Abstract
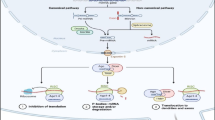
MicroRNAs (miRNAs) are small endogenous non-coding RNAs which can specifically silence gene expression, and thereby alter cell and organism phenotype. Deregulation of miRNA expression has been discovered in a variety of tumors and it is now clear that they contribute to cancer development and progression. Previous studies have indicated that miRNAs are involved in developmental timing, cell proliferation, apoptosis, morphogenesis [1], antiviral defense [2], and tumorigenesis [3]. In cancer pathways, altered expression of tumor suppressive or oncogenic miRNAs can disrupt regulatory mechanisms normal. Altered miRNAs expression patterns have been observed in a variety of diseased tissues. Cervical cancer is the most common malignant tumor in female reproductive tract. Recently more and more study showed a large number of miRNAs were down-regulated or up-regulated in cervical cancer. Recent data revealed that miRNA-218 (miR-218) played important roles in tumor initiation and development. This review focuses on analysis of miR-218 and will provide some insight into the progress of cervical cancer.
Similar content being viewed by others
References
Kim VN. Small RNAs: classification, biogenesis, and function. Mol Cells, 2005, 19: 1–15.
Lecellier CH, Dunoyer P, Arar K, et al. A cellular microRNA mediates antiviral defense in human cells. Science, 2005, 308: 557–560.
O’Donnell KA, Wentzel EA, Zeller KI, et al. c-Myc-regulated microRNAs modulate E2F1 expression. Nature, 2005, 435: 839–843.
Novina CD, Sharp PA. The RNAi revolution. Nature, 2004, 430: 161–164.
Sullivan CS, Grundhoff AT, Tevethia S, et al. SV40-encoded microRNAs regulate viral gene expression and reduce susceptibility to cytotoxic T cells. Nature, 2005, 435: 682–686.
Gupta A, Gartner JJ, Sethupathy P, et al. Anti-apoptotic function of a microRNA encoded by the HSV-1 latency-associated transcript. Nature, 2006, 442: 82–85.
Burnside J, Bernberg E, Anderson A, et al. Marek’s disease virus encodes MicroRNAs that map to meq and the latency-associated transcript. J Virology, 2006, 80: 8778–8786.
Calin GA, Sevignani C, Dumitru CD, et al. Human microRNA genes are frequently located at fragile sites and genomic regions involved in cancers. Proc Natl Acad Sci U S A, 2004, 101: 2999–3004.
Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell, 2004, 116: 281–297.
Zeng Y, Yi R, Cullen BR. MicroRNAs and small interfering RNAs can inhibit mRNA expression by similar mechanisms. Proc Natl Acad Sci U S A, 2003, 100: 9779–9784.
John B, Enright AJ, Aravin A, et al. Human MicroRNA targets. PLoS Biol, 2004, 2: e363.
Lewis BP, Burge CB, Bartel DP. Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microRNA targets. Cell, 2005, 120: 15–20.
Tran N, O’Brien CJ, Clark J, et al. Potential role of micro-RNAs in head and neck tumorigenesis. Head Neck, 2010, 32: 1099–1111.
Esquela-Kerscher A, Slack FJ. Oncomirs-microRNAs with a role in cancer. Nat Rev Cancer, 2006, 6: 259–269.
Li Y, Wang F, Xu J, et al. Progressive miRNA expression profiles in cervical carcinogenesis and identification of HPV-related target genes for miR-29. J Pathol, 2011, 224: 484–495.
Wang X, Tang S, Le SY, et al. Aberrant expression of oncogenic and tumor-suppressive microRNAs in cervical cancer is required for cancer cell growth. PLoS One, 2008, 3: e2557.
Wang X, Wang HK, McCoy JP, et al. Oncogenic HPV infection interrupts the expression of tumor-suppressive miR-34a through viral oncoprotein E6. RNA, 2009, 15: 637–647.
