Abstract
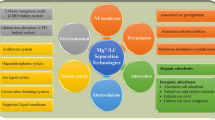
The demand for lithium is growing rapidly with the increase in electric vehicles, batteries and electronic equipments. Lithium can be extracted from brines, yet the separation of lithium ions Li+ from magnesium ions Mg2+ is challenging at high Mg/Li ratios. Here, we review methods to extract lithium from brines, such as extraction, adsorption, nanofiltration, selective electrodialysis and electrochemical intercalation/de-intercalation, with focus on the selective separation of lithium at high Mg/Li ratios. Extraction can be done with organic solvents and ionic liquids. Adsorption is performed with aluminum-, manganese- and titanate-based adsorbents.






Reproduced with permission from Huang et al. (2022) Copyright 2022, Journal of Membrane Science



Reproduced with permission from ref. (Zhang et al., 2021b) Copyright 2021, Journal of Environmental Chemical Engineering
Similar content being viewed by others
References
Al-Amshawee S et al (2020) Electrodialysis desalination for water and wastewater: A review. Chem Eng J 380:122231. https://doi.org/10.1016/j.cej.2019.122231
An JW et al (2012) Recovery of lithium from Uyuni salar brine. Hydrometallurgy 117–118:64–70. https://doi.org/10.1016/j.hydromet.2012.02.008
Bian SJ, Liu X, Gao DD, Hao Y, Li W (2013) Study on natural evaporation process of Lon-gmucuo Brine. Adv Mater Res 807–809:2408–2412. https://doi.org/10.4028/www.scientific.net/AMR.807-809.2408
Bowell RJ, Lagos L, de los Hoyos CR, Declercq J, (2020) Classification and characteristics of natural lithium resources. Elements 16(4):259–264. https://doi.org/10.2138/gselements.16.4.259
Cai C, Hanada T, Fajar ATN, Goto M (2021) An ionic liquid extractant dissolved in an ionic liquid diluent for selective extraction of Li(I) from salt lakes. Desalination 509:115073. https://doi.org/10.1016/j.desal.2021.115073
Charcosset C (2021) Classical and recent applications of membrane processes in the food industry. Food Eng Rev 13(2):322–343. https://doi.org/10.1007/s12393-020-09262-9
Chen Q-B et al (2018) Development of recovering lithium from brines by selective-electrodialy-sis: effect of coexisting cations on the migration of lithium. J Memb Sci 548:408–420. https://doi.org/10.1016/j.memsci.2017.11.040
Chen J, Lin S, Yu J (2020) Quantitative effects of Fe3O4 nanoparticle content on Li+ adsorpti-on and magnetic recovery performances of magnetic lithium-aluminum layered double hydr-oxides in ultrahigh Mg/Li ratio brines. J Hazardous Mater 388:122101. https://doi.org/10.1016/j.jhazmat.2020.122101
Christmann P, Gloaguen E, Labbé J-F, Melleton J, Piantone P, (2015) Global lithium resourc-es and sustainability issues. lithium process chemistry, pp 1–40. https://doi.org/10.1016/B978-0-12-801417-2.00001-3
Doornbusch GJ, Tedesco M, Post JW, Borneman Z, Nijmeijer K (2019) Experimental investigat-ion of multistage electrodialysis for seawater desalination. Desalination 464:105–114. https://doi.org/10.1016/j.desal.2019.04.025
Fogg AM, O’Hare D (1999) Study of the Intercalation of lithium salt in gibbsite using time-resolved in situ X-ray diffraction. Chem Mater 11(7):1771–1775. https://doi.org/10.1021/cm981151s
Globa NI, Shmatok YV, Prisyazhnyi VD (2013) Structural and electrochemical characteristics of LixMn2O4 spinel synthesized using a microwave-assisted method. Surf Eng Appl Electrochem 49(6):488–492. https://doi.org/10.3103/s1068375513060094
Goh PS, Matsuura T, Ismail AF, Hilal N (2016) Recent trends in membranes and membrane p-rocesses for desalination. Desalination 391:43–60. https://doi.org/10.1016/j.desal.2015.12.016
