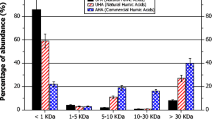
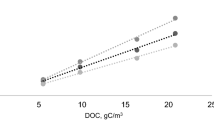
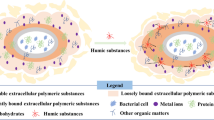
Abstract
Humic acids are complex mixtures of organic molecules of different sizes, molecular weights and functional groups such as phenols, carboxyls, quinones and amino acids. Humic acids occur ubiquitously in media where organic matter is decomposing, such as waters, soils, sediments and organic wastes. The presence of humic acids in untreated water inhibits the oxidation of target pollutants through competing reactions and generates toxic disinfection by-products during the classical disinfection method of chlorination. There is therefore a need for alternative methods that remove humic acids. Here, degradation of humic acids at 10 mg/L was tested using 1000 W microwave irradiation assisted by 0.25–1.2 mM persulfate, with granular activated carbon at 10–50 g/L, at pH 3.0–12.0. In 90s, the highest removal of humic acids, of 75%, and humic acids mineralization, of 41%, was obtained using 1000 W microwave, 50 g/L granular activated carbon and 0.5 mM persulfate at pH 8.0. Under the same conditions without persulfate, removal was only 42% and mineralization 24%. Removal was lower than 7% using either persulfate or microwave alone. High removal with microwave, persulfate and granular activated carbon may be explained by enhanced generation of SO ·−4 and ·OH radicals and also by better trapping/encapsulation/binding of humic acids in the granular activated carbon matrix.



Similar content being viewed by others
References
Bo LL, Zhang YB, Quan X, Zhao B (2008) Microwave assisted catalytic oxidation of p-nitrophenol in aqueous solution using carbon-supported copper catalyst. J Hazard Mater. https://doi.org/10.1016/j.jhazmat.2007.09.082
Chen YQ, Deng PY, Xie PC, Shang R, Wang ZP, Wang SL (2017) Heat-activated persulfate oxidation of methyl- and ethyl-parabens: effect, kinetics, and mechanism. Chemosphere. https://doi.org/10.1016/j.chemosphere.2016.11.143
Chou YC, Lo SL, Kuo J, Yeh CJ (2015) Microwave-enhanced persulfate oxidation to treat mature landfill leachate. J Hazard Mater. https://doi.org/10.1016/j.jhazmat.2014.10.043
Feng M, Wang X, Chen J, Qu R, Sui Y, Cizmas L, Wang Z, Sharma VK (2016) Degradation of fluoroquinolone antibiotics by ferrate(VI): effects of water constituents and oxidized products. Water Res. https://doi.org/10.1016/j.watres.2016.07.014
Feng M, Wang Z, Dionysiou DD, Sharma VK (2017) Metal-mediated oxidation of fluoroquinolone antibiotics in water: a review on kinetics, transformation products, and toxicity assessment. J Hazard Mater. https://doi.org/10.1016/j.jhazmat.2017.08.067
Gayathri PV, Yesodharan S, Yesodharan EP (2017) Purification of water contaminated with traces of Rhodamine B dye by microwave-assisted, oxidant-induced and zinc oxide catalyzed advanced oxidation process. Desalin Water Treat. https://doi.org/10.5004/dwt.2017.21286
Ghanbari F, Moradi M (2017) Application of peroxymonosulfate and its activation methods for degradation of environmental organic pollutants: review. Chem Eng J. https://doi.org/10.1016/j.cej.2016.10.064
Heeb MB, Criquet J, Zimmermann-Steffens SG, Von Gunten U (2014) Oxidative treatment of bromide-containing waters: formation of bromine and its reactions with inorganic and organic compounds—a critical review. Water Res. https://doi.org/10.1016/j.watres.2013.08.030