Li B, Hu Y, Ye F, et al. Reduced miR-34a expression in normal cervical tissues and cervical lesions with high-risk human papillomavirus infection. Int J Gynecol Cancer, 2010, 20: 597–604.
Petrocca F, Visone R, Onelli MR, et al. E2F1-regulated microRNAs impair TGFbeta-dependent cell-cycle arrest and apoptosis in gastric cancer. Cancer Cell, 2008, 13: 272–286.
Volinia S, Calin GA, Liu CG, et al. A microRNA expression signature of human solid tumors defines cancer gene targets. Proc Natl Acad Sci U S A, 2006, 103: 2257–2261.
Wu DW, Cheng YW, Wang J, et al. Paxillin predicts survival and relapse in non-small cell lung cancer by microRNA-218 targeting. Cancer Res, 2010, 70: 10392–10401.
Schembri F, Sridhar S, Perdomo C, et al. MicroRNAs as modulators of smoking-induced gene expression changes in human airway epithelium. Proc Natl Acad Sci U S A, 2009, 106: 2319–2324.
Yanaihara N, Caplen N, Bowman E, et al. Unique microRNA molecular profiles in lung cancer diagnosis and prognosis. Cancer Cell, 2006, 9: 189–198.
Alajez NM, Lenarduzzi M, Ito E, et al. MiR-218 suppresses nasopharyngeal cancer progression through downregulation of survivin and the SLIT2-ROBO1 pathway. Cancer Res, 2011, 71: 2381–2391.
Griffiths-Jones S, Grocock RJ, van Dongen S, et al. miRBase: microRNA sequences, targets and gene nomenclature. Nucleic Acids Res, 2006, 34: D140–D144.
Calin GA, Croce CM. MicroRNA-cancer connection: the beginning of a new tale. Cancer Res, 2006, 66: 7390–7394.
Zhang L, Huang J, Yang N, et al. microRNAs exhibit high frequency genomic alterations in human cancer. Proc Natl Acad Sci U S A, 2006, 103: 9136–9141.
Tie J, Pan Y, Zhao L, Shrover KR, et al. MiR-218 inhibits invasion and metastasis of gastric cancer by targeting the Robo1 receptor. PLoS Genet, 2010, 6: e1000879.
Thomison J 3rd, Thomas LK, et al. Human papillomavirus: molecular and cytologic/histologic aspects related to cervical intraepithelial neoplasia and carcinoma. Hum Pathol, 2008, 39: 154–166.
Stoler MH. Human papillomaviruses and cervical neoplasia: a model for carcinogenesis. Int J Gynecol Pathol, 2000, 19: 16–28.
Bernard HU, Burk RD, Chen Z, et al. Classification of papillomaviruses (PVs) based on 189 PV types and proposal of taxonomic amendments. Virology, 2010, 401: 70–79.
Clifford G, Franceschi S, Diaz M, et al. Chapter 3: HPV type-distribution in women with and without cervical neoplastic diseases. Vaccine, 2006, 3: S3/26–34.
Parkin DM, Bray F. Chapter 2: The burden of HPV-related cancers. Vaccine, 2006, 3: S3/11–25.
Wald AI, Hoskins EE, Wells SI, et al. Alteration of microRNA profiles in squamous cell carcinoma of the head and neck cell lines by human papillomavirus. Head Neck, 2011, 33: 504–512.
Nominé Y, Masson M, Charbonnier S, et al. Structural and functional analysis of E6 oncoprotein: insights in the molecular pathways of human papillomavirus-mediated pathogenesis. Mol Cell, 2006, 21: 665–678.
Ristriani T, Fournane S, Orfanoudakis G, et al. A single-codon mutation converts HPV16 E6 oncoprotein into a potential tumor suppressor, which induces p53-dependent senescence of HPV-positive HeLa cervical cancer cells. Oncogene, 2009, 28: 762–772.