Gu F et al (2017) An investigation of the current status of recycling spent lithium-ion batteri-es from consumer electronics in China. J Clean Prod 161:765–780. https://doi.org/10.1016/j.jclepro.2017.05.181
Guo X, Hu S, Wang C, Duan H, Xiang X (2018a) Highly efficient separation of magnesium and lithium and high-valued utilization of magnesium from salt lake brine by a react-ion-coupled separation technology. Ind Eng Chem Res 57(19):6618–6626. https://doi.org/10.1021/acs.iecr.8b01147
Guo ZY et al (2018b) Prefractionation of LiCl from concentrated seawater/salt lake brines by electrodialysis with monovalent selective ion exchange membranes. J Cleaner Prod 193:338–350. https://doi.org/10.1016/j.jclepro.2018.05.077
He G, Zhang L, Zhou D, Zou Y, Wang F (2015) The optimal condition for H2TiO3-lithium ad-sorbent preparation and Li+ adsorption confirmed by an orthogonal test design. Ionics 21(8):2219–2226. https://doi.org/10.1007/s11581-015-1393-3
He L et al (2018) New Insights into the application of lithium-ion battery materials: selecti-ve extraction of lithium from brines via a rocking-chair lithium-ion battery system. Global Chall 2(2):1700079. https://doi.org/10.1002/gch2.201700079
Hou X-J, Li H, He P, Sun Z, Li S (2017) Structural and electronic analysis of Li/Al layered double hydroxides and their adsorption for CO2. Appl Surf Sci 416:411–423. https://doi.org/10.1016/j.apsusc.2017.04.187
Huang B, Pan Z, Su X, An L (2018) Tin-based materials as versatile anodes for alkali (earth)-ion batteries. J Power Sour 395:41–59. https://doi.org/10.1016/j.jpowsour.2018.05.063
Huang Q, Liu S, Guo Y, Liu G, Jin W (2022) Separation of mono-/di-valent ions via charged interlayer channels of graphene oxide membranes. J Membr Sci 645:120212. https://doi.org/10.1016/j.memsci.2021.120212
Isupov VP et al (2007) Mechanism of formation of nanosized copper particles in a nanoreact-or based on the [LiAl2(OH)6]2[Cuedta]·nH2O supramolecular system. J Struct Chem 48(2):350–357. https://doi.org/10.1007/s10947-007-0052-6
Jiang J, Li W, Deng H, Gong G, Li N (2019) Research on improving the electrochemical performance of LiMn2O4 via Cr-Doping. J Nanosci Nanotechnol 19(1):125–129. https://doi.org/10.1166/jnn.2019.16386
Kesler SE et al (2012) Global lithium resources: relative importance of pegmatite, brine and other deposits. Ore Geol Rev 48:55–69. https://doi.org/10.1016/j.oregeorev.2012.05.006
Kharaka YK, Hanor JSJToG, (2003) Deep fluids in the continents: i. Sediment Basins 5:605. https://doi.org/10.1016/b0-08-043751-6/05085-4
Khor A et al (2018) Review of zinc-based hybrid flow batteries: from fundamentals to applications. Mater Today Energy 8:80–108. https://doi.org/10.1016/j.mtener.2017.12.012
Kim T-H et al (2012) The current move of lithium ion batteries towards the next phase. Adv Energy Mater 2(7):860–872. https://doi.org/10.1002/aenm.201200028
Kozlova SG, Gabuda SP, Isupov VP, Chupakhina LÉ (2003) Using NMR in structural studies of aluminum hydroxide intercalation compounds with lithium salts. J Struct Chem 44(2):198–205. https://doi.org/10.1023/A:1025586321484
Lai X et al (2020) Adsorption-desorption properties of granular EP/HMO composite and its application in lithium recovery from brine. Ind Eng Chem Res 59(16):7913–7925. https://doi.org/10.1021/acs.iecr.0c00668
Lawagon CP et al (2019) Development of high capacity Li+ adsorbents from H2TiO3/polymer- nanofiber composites: systematic polymer screening, characterization and evaluation. J Ind Eng Chem 70:124–135. https://doi.org/10.1016/j.jiec.2018.10.003
