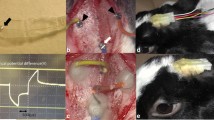
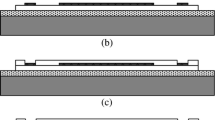
Abstract
Purpose
To determine the influence of stimulating electrode conditions on the amplitudes and latencies of electrically evoked potentials (EEPs) and the resistance at the electrode–tissue interface in the suprachoroidal transretinal stimulation (STS) system.
Study design
Experimental study.
Methods
A scleral pocket (3 × 5 mm) was created just over the visual streak in anesthetized pigmented rabbits (weight, 1.9-2.7 kg), and STS stimulating electrodes were implanted into the pocket. Measurements were obtained with stimulating electrodes of different lengths (0.3 or 0.5 mm) and different surface characteristics (smooth or porous). EEPs elicited with a fixed current under each set of electrode conditions were recorded; three measurement sessions were performed for each rabbit. The resistance at each electrode–tissue interface was measured.
Results
The latencies and amplitudes of the EEPs did not differ significantly with changes in the height and surface characteristics of the stimulating electrodes, but the resistances at the electrode–tissue interface differed significantly (P = 0.001; the resistance values for the 0.3-mm-long electrode with a porous surface was 5.24 ± 0.67 kΩ and with the 0.3- and 0.5-mm-long electrodes with smooth surfaces were 7.63 ± 0.12 kΩ and 6.77 ± 0.20 kΩ).
Conclusion
Being shorter did not affect the EEPs of the stimulating electrodes with a porous surface while decreasing the resistance at the electrode–tissue interface.


Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this article. Further enquiries can be directed to the corresponding author.
References
World Health Organization. Blindness and vision impairment. 2021. https://www.who.int/news-room/fact-sheets/detail/blindness-and-visual-impairment. Accessed 3 Jan 2022.
Morizane Y, Morimoto N, Fujiwara A, Kawasaki R, Yamashita H, Ogura Y, et al. Incidence and causes of visual impairment in Japan: the first nation-wide complete enumeration survey of newly certified visually impaired individuals. Jpn J Ophthalmol. 2019;63:26–33.
Russell S, Bennett J, Wellman JA, Chung DC, Yu ZF, Tillman A, et al. Efficacy and safety of voretigene neparvovec (AAV2-hRPE65v2) in patients with RPE65-mediated inherited retinal dystrophy: a randomised, controlled, open-label, phase 3 trial. Lancet. 2017;390:849–60.
Sahel JA, Boulanger-Scemama E, Pagot C, Arleo A, Galluppi F, Martel JN, et al. Partial recovery of visual function in a blind patient after optogenetic therapy. Nat Med. 2021;27:1223–9.
Shirai H, Mandai M, Matsushita K, Kuwahara A, Yonemura S, Nakano T, et al. Transplantation of human embryonic stem cell-derived retinal tissue in two primate models of retinal degeneration. Proc Natl Acad Sci USA. 2016;113:E81–90.
da Cruz L, Dorn JD, Humayun MS, Dagnelie G, Handa J, Barale PO, et al. Five-year safety and performance results from the Argus II retinal prosthesis system clinical trial. Ophthalmology. 2016;123:2248–54.
Chen J, Poulaki V, Kim SJ, Eldred WD, Kane S, Gingerich M, et al. Implantation and extraction of penetrating electrode arrays in minipig retinas. Transl Vis Sci Technol. 2020;9:19.
Edwards TL, Cottriall CL, Xue K, Simunovic MP, Ramsden JD, Zrenner E, et al. Assessment of the electronic retinal implant alpha AMS in restoring vision to blind patients with end-stage retinitis pigmentosa. Ophthalmology. 2018;125:432–43.
Sinclair NC, Shivdasani MN, Perera T, Gillespie LN, McDermott HJ, Ayton LN, et al. The appearance of phosphenes elicited using a suprachoroidal retinal prosthesis. Invest Ophthalmol Vis Sci. 2016;57:4948–61.
Sakaguchi H, Fujikado T, Fang X, Kanda H, Osanai M, Nakauchi K, et al. Transretinal electrical stimulation with a suprachoroidal multichannel electrode in rabbit eyes. Jpn J Ophthalmol. 2004;48:256–61.
