Abstract
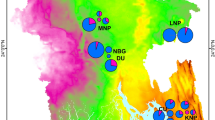
The emerging infectious disease chytridiomycosis is prevalent in Central and South America, and has caused catastrophic declines of amphibian populations in the Neotropics. The responsible organism, Batrachochytrium dendrobatidis, has been recorded on three West Indian islands, but the whole of the Caribbean region is predicted to offer a suitable environment for the disease. Monitoring the spread of chytridiomycosis is thus a priority in this region, which has exceptionally high levels of amphibian endemism. PCR analysis of 124 amphibian skin swabs in Tobago (Republic of Trinidad and Tobago) demonstrated the presence of B. dendrobatidis in three widely separated populations of the frog Mannophryne olmonae, which is listed as Critically Endangered on the basis of recent population declines. Chytridiomycosis is presently endemic in this species, with a prevalence of about 20% and no associated clinical disease. Increased susceptibility to chytridiomycosis from climate change is unlikely in amphibian populations in Tobago, as this island does not have high montane environments, but remains a possibility in the sister island of Trinidad. Preventing the spread of chytridiomycosis within and between these and other Caribbean islands should be a major goal of practical conservation measures for amphibians in the region.

Similar content being viewed by others
References
Alemu JB I, Cazabon MNE, Dempewolf L, Hailey A, Lehtinen RM, Mannette RP, et al. (2007) Ecological observations on the critically endangered Tobago endemic frog Mannophryne olmonae. Applied Herpetology 4:377–386
Barrionuevo S, Mangione S (2006) Chytridiomycosis in two species of Telmatobius (Anura: Leptodactylidae) from Argentina. Diseases of Aquatic Organisms 73:171–174
Berger L, Speare R, Daszak P, Green DE, Cunningham AA, Goggin CL, et al. (1998) Chytridiomycosis causes amphibian mortality associated with population declines in the rain forests of Australia and Central America. Proceedings of the National Academy of Sciences of the United States of America 95:9031–9036
Berger L, Speare R, Hines HB, Marantelli G, Hyatt AD, McDonald KR, et al. (2004) Effect of season and temperature on mortality in amphibians due to chytridiomycosis. Australian Veterinary Journal 82:434–439
Berger L, Speare R, Skerratt LF (2005) Distribution of Batrachochytrium dendrobatidis and pathology in the skin of green tree frogs Litoria caerulea with severe chytridiomycosis. Diseases of Aquatic Organisms 68:65–70
Blaustein AR, Romansic JM, Scheessele EA, Han BA, Pessier AP, Longcore JE (2005) Interspecific variation in susceptibility of frog tadpoles to the pathogenic fungus Batrachochytrium dendrobatidis. Conservation Biology 19:1460–1468
Boyle DG, Boyle DB, Olsen V, Morgan JAT, Hyatt AD (2004) Rapid quantitative detection of chytridiomycosis (Batrachochytrium dendrobatidis) in amphibian samples using real-time Taqman PCR assay. Diseases of Aquatic Organisms 60:141–148
Burrowes PA, Joglar RL, Green DE (2004) Potential causes for amphibian declines in Puerto Rico. Herpetologica 60:141–154
Carey C, Bruzgul JE, Livo LJ, Walling ML, Kuehl KA, Dixon BF, et al. (2006) Experimental exposures of boreal toads (Bufo boreas) to a pathogenic chytrid fungus (Batrachochytrium dendrobatidis). EcoHealth 3:5–21
Carnaval ACOQ, Puschenforf R, Peixoto OL, Verdade VK, Rodrigues MT (2006) Amphibian chytrid fungus broadly distributed in the Brazilian Atlantic rain forest. EcoHealth 3:41–48
Daszak P, Strieby A, Cunningham AA, Longcore JE, Brown CC, Porter D (2004) Experimental evidence that the bullfrog (Rana catesbeiana) is a potential carrier of chytridiomycosis, an emerging fungal disease of amphibians. Herpetological Journal 14:201–207
Díaz LM, Cadiz A, Chong A, Silva A (2007) First report of chytridiomycosis in a dying toad (Anura: Bufonidae) from Cuba: a new conservation challenge for the island. EcoHealth 4:172–175
Drew A, Allen EJ, Allen LJS (2006) Analysis of climatic and geographic factors affecting the presence of chytridiomycosis in Australia. Diseases of Aquatic Organisms 68:245–250