Li X et al (2015a) Preparation and characterization of positively charged polyamide composite nanofiltration hollow fiber membrane for lithium and magnesium separation. Desalination 369:26–36. https://doi.org/10.1016/j.desal.2015.04.027
Li Y, Wee LH, Volodin A, Martens JA, Vankelecom IF (2015b) Polymer supported ZIF-8 me-mbranes prepared via an interfacial synthesis method. Chem Commun 51(5):918–920. https://doi.org/10.1039/c4cc06699e
Li H-F, Li L-J, Peng X-W, Ji L-M, Li W (2018) Extraction kinetics of lithium from salt lake brine by N, N-bis(2-ethylhexyl) acetamide using Lewis Cell. Hydrometallurgy 178:84–87. https://doi.org/10.1016/j.hydromet.2018.03.022
Li X et al (2019) Membrane-based technologies for lithium recovery from water lithium reso-urces: a review. J Membr Sci 591:117317. https://doi.org/10.1016/j.memsci.2019.117317
Li J et al (2022a) Synthesis of granulated Li/Al-LDHs adsorbent and application for recovery of Li from synthetic and real salt lake brines. Hydrometallurgy 209:105828. https://doi.org/10.1016/j.hydromet.2022a.105828
Li P et al (2022b) Chelation-based metal cation stabilization of graphene oxide membranes to-wards efficient sieving of mono/divalent ions. J Membr Sci 655:120604. https://doi.org/10.1016/j.memsci.2022b.120604
Li P et al (2022c) Novel high-flux positively charged aliphatic polyamide nanofiltration membrane for selective removal of heavy metals. Sep Purif Technol 280:119949. https://doi.org/10.1016/j.seppur.2021.119949
Lin S, Zhang T, Pan X, Zhang J (2021) Eco-friendly extraction of magnesium and lithium fro-m salt lake brine for lithium-ion battery. J Cleaner Prod 327:129481. https://doi.org/10.1016/j.jclepro.2021.129481
Liu X, Chen X, Zhao Z, Liang X (2014) Effect of Na+ on Li extraction from brine using LiFePO4/FePO4 electrodes. Hydrometallurgy 146:24–28. https://doi.org/10.1016/j.hydromet.2014.03.010
Liu X, Chen X, He L, Zhao Z (2015) Study on extraction of lithium from salt lake brine by membrane electrolysis. Desalination 376:35–40. https://doi.org/10.1016/j.desal.2015.08.013
Liu D et al (2019) Supply and demand response trends of lithium resources driven by the de-mand of emerging renewable energy technologies in China. Resour Conserv R-Ecycling 145:311–321. https://doi.org/10.1016/j.resconrec.2019.02.043
Liu G, Zhao Z, He L (2020) Highly selective lithium recovery from high Mg/Li ratio brines. Desalination 474:114185. https://doi.org/10.1016/j.desal.2019.114185
Liu Y et al (2022) A review on lithium extraction from natural resources. Int J Miner Metall Mater 30:209–224. https://doi.org/10.1007/s12613-022-2544-y
Lucrecia López Steinmetz R et al (2018) Northern Puna Plateau-scale survey of Li brine-type deposits in the Andes of NW Argentina. J Geochem Explor 190:26–38. https://doi.org/10.1016/j.gexplo.2018.02.013
Luo Q et al (2021) Extraction of lithium from salt lake brines by granulated adsorbents. Coll-Oids Surf a: Physicochem and Eng Aspects 628:127256. https://doi.org/10.1016/j.colsurfa.2021.127256
Marthi R, Asgar H, Gadikota G, Smith YR (2021) On the structure and lithium adsorption mechanism of layered H2TiO3. ACS Appl Mater Interfaces 13(7):8361–8369. https://doi.org/10.1021/acsami.0c20691
Meng Z et al (2022) Highly flexible interconnected Li+ ion-sieve porous hydrogels with selfre-gulating nanonetwork structure for marine lithium recovery. Chem Eng J 445:136780. https://doi.org/10.1016/j.cej.2022.136780