Hua LC, Lin JL, Chen PC, Huang CP (2017) Chemical structures of extra- and intra-cellular algogenic organic matters as precursors to the formation of carbonaceous disinfection byproducts. Chem Eng J. https://doi.org/10.1016/j.cej.2017.07.123
Jasemizad T, Ghaneian MT, Ehrampoush MH, Rezapour I, Jebali A, Sahlabadi F (2016) Effects of radical scavengers on humic acid removal by electron beam irradiation. Desalin Water Treat. https://doi.org/10.1080/19443994.2016.1177798
Jiang JY, Zhang XG, Zhu XH, Li Y (2017) Removal of intermediate aromatic halogenated DBPs by activated carbon adsorption: a new approach to controlling halogenated DBPs in chlorinated drinking water. Environ Sci Technol. https://doi.org/10.1021/acs.est.6b06161
Kim YB, Ahn JH (2017) Changes of absorption spectra, SUVA(254), and color in treating landfill leachate using microwave-assisted persulfate oxidation. Korean J Chem Eng. https://doi.org/10.1007/s11814-017-0104-3
Leenheer JA, Croué JP (2003) Peer reviewed: characterizing aquatic dissolved organic matter understanding the unknown structures is key to better treatment of drinking water. Environ Sci Technol 37(1):18A–26A. https://doi.org/10.1021/es032333c
Li C, Dong F, Feng L, Zhao J, Zhang T, Cizmas L, Sharma VK (2017) Bacterial community structure and microorganism inactivation following water treatment with ferrate(VI) or chlorine. Environ Chem Lett 15(3):525–530. https://doi.org/10.1007/s10311-017-0623-5
Liao W, Sharma VK, Xu S, Li Q, Wang L (2017a) Microwave-enhanced photolysis of norfloxacin: kinetics, matrix effects, and degradation pathways. Int J Environ Res Public Health 14:1564. https://doi.org/10.3390/ijerph14121564
Liao YJ, Chen WC, Borsali R (2017b) Carbohydrate-based block copolymer thin films: ultrafast nano-organization with 7 nm resolution using microwave energy. Adv Mater 29(35):1701645. https://doi.org/10.1002/adma.201701645
Lichtfouse E (1997) Soil, a sponge for pollutants. Analusis, EDP Sci 25(8):M16–M23. https://hal.archives-ouvertes.fr/hal-00193259/
Liu XT, Quan X, Bo LL, Chen S, Zhao YZ, Chang M (2004) Temperature measurement of GAC and decomposition of PCP loaded on GAC and GAC-supported copper catalyst in microwave irradiation. Appl Catal A Gen. https://doi.org/10.1016/j.apcata.2003.12.026
Lv SS, Chen XG, Ye Y, Yin SH, Cheng JP, Xia MS (2009) Rice hull/MnFe2O4 composite: preparation, characterization and its rapid microwave-assisted COD removal for organic wastewater. J Hazard Mater. https://doi.org/10.1016/j.jhazmat.2009.06.039
Ma J, Yang Y, Jiang X, Xie Z, Li X, Chen C, Chen H (2018) Impacts of inorganic anions and natural organic matter on thermally activated persulfate oxidation of BTEX in water. Chemosphere. https://doi.org/10.1016/j.chemosphere.2017.09.148
McKnight D, Thurman EM, Wershaw RL, Hemond H (1985) Biogeochemistry of aquatic humic substances in thoreau's bog, concord, massachusetts. Ecol 66(4):1339–1352. https://doi.org/10.2307/1939187
Ni QQ, Zhu YF, Yu LJ, Fu YQ (2015) One-dimensional carbon nanotube@barium titanate@polyaniline multiheterostructures for microwave absorbing application. Nanoscale Res Lett. https://doi.org/10.1186/s11671-015-0875-6
Quan X, Zhang YB, Chen S, Zhao YZ, Yang FL (2007) Generation of hydroxyl radical in aqueous solution by microwave energy using activated carbon as catalyst and its potential in removal of persistent organic substances. J Mol Catal A Chem. https://doi.org/10.1016/j.molcata.2006.08.079