Nishida K, Kamei M, Kondo M, Sakaguchi H, Suzuki M, Fujikado T, et al. Efficacy of suprachoroidal-transretinal stimulation in a rabbit model of retinal degeneration. Invest Ophthalmol Vis Sci. 2010;51:2263–8.
Fujikado T, Kamei M, Sakaguchi H, Kanda H, Endo T, Hirota M, et al. One-year outcome of 49-channel suprachoroidal-transretinal stimulation prosthesis in patients with advanced retinitis pigmentosa. Invest Ophthalmol Vis Sci. 2016;57:6147–57.
Morimoto T, Fujikado T, Kanda H, Miyoshi T, Endo T, Nishida K, et al. Testing of newly developed wide-field dual-array suprachoroidal-transretinal stimulation prosthesis in dogs. Transl Vis Sci Technol. 2021;10:13.
Santos A, Humayun MS, de Juan E Jr, Greenburg RJ, Marsh MJ, Klock IB, et al. Preservation of the inner retina in retinitis pigmentosa. A morphometric analysis. Arch Ophthalmol. 1997;115:511–5.
Sakaguchi H, Fujikado T, Kanda H, Osanai M, Fang X, Nakauchi K, et al. Electrical stimulation with a needle-type electrode inserted into the optic nerve in rabbit eyes. Jpn J Ophthalmol. 2004;48:552–7.
Fang X, Sakaguchi H, Fujikado T, Osanai M, Ikuno Y, Kamei M, et al. Electrophysiological and histological studies of chronically implanted intrapapillary microelectrodes in rabbit eyes. Graefes Arch Clin Exp Ophthalmol. 2006;244:364–75.
Sakaguchi H, Kamei M, Fujikado T, Yonezawa E, Ozawa M, Cecilia-Gonzalez C, et al. Artificial vision by direct optic nerve electrode (AV-DONE) implantation in a blind patient with retinitis pigmentosa. J Artif Organs. 2009;12:206–9.
Nishida K, Sakaguchi H, Kamei M, Cecilia-Gonzalez C, Terasawa Y, Velez-Montoya R, et al. Visual sensation by electrical stimulation using a new direct optic nerve electrode device. Brain Stimul. 2015;8:678–81.
Nishida K, Sakaguchi H, Kamei M, Saito T, Fujikado T, Nishida K. Electrophysiological evaluation of a chronically implanted electrode for suprachoroidal transretinal stimulation in rabbit eyes. J Artif Organs. 2019;22:237–45.
Terasawa Y, Tashiro H, Nakano Y, Ohta J. Safety and efficacy of semichronic suprachoroidal transretinal stimulation with femtosecond laser-induced porosity and smooth-surface electrodes. Sens Mater. 2018;30(2):235–49.
Hagihira S. Changes in the electroencephalogram during anaesthesia and their physiological basis. Br J Anaesth. 2015;115(Suppl 1):i27–31.
Shin HW, Kim HJ, Jang YK, You HS, Huh H, Choi YJ, et al. Monitoring of anesthetic depth and EEG band power using phase lag entropy during propofol anesthesia. BMC Anesthesiol. 2020;20:49.
Acknowledgments
This work was supported by JSPS KAKENHI Grant Number 21K12809 and JRPS Grant 2020. Special thanks to Prof. Ryo Kawasaki for advice for statistical analysis. Medical writing and editing support were provided by Editage.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interst
Kentaro Nishida, None; T. Morimoto, None; Y. Terasawa, Employee (NIDEK), NIDEK and the author own the patent for Stimulus electrode for biological tissue and method of producing the stimulus electrode; H. Sakaguchi, None; M. Kamei, None; T. Miyoshi, None; T. Fujikado, None; Kohji Nishida, None.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Corresponding Author: Kentaro Nishida
About this article
Cite this article
Nishida, K., Morimoto, T., Terasawa, Y. et al. The influence of stimulating electrode conditions on electrically evoked potentials and resistance in suprachoroidal transretinal stimulation. Jpn J Ophthalmol 67, 182–188 (2023). https://doi.org/10.1007/s10384-022-00972-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10384-022-00972-7