Mineral commodity summaries (2022). USGS Numbered Series VA. https://doi.org/10.3133/mcs2022
Moazeni M, Hajipour H, Askari M, Nusheh M (2015) Hydrothermal synthesis and characterizat-ion of titanium dioxide nanotubes as novel lithium adsorbents. Mater Res Bull 61:70–75. https://doi.org/10.1016/j.materresbull.2014.09.069
Mudring AV (2010) Tang S (2010) Ionic liquids for lanthanide and actinide chemistry. Eur J-Ournal of Inorganic Chem 18:2569–2581. https://doi.org/10.1002/ejic.201000297
Narins TP (2017) The battery business: lithium availability and the growth of the global elect-ric car industry. Extractive Ind Soc 4(2):321–328. https://doi.org/10.1016/j.exis.2017.01.013
Nayak M, Kutty TRN, Jayaraman V, Periaswamy G (1997) Preparation of the layered double hydroxide (LDH) LiAl2 (OH) 7· 2H2O, by gel to crystallite conversion and a hydrothermal method, and its conversion to lithium aluminates. J Mater Chem 7(10):2131–2137. https://doi.org/10.1039/A702065A
Nie Z, Bu L, Zheng M (2010) Lithium resources industrialization of salt lakes in China: a c-ase study of the Xitaijinaier Salt Lake and the Zabuye Salt Lake (in Chinese). Acta Geoscientica Sinic-a 31(1):95–101
Nightingale ER Jr (1959) Phenomenological theory of ion solvation Effective radii of hydrated ions. J Phys Chem 63(9):1381–1387. https://doi.org/10.1021/j150579a011
Ogawa Y, Koibuchi H, Suto K, Inoue C (2014) Effects of the Chemical Compositions of Sala-rs de Uyuni and Atacama Brines on Lithium Concentration during Evaporation. Resour Geol 64(2):91–101. https://doi.org/10.1111/rge.12030
Orooji Y et al (2022) Recent advances in nanomaterial development for lithium ion-sieving te-chnologies. Desalination 529:115624. https://doi.org/10.1016/j.desal.2022.115624
Paranthaman MP et al (2017) Recovery of lithium from geothermal brine with lithium-alu-minum layered double hydroxide chloride sorbents. Environ Sci Technol 51(22):13481–13486. https://doi.org/10.1021/acs.est.7b03464
Pulido R et al (2021) Hydrothermal control of the lithium-rich Li2MnO3 phase in lithium ma-nganese oxide nanocomposites and their application as precursors for lithium adsorbents. Dalton Trans 50(31):10765–10778. https://doi.org/10.1039/d1dt01638e
Purkait MK, Sinha MK, Mondal P, Singh R (2018) Stimuli responsive polymeric membranes: smart polymeric membranes. Academic Press, London, pp 1–37
Qiao D, Wang G, Gao T, Wen B, Dai T (2021) Potential impact of the end-of-life batteries r-ecycling of electric vehicles on lithium demand in China: 2010–2050. Sci Total Environ 764:142835. https://doi.org/10.1016/j.scitotenv.2020.142835
Quist-Jensen CA, Ali A, Drioli E, Macedonio F (2019) Perspectives on mining from sea and o-ther alternative strategies for minerals and water recovery-The development of novel mem-brane operations. J Taiwan Inst Chem E 94:129–134. https://doi.org/10.1016/j.jtice.2018.02.002
Razmjou A, Asadnia M, Hosseini E, Habibnejad Korayem A, Chen V (2019) Design principles of ion selective nanostructured membranes for the extraction of lithium ions. Nat Commu-n 10(1):5793. https://doi.org/10.1038/s41467-019-13648-7
Ren Z et al (2021) Highly selective extraction of lithium ions from salt lake brines with sodi-um tetraphenylborate as co-extractant. Sep Purif Technol 269:118756. https://doi.org/10.1016/j.seppur.2021.118756
Richards LA, Schäfer AI, Richards BS, Corry BJS (2012) The importance of dehydration in determining ion transport in narrow pores. Small 8(11):1701–1709. https://doi.org/10.1002/smll.201102056
