvand, M. L. Lindsey, and H. C. Han. Artery buckling stimulates cell proliferation and NF-kappaB signaling. Am. J. Physiol. Heart Circ. Physiol. 307:H542–H551, 2014.
Yao, Q., D. M. Hayman, Q. Dai, M. L. Lindsey, and H. C. Han. Alterations of pulse pressure stimulate arterial wall matrix remodeling. J. Biomech. Eng. 131(10):101011, 2009.
Zhao, S., A. Suciu, T. Ziegler, J. E. Moore, Jr., E. Burki, J. J. Meister, and H. R. Brunner. Synergistic effects of fluid shear stress and cyclic circumferential stretch on vascular endothelial cell morphology and cytoskeleton. Arterioscler. Thromb. Vasc. Biol. 15(10):1781–1786, 1995.
Acknowledgments
This work was supported by National Natural Science Foundation of China through Grant 11229202 and by the National Institutes of Health through Grant R01HL095852. It was also partially supported through HHSN 268201000036C (N01-HV-00244) for the San Antonio Cardiovascular Proteomics Center. The authors thank Granzins Meat Market at New Braunfels, Texas for their help in this work. We also thank Dr. Coleen Witt from the Computational Biology Initiatives at UTSA for her help in this study and thank the RCMI facility center supported by Grant G12MD007591 from the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Jane Grande-Allen oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Wang, GL., Xiao, Y., Voorhees, A. et al. Artery Remodeling Under Axial Twist in Three Days Organ Culture. Ann Biomed Eng 43, 1738–1747 (2015). https://doi.org/10.1007/s10439-014-1215-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-014-1215-1