Garcia TS, Romansic JM, Blaustein AR (2006) Survival of three species of anuran metamorphs exposed to UV-B and the pathogenic fungus Batrachochytrium dendrobatidis. Diseases of Aquatic Organisms 72:163–169
Grant T, Frost DR, Caldwell JP, Gagliardo R, Haddad CFB, Kok PJR, et al. (2006) Phylogenetic systematics of dart-poison frogs and their relatives (Amphibia: Athesphatanura: Dendrobatidae). Bulletin of the American Museum of Natural History 299:1–262
Hanselmann R, Rodriguez A, Lampo M, Fajardo-Ramos L, Aguirre AA, Kilpatrick AM, et al. (2004) Presence of an emerging pathogen of amphibians in introduced bullfrogs Rana catesbeiana in Venezuela. Biological Conservation 120:115–119
Hardy J (2004) Mannophryne olmonae. In: 2006 IUCN Red List of Threatened Species. Available: http://www.iucnredlist.org
Hardy JD Jr (1983) A new frog of the genus Colostethus from the island of Tobago, West Indies (Anura: Dendrobatidae). Bulletin of the Maryland Herpetological Society 19:47–57
Hedges SB (2006) An overview of the evolution and conservation of West Indian amphibians and reptiles. Applied Herpetology 3:281–292
Herrera RA, Steciow MM, Natale GS (2005) Chytrid fungus parasitizing the wild amphibian Leptodactylus ocellatus (Anura, Leptodactylidae) in Argentina. Diseases of Aquatic Organisms 64:247–252
Johnson ML, Speare R (2005) Possible modes of dissemination of the amphibian chytrid Batrachochytrium dendrobatidis in the environment. Diseases of Aquatic Organisms 65:181–186
Kriger KM, Hero J-M (2006) Survivorship in wild frogs infected with chytridiomycosis. EcoHealth 3:171–177
Kriger KM, Hero J-M (2007) Large-scale seasonal variation in the prevalence and severity of chytridiomycosis. Journal of Zoology 271:352–359
Kriger KM, Hines HB, Hyatt AD, Boyle DG, Hero J-M (2006) Techniques for detecting chytridiomycosis in wild frogs: comparing histology with real-time Taqman PCR. Diseases of Aquatic Organisms 71:141–148
La Marca E, Lips K, Lotters S, Puschendorf R, Ibáñez R, Rueda-Almonacid J, et al. (2005) Catastrophic population declines and extinction in neotropical harlequin frog (Bufonidae: Atelopus). Biotropica 37:190–201
Lampo M, Barrio-Amoros C, Han B (2006a) Batrachochytrium dendrobatidis infection in the recently rediscovered Atelopus mucubajiensis (Anura, Bufonidae), a critically endangered frog from the Venezuelan Andes. EcoHealth 3:299–302
Lampo M, Rodríguez-Contreras A, La Marca E, Daszak P (2006b) A chytridiomycosis epidemic and a severe dry season precede the disappearance of Atelopus species from the Venezuelan Andes. Herpetological Journal 16:395–402
Lips KR (1999) Mass mortality and population declines of anurans at an upland site in western Panama. Conservation Biology 13:117–125
Lips KR, Brem F, Brenes R, Reeve JD, Alford RA, Voyles J, et al. (2006) Emerging infectious disease and the loss of biodiversity in a neotropical amphibian community. Proceedings of the National Academy of Sciences of the United States of America 103:3165–3170
Lips KR, Green DE, Papendick R (2003a) Chytridiomycosis in wild frogs from southern Costa Rica. Journal of Herpetology 37:215–218
Lips KR, Mendelson JR III, Munoz-Alonso A, Canseco-Marquez L, Mulcahy DG (2004) Amphibian population declines in montane southern Mexico: resurveys of historical localities. Biological Conservation 119:555–564
Lips KR, Reeve JD, Witters LR (2003b) Ecological traits predicting amphibian declines in Central America. Conservation Biology 17:1078–1088
Longcore JE, Pessier AP, Nichols DK (1999) Batrachochytrium dendrobatidis gen. et sp. nov., a chytrid pathogenic to amphibians. Mycologia 91:219–227
Malhotra A, Thorpe RS, Hypolite E, James A (2007) A report on the status of the herpetofauna of the Commonwealth of Dominica, West Indies. Applied Herpetology 4:177–194
Mazzoni R, Cunningham AA, Daszak P, Apolo A, Perdomo E, Speranza G (2003) Emerging pathogen of wild amphibians in frogs (Rana catesbeiana) farmed for international trade. Emerging Infectious Diseases 9:995–998
McDonald KR, Mendez D, Muller R, Freeman AB, Speare R (2005) Decline in the prevalence of chytridiomycosis in frog populations in North Queensland, Australia. Pacific Conservation Biology 11:114–120