Sahu S, Di Ventra M, Zwolak M (2017) Dehydration as a universal mechanism for ion select-ivity in graphene and other atomically thin pores. Nano Lett 17(8):4719–4724. https://doi.org/10.1021/acs.nanolett.7b01399
Shang X et al (2021) LiNi0.5Mn1.5O4-based hybrid capacitive deionization for highly selective adsorption of lithium from brine. Sep Purif Technol 258:118009. https://doi.org/10.1016/j.seppur.2020.118009
Shi C, Duan D, Jia Y, Jing Y (2014) A highly efficient solvent system containing ionic liquid in tributyl phosphate for lithium ion extraction. J Mol Liq 200:191–195. https://doi.org/10.1016/j.molliq.2014.10.004
Shi C, Jia Y, Zhang C, Liu H, Jing Y (2015) Extraction of lithium from salt lake brine using room temperature ionic liquid in tributyl phosphate. Fusion Eng Des 90:1–6. https://doi.org/10.1016/j.fusengdes.2014.09.021
Shi D et al (2019) Lithium extraction from low-grade salt lake brine with ultrahigh Mg/Li ra-tio using TBP-kerosene-FeCl3 system. Sep Purif Technol 211:303–309. https://doi.org/10.1016/j.seppur.2018.09.087
Snydacker DH, Hegde VI, Aykol M, Wolverton C (2018) Computational discovery of Li-M-O ion exchange materials for lithium extraction from brines. Chem Mater 30(20):6961–6968. https://doi.org/10.1021/acs.chemmater.7b03509
Somrani A, Hamzaoui AH, Pontie M (2013) Study on lithium separation from salt lake brines by nanofiltration (NF) and low pressure reverse osmosis (LPRO). Desalination 317:184–192. https://doi.org/10.1016/j.desal.2013.03.009
Song Y et al (2017) Insight into the atomic structure of Li2MnO3 in Li-rich Mn-based catho-de materials and the impact of its atomic arrangement on electrochemical performance. J Mater Chem A 5(22):11214–11223. https://doi.org/10.1039/c7ta02151h
Su H et al (2020) Recovery of lithium from salt lake brine using a mixed ternary solvent ex-traction system consisting of TBP, FeCl3 and P507. Hydrometallurgy 197:105487. https://doi.org/10.1016/j.hydromet.2020.105487
Sun X, Luo H, Dai S (2012) Ionic liquids-based extraction: a promising strategy for the adva-nced nuclear fuel cycle. Chem Rev 112(4):2100–2128. https://doi.org/10.1021/cr200193x
Sun SY, Xiao JL, Wang J, Song X, Yu JG (2014) Synthesis and adsorption properties of Li1.6Mn1.6O4 by a combination of redox precipitation and solid-phase reaction. Ind Eng Chem Res 53(40):15517–15521. https://doi.org/10.1021/ie5004625
Sun SY, Cai LJ, Nie XY, Song X, Yu JG (2015) Separation of magnesium and lithium fro-m brine using a Desal nanofiltration membrane. J Water Process Eng 7:210–217. https://doi.org/10.1016/j.jwpe.2015.06.012
Sun X, Hao H, Hartmann P, Liu Z, Zhao F (2019) Supply risks of lithium-ion battery materia-ls: An entire supply chain estimation. Materials Today Energy 14:100347. https://doi.org/10.1016/j.mtener.2019.100347
Swain B (2017) Recovery and recycling of lithium: a review. Sep Purif Technol 172:388–403. https://doi.org/10.1016/j.seppur.2016.08.031
Takemoto M et al (2021) Synthesis of high-specific-surface-area Li-Al mixed metal oxide: th-rough nanoseed-assisted growth of layered double hydroxide. Appl Clay Sci 203:106006. https://doi.org/10.1016/j.clay.2021.106006
Tansel B et al (2006) Significance of hydrated radius and hydration shells on ionic permeabilit-y during nanofiltration in dead end and cross flow modes. Sep Purif Technol 51(1):40–47. https://doi.org/10.1016/j.seppur.2005.12.020
Volkov AG, Paula S, Deamer DW (1997) Two mechanisms of permeation of small neutral molecules and hydrated ions across phospholipid bilayers. Bioelectrochem Bioenergeti-Cs 42(2):153–160. https://doi.org/10.1016/S0302-4598(96)05097-0