Martinez I, Gardiner AS, Board KF, et al. Human papillomavirus type 16 reduces the expression of microRNA-218 in cervical carcinoma cells. Oncogene, 2008, 27: 2575–2582.
Nambaru L, Meenakumari B, Swaminathan R, et al. Prognostic significance of HPV physical status and integration sites in cervical cancer. Asian Pac J Cancer Prev, 2009, 10: 355–360.
Li Y, Liu J, Yuan C, et al. High-risk human papillomavirus reduces the expression of microRNA-218 in women with cervical intraepithelial neoplasia. J Int Med Res, 2010, 38: 1730–1736.
Liu Z, Wei S, Ma H, et al. A functional variant at the miR-184 binding site in TNFAIP2 and risk of squamous cell carcinoma of the head and neck. Carcinogenesis, 2011, 32: 1668–1674.
Chen AX, Yu KD, Fan L, et al. Germline genetic variants disturbing the Let-7/LIN28 double-negative feedback loop alter breast cancer susceptibility. PLoS Genet, 2011, 7: e1002259.
Zhou X, Chen X, Hu L, et al. Polymorphisms involved in the miR-218-LAMB3 pathway and susceptibility of cervical cancer, a case-control study in Chinese women. Gynecol Oncol, 2010, 117: 287–290.
Uesugi A, Kozaki K, Tsuruta T, et al. The tumor suppressive microRNA miR-218 targets the mTOR component Rictor and inhibits AKT phosphorylation in oral cancer. Cancer Res, 2011, 71: 5765–5778.
Davidson MR, Larsen JE, Yang IA, et al. MicroRNA-218 is deleted and downregulated in lung squamous cell carcinoma. PLoS One, 2010, 5: e12560.
Lu P, Weaver VM, Werb Z. The extracellular matrix: a dynamic niche in cancer progression. J Cell Biol, 2012, 196: 395–406.
Marinkovich MP. Tumour microenvironment: laminin 332 in squamous-cell carcinoma. Nat Rev Cancer, 2007, 7: 370–380.
Culp TD, Budgeon LR, Marinkovich MP, et al. Keratinocyte-secreted laminin 5 can function as a transient receptor for human papillomaviruses by binding virions and transferring them to adjacent cells. J Virol, 2006, 80: 8940–8950.
Legg JA, Herbert JM, Clissold P, et al. Slits and Roundabouts in cancer, tumour angiogenesis and endothelial cell migration. Angiogenesis, 2008, 11: 13–21.
Wang B, Xiao Y, Ding BB, et al. Induction of tumor angiogenesis by Slit-Robo signaling and inhibition of cancer growth by blocking Robo activity. Cancer Cell, 2003, 4: 19–29.
Wang XY, Smith DI, Frederick L, et al. Analysis of EGF receptor amplicons reveals amplification of multiple expressed sequences. Oncogene, 1998, 16: 191–195.
Eley GD, Reiter JL, Pandita A, et al. A chromosomal region 7p11.2 transcript map: its development and application to the study of EGFR amplicons in glioblastoma. Neuro Oncol, 2002, 4: 86–94.
Park S, James CD. ECop (EGFR-coamplified and overexpressed protein), a novel protein, regulates NF-kappaB transcriptional activity and associated apoptotic response in an IkappaBalpha-dependent manner. Oncogene, 2005, 24: 2495–2502.
Pikarsky E, Porat RM, Stein I, et al. NF-kappaB functions as a tumour promoter in inflammation-associated cancer. Nature, 2004, 431: 461–466.
Karin M. Nuclear factor-kappaB in cancer development and progression. Nature, 2006, 441: 431–436.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Zeng, K., Zhang, W. & Hu, X. Progress of research in miR-218 and cervical cancer. Chin. -Ger. J. Clin. Oncol. 12, 399–402 (2013). https://doi.org/10.1007/s10330-012-1186-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10330-012-1186-3