Mendelson JR, Brodie ED Jr, Malone JH, Acevedo ME, Baker MA, Smatresk MJ, et al. (2005) Factors associated with the catastrophic decline of a cloudforest frog fauna in Guatemala. Revista de Biologia Tropical 52:991–1000
Murphy JC (1997) Amphibians and Reptiles of Trinidad and Tobago, Malabar, FL: Krieger Publishing
Nichols DK, Lamirande EW, Pessier AP, Longcore JE (2001) Experimental transmission of cutaneous chytridiomycosis in dendrobatid frogs. Journal of Wildlife Diseases 37:1–11
Ouellet M, Mikaelian I, Pauli BD, Rodrigues J, Green DM (2005) Historical evidence of widespread chytrid infection in North American amphibian populations. Conservation Biology 19:1431–1440
Piotrowski JS, Annis SL, Longcore JE (2004) Physiology of Batrachochytrium dendrobatidis, a chytrid pathogen of amphibians. Mycologia 96:9–15
Pough FH (1980) The advantages of ectothermy for tetrapods. American Naturalist 115:92–112
Pounds JA, Bustamante MR, Coloma LA, Consuegra JA, Fogden MPL, Foster PN, et al. (2006) Widespread amphibian extinctions from epidemic disease driven by global warming. Nature 439:161–167
Puschendorf R, Bolanos F, Chaves G (2006a) The amphibian chytrid fungus along an altitudinal transect before the first reported declines in Costa Rica. Biological Conservation 132:136–142
Puschendorf R, Castañeda F, McCranie JR (2006b) Chytridiomycosis in wild frogs from Pico Bonito National Park, Honduras. EcoHealth 3:178–181
Retallick RWR, McCallum H, Speare R (2004) Endemic infection of the amphibian chytrid fungus in a frog community post-decline. PLoS Biology 2:1965–1971
Ron S (2005) Predicting the distribution of the amphibian pathogen Batrachochytrium dendrobatidis in the New World. Biotropica 37:209–221
Schloegel LM, Hero J-M, Berger L, Speare R, McDonald K, Daszak P (2006) The decline of the sharp-snouted day frog (Taudactylus acutirostris): The first documented case of extinction by infection in a free-ranging wildlife species? EcoHealth 3:35–40
Smith KG, Weldon C, Conradie W, Du Preez LH (2007) Relationships among size, development, and Batrachochytrium dendrobatidis infection in African tadpoles. Diseases of Aquatic Organisms 74:159–164
Weldon C, Preez LH, Hyatt AD, Muller R, Speare R (2004) Origin of the amphibian chytrid fungus. Emerging Infectious Diseases 10:2100–2105
Wilson BS, Horrocks JA, Hailey A (2006) Conservation of insular herpetofaunas in the West Indies. Applied Herpetology 3:181–195
Woodhams DC, Alford RA (2005) Ecology of chytridiomycosis in rainforest stream frog assemblages of tropical Queensland. Conservation Biology 19:1449–1459
Woodhams DC, Alford RA, Marantelli G (2003) Emerging disease of amphibians cured by elevated body temperature. Diseases of Aquatic Organisms 55:65–67
Acknowledgments
Fieldwork was supported by a 2006 Future Conservationist Award from the bp Conservation Programme (Mannophryne olmonae: An Ecological Study in Tobago—Project L.E.A.P.), and a seed grant from the Declining Amphibian Populations Task Force (health monitoring in amphibian populations in Trinidad and Tobago). Analysis of DNA samples was funded by a seed grant from Conservation International. We thank Dave Hardy for information on locations of M. olmonae, John E. Cooper, Jodi Rowley, and Robin Moore for discussion of chytridiomycosis, and Gian Lalsingh and Pat Turpin of Environment Tobago for accommodation and logistical help. Handling frogs was under permit from the Wildlife Section of the Forestry Division of Trinidad and Tobago, and the Department of Natural Resources and the Environment of the Tobago House of Assembly, courtesy of Nadra Nathai-Gyan and Angela Ramsey, respectively.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Alemu I, J.B., Cazabon, M.N.E., Dempewolf, L. et al. Presence of the Chytrid Fungus Batrachochytrium dendrobatidis in Populations of the Critically Endangered Frog Mannophryne olmonae in Tobago, West Indies. EcoHealth 5, 34–39 (2008). https://doi.org/10.1007/s10393-008-0154-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10393-008-0154-4