Wahib SA, Da’na DA, Zaouri N, Hijji YM, Al-Ghouti MA (2022) Adsorption and recovery of lithium ions from groundwater using date pits impregnated with cellulose nanocrystals and ionic liquid. J Hazard Mater 421:126657. https://doi.org/10.1016/j.jhazmat.2021.126657
Wang L, Lin Y, Tang Y, Ren D, Wang X (2021) Fabrication of oppositely charged thin-film c-omposite polyamide membranes with tunable nanofiltration performance by using a pipera-zine derivative. J Membr Sci 634:119405. https://doi.org/10.1016/j.memsci.2021.119405
Wang Z et al (2022) Ultrathin polyamide nanofiltration membranes with tunable chargeability f-or multivalent cation removal. J Membr Sci 642:119971. https://doi.org/10.1016/j.memsci.2021.119971
Wei S, Wei Y, Chen T, Liu C, Tang Y (2020) Porous lithium ion sieves nanofibers: General synthesis strategy and highly selective recovery of lithium from brine water. Chem Eng J 379:122407. https://doi.org/10.1016/j.cej.2019.122407
Weng D et al (2020) Introduction of manganese based lithium-ion Sieve-A review. Progress Nat Sci: Mater Int 30(2):139–152. https://doi.org/10.1016/j.pnsc.2020.01.017
Wietelmann U, Steinbild M (2014) Lithium and Lithium Compounds. In Ullmann's Encyclopedia of Industrial Chemistry pp. 1–38. https://doi.org/10.1002/14356007.a15_393.pub2
Wu QX, Pan ZF, An L (2018) Recent advances in alkali-doped polybenzimidazole membranes for fuel cell applications. Renew Sustain Energy Rev 89:168–183. https://doi.org/10.1016/j.rser.2018.03.024
Xiang W, Liang S, Zhou Z, Qin W, Fei W (2016) Extraction of lithium from salt lake brine containing borate anion and high concentration of magnesium. Hydrometallurgy 166:9–15. https://doi.org/10.1016/j.hydromet.2016.08.005
Xiang W, Liang S, Zhou Z, Qin W, Fei W (2017) Lithium recovery from salt lake brine by counter-current extraction using tributyl phosphate/FeCl3 in methyl isobutyl ketone. Hydro-Metallurgy 171:27–32. https://doi.org/10.1016/j.hydromet.2017.04.007
Xiao W et al (2018) Insight into fast Li diffusion in Li-excess spinel lithium manganese oxid-e. J Mater Chem A 6(21):9893–9898. https://doi.org/10.1039/c8ta01428k
Xu P et al (2019) Positive charged PEI-TMC composite nanofiltration membrane for separation of Li+ and Mg2+ from brine with high Mg2+/Li+ ratio. Desalination 449:57–68. https://doi.org/10.1016/j.desal.2018.10.019
Xu T et al (2020) Ti-exchanged UiO-66-NH2–containing polyamide membranes with remarkabl-e cation permselectivity. J Membr Sci 615:118608. https://doi.org/10.1016/j.mem-sci.2020.118608
Xu S et al (2021) Extraction of lithium from Chinese salt-lake brines by membranes: design and practice. J Memb Sci 635:119441. https://doi.org/10.1016/j.memsci.2021.119441
Xu W, He L, Zhao Z (2021) Lithium extraction from high Mg/Li brine via electrochemical i-ntercalation/de-intercalation system using LiMn2O4 materials. Desalination 503:114935. https://doi.org/10.1016/j.desal.2021.114935
Yang X, Yan L, Ma J, Bai Y, Shao L (2019) Bioadhesion-inspired surface engineering constru-cting robust, hydrophilic membranes for highly-efficient wastewater remediation. J Memb Sci 591:117353. https://doi.org/10.1016/j.memsci.2019.117353
Yu M, Li H, Du N, Hou W (2019) Understanding Li-Al-CO3 layered double hydroxides. (I) Urea-supported hydrothermal synthesis. J Colloid Interface Sci 547:183–189. https://doi.org/10.1016/j.jcis.2019.03.101
Zhang H et al (2018) Ultrafast selective transport of alkali metal ions in metal organic frameworks with subnanometer pores. Sci Adv 4(2):eaqq0066. https://doi.org/10.1126/sciadv.aaq0066
