Abstract
Purpose
To clarify the redox states of the aqueous humor and serum in senile cataract patients.
Methods
We analyzed the reduced and oxidized albumin fractions in 58 patients. The patients had no detectable evidence of other eye or systemic diseases and were divided into two age groups (53–75 and 76-96 years). Analysis was performed with a high-performance liquid chromatographic system with a fluorescence detector.
Results
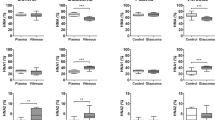
Extremely small amounts of reduced albumin (3.8% and 3.6% in the younger and older groups, respectively) and markedly large amounts of oxidized albumin (96.2% and 96.4%, respectively) were found in the aqueous humor. Moreover, a higher percentage of oxidized albumin, particularly a highly oxidized irreversible form of albumin, was found in the aqueous humor (12.3% and 12.0%) than in the serum (2.4% and 3.0%).
Conclusions
In senile cataract patients with no complications, the redox state of albumin in the aqueous humor differs considerably, both quantitatively and qualitatively, from that in the serum.
Similar content being viewed by others
References
Peters, T, Jr. All about albumin. Biochemistry, genetics, and medical applications. New York: Academic Press; 1996. p. 9–75.
King TP. On the sulfhydryl group of human plasma albumin. J Biol Chem 1961;236:PC5.
Kawakami A, Kubota K, Yamada N, et al. Identifi cation and characterization of oxidized human serum albumin. A slight structural change impairs its ligand-binding and antioxidant functions. FEBS J 2006;273:3346–3357.
Sogami M, Era S, Nagaoka S, et al. High-performance liquid chromatographic studies on non-mercapt ←→ mercapt conversion of human serum albumin. II. J Chromatogr 1985;332:19–27.
Fukushima H, Miwa Y, Shiraki M, et al. Oral branched-chain amino acid supplementation improves the oxidized/reduced albumin ratio in patients with liver cirrhosis. Hepatol Res 2007;37:765–770.
Soejima A, Kaneda F, Manno S, et al. Useful markers for detecting decreased serum antioxidant activity in hemodialysis patients. Am J Kidney Dis 2002;39:1040–1046.
Soejima A, Matsuzawa N, Hayashi T, et al. Alteration of redox state of human serum albumin before and after hemodialysis. Blood Purif 2004;22:525–529.
Terawaki H, Yoshimura K, Hasegawa T, et al. Oxidative stress is enhanced in correlation with renal dysfunction: examination with the redox state of albumin. Kidney Int 2004;66:1988–1993.
Terawaki H, Matsuyama Y, Era S, et al. Elevated oxidative stress measured as albumin redox state in continuous ambulatory peritoneal dialysis patients correlates with small uraemic solutes. Nephrol Dial Transplant 2007;22:968.
Terawaki H, Nakayama K, Matsuyama Y, et al. Dialyzable uremic solutes contribute to enhanced oxidation of serum albumin in regular hemodialysis patients. Blood Purif 2007;25:274–279.
Suzuki E, Yasuda K, Takeda N, et al. Increased oxidized form of serum albumin in patients with diabetes mellitus. Diabetes Res Clin Pract 1992;18:153–158.
Kawai K, Yoh M, Hayashi T, et al. Effect of diabetic retinopathy on redox state of aqueous humor and serum albumin in patients with senile cataract. Tokai J Exp Clin Med 2001;26:93–99.
Hayakawa A, Kuwata K, Era S, et al. Alteration of redox state of human serum albumin in patients under anesthesia and invasive surgery. J Chromatogr B 1997;698:27–33.
Hayashi T, Era S, Kawai K, et al. Observation for redox state of human serum and aqueous humor albumin from patients with senile cataract. Pathophysiology 2000;6:237–243.
Tomida M, Ishimaru J-I, Hayashi T, Nakamura K, Murayama K, Era S. The redox states of serum and synovial fl uid of patients with temporomandibular joint disorders. Jpn J Physiol 2003;53: 351–355.
Era S, Hamaguchi T, Sogami M, et al. Further studies on the resolution of human mercapt- and non-mercaptalbumin and on human serum albumin in the elderly by high-performance liquid chromatography. Int J Peptide Protein Res 1988;31:435–442.
Era S, Kuwata K, Imai H, Nakamura K, Hayashi T, Sogami M. Arerelated change in redox state of human serum albumin. Biochim Biophys Acta 1995;1247:12–16.
Imai H, Hayashi T, Negawa T, et al. Strenuous exercise-induced change in redox state of human serum albumin during intensive kendo training. Jpn J Physiol 2000;52:135–140.
Zirm M. Proteins in aqueous humor. Adv Ophthalmol 1980; 40:100–172.
Dernouchamps JP. The proteins of the aqueous humour. Doc Ophthalmol 1982;53:193–248.
Inada K, Murata T, Baba H, Murata Y, Ozaki M. Increase of aqueous humor proteins with aging. Jpn J Ophthalmol 1988;32:126–131.
Saari KM, Guillén-Monterrubio OM, Hartikainen J, Hämäläinen MM, Taskinen K. Measurement of protein concentration of aqueous humor in vivo: correlation between laser fl are measurements and chemical protein determination. Acta Ophthalmol Scand 1997;75:63–66.
Halliwell B. Oxidants and human disease: some new concepts. FASEB J 1987;1:358–364.
Spector A, Garner WH. Hydrogen peroxide and human cataract. Exp Eye Res 1981;33:673–681.
Varma SD, Devamanoharan PS, Morris SM. Prevention of cataracts by nutritional and metabolic antioxidants. Crit Rev Food Sci Nutri 1995;35:111–129.
Spector A. Oxidative stress-induced cataract: mechanism of action. FASEB J 1995;9:1173–1182.
Berlett BS, Stadtman ER. Protein oxidation in aging, disease, and oxidative stress. J Biol Chem 1997;272:20313–20316.
Finch JW, Crouch RK, Knapp DR, Schey KL. Mass spectrometric identification of modifications to human serum albumin treated with hydrogen peroxide. Arch Biochem Biophys 1994;312: 180–188.
Carballal S, Radi R, Kirk MC, Barnes S, Freeman BA, Alvarez B. Sulfenic acid formation in human serum albumin by hydrogen peroxide and peroxynitrite. Biochemistry 2003;42:9906–9914.
Vîrgolici B, Stoian I, Muscurel C, Mãrãcine M, Moraru C, Dinu V. Plasma redox status and premature onset of senile cataract. Rom J Intern Med 2007;45:59–65.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Kawai, K., Hayashi, T., Matsuyama, Y. et al. Difference in redox status of serum and aqueous humor in senile cataract patients as monitored via the albumin thiol-redox state. Jpn J Ophthalmol 54, 584–588 (2010). https://doi.org/10.1007/s10384-010-0877-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10384-010-0877-y