Zhang H, He Q, Luo J, Wan Y, Darling SB (2020a) Sharpening nanofiltration: strategies for enhanced membrane selectivity. ACS Appl Mater Interfaces 12(36):39948–39966. https://doi.org/10.1021/acsami.0c11136
Zhang C, Mu Y, Zhang W, Zhao S, Wang Y (2020b) PVC-based hybrid membranes containing metal-organic frameworks for Li+/Mg2+ separation. J Membr Sci 596:117724. https://doi.org/10.1016/j.memsci.2019.117724
Zhang S et al (2021a) Thermally regenerable metal-organic framework with high monovalent metal ion selectivity. Chem Eng J 405:127037. https://doi.org/10.1016/j.cej.2020.127037
Zhang X-C et al (2021b) Preparation of Li2CO3 from high Mg2+/Li+ brines based on selective electrodialysis with feed and bleed mode. J Environ Chem Eng 9(6):106635. https://doi.org/10.1016/j.jece.2021b.106635
Zhao Z, Si X, Liu X, He L, Liang X (2013) Li extraction from high Mg/Li ratio brine with LiFePO4/FePO4 as electrode materials. Hydrometallurgy 133:75–83. https://doi.org/10.1016/j.hydromet.2012.11.013
Zhao Y, Wang H, Li Y, Wang M, Xiang X (2020) An integrated membrane process for prepa-ration of lithium hydroxide from high Mg/Li ratio salt lake brine. Desalination 493:114620. https://doi.org/10.1016/j.desal.2020.114620
Zhao B, Qian Z, Guo M, Wu Z, Liu Z (2021) The performance and mechanism of recovering lithium on H4Ti5O12 adsorbents influenced by (110) and (111) facets exposed. Chem Eng J 414:128729. https://doi.org/10.1016/j.cej.2021.128729
Zhong J, Lin S, Yu J (2021) Li+ adsorption performance and mechanism using lithium/alumin-um layered double hydroxides in low grade brines. Desalination 505(1):114983. https://doi.org/10.1016/j.desal.2021.114983
Zhou Z, Qin W, Liang S, Tan Y, Fei W (2012a) Recovery of Lithium Using Tributyl Phosph-ate in Methyl Isobutyl Ketone and FeCl3. Ind Eng Chem Res 51(39):12926–12932. https://doi.org/10.1021/ie3015236
Zhou Z, Qin W, Fei W, Li Y (2012b) A study on stoichiometry of complexes of tributyl p-hosphate and methyl isobutyl ketone with lithium in the presence of FeCl3. Chin J Chem Eng 20(1):36–39. https://doi.org/10.1016/S1004-9541(12)60360-7
Zhou Z, Qin W, Chu Y, Fei W (2013) Elucidation of the structures of tributyl phosphate/Li c-omplexes in the presence of FeCl3 via UV–visible, Raman and IR spectroscopy and the method of continuous variation. Chem Eng Sci 101:577–585. https://doi.org/10.1016/j.ces.2013.07.020
Zhou W, Xu S, Li Z (2021) Recovery of lithium from brine with a High Mg/Li Ratio Using hydroxyl-functionalized ionic liquid and tri-n-butyl phosphate. J Sustain Metall 7(1):256–265. https://doi.org/10.1007/s40831-020-00331-1
Zhu X et al (2021a) Study on adsorption extraction process of lithium ion from West Taijinar brine by shaped titanium-based lithium ion sieves. Sep Purif Technol 274:119099. https://doi.org/10.1016/j.seppur.2021a.119099
Zou C et al (2021) The role of new energy in carbon neutral. Petrol Explor Dev 48(2):480–491. https://doi.org/10.1016/s1876-3804(21)60039-3
Acknowledgements
Financial aid from the following programs is gratefully acknowledged: National Key research and development Program (Grant No. 2021YFC2901300).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing financial interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhu, R., Wang, S., Srinivasakannan, C. et al. Lithium extraction from salt lake brines with high magnesium/lithium ratio: a review. Environ Chem Lett 21, 1611–1626 (2023). https://doi.org/10.1007/s10311-023-01571-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10311-023-01571-9