Remya N, Lin JG (2011a) Current status of microwave application in wastewater treatment—a review. Chem Eng J. https://doi.org/10.1016/j.cej.2010.11.100
Remya N, Lin JG (2011b) Microwave-assisted carbofuran degradation in the presence of GAC, ZVI and H2O2: influence of reaction temperature and pH. Sep Purif Technol. https://doi.org/10.1016/j.seppur.2010.10.012
Remya N, Lin JG (2015) Microwave-granular activated carbon (MW-GAC) system for carbofuran degradation: analysis of characteristics and recyclability of the spent GAC. Desalin Water Treat. https://doi.org/10.1080/19443994.2013.855673
Richardson SD, Kimura SY (2016) Water analysis: emerging contaminants and current issues. Anal Chem 88:546–582. https://doi.org/10.1021/acs.analchem.5b04493
Sharma VK (2013) Oxidation of amino acids, peptides, and proteins. Wiley, Hoboken
Sharma VK, Yang X, Cizmas L, McDonald TJ, Luque R, Sayes CM, Yuan B, Dionysiou DD (2017) Impact of metal ions, metal oxides, and nanoparticles on the formation of disinfection byproducts during chlorination. Chem Eng J. https://doi.org/10.1016/j.cej.2017.02.071
Sillanpää M, Mohamed CN, Matilainen A (2017) Advanced oxidation processes for the removal of natural organic matter from drinking water sources: a comprehensive review. J Environ Manag 208:56–76. https://doi.org/10.1016/j.jenvman.2017.12.009
Song SA, Huang SY, Zhang R, Chen ZS, Wen T, Wang SH, Wang XK (2017) Simultaneous removal of U(VI) and humic acid on defective TiO2-x investigated by batch and spectroscopy techniques. Chem Eng J. https://doi.org/10.1016/j.cej.2017.05.125
Stevenson FJ (1982) Humus chemistry: genesis, composition, reactions. Wiley, New York, pp 35–37
Trellu C, Mousset E, Pechaud Y, Huguenot D, Van Hullebusch ED, Esposito G, Oturan MA (2016) Removal of hydrophobic organic pollutants from soil washing/flushing solutions: a review. J Hazard Mater 306:149–174. https://doi.org/10.1016/j.jhazmat.2015.12.008
Villata LS, Gonzalez MC, Martire DO (2010) A kinetic study of the reactions of sulfate and dihydrogen phosphate radicals with epicatechin, epicatechingallate, and epigalocatechingallate. Int J Chem Kinet 42(7):391–396. https://doi.org/10.1002/kin.20492
Waclawek S, Lutze HV, Grubel K, Padil VVT, Cernik M, Dionysiou DD (2017) Chemistry of persulfates in water and wastewater treatment: a review. Chem Eng J. https://doi.org/10.1016/j.cej.2017.07.132
Wall NA, Choppin GR (2003) Humic acids coagulation: influence of divalent cations. Appl Geochem 18(10):1573–1582. https://doi.org/10.1016/S0883-2927(03)00046-5
Wang NN, Wang P (2016) Study and application status of microwave in organic wastewater treatment—a review. Chem Eng J. https://doi.org/10.1016/j.cej.2015.07.046
Wei MC, Wang KS, Lin IC, Hsiao TE, Lin YN, Tang CT, Chang SH (2012) Rapid regeneration of sulfanilic acid-sorbed activated carbon by microwave with persulfate. Chem Eng J. https://doi.org/10.1016/j.cej.2012.04.072
Yang SY, Wang P, Yang X, Wei G, Zhang WY, Shan L (2009) A novel advanced oxidation process to degrade organic pollutants in wastewater: microwaveactivated persulfate oxidation. J Environ Sci. https://doi.org/10.1016/S1001-0742(08)62399-2
Zhen GY, Lu XQ, Kato H, Zhao YC, Li YY (2017) Overview of pretreatment strategies for enhancing sewage sludge disintegration and subsequent anaerobic digestion: current advances, full-scale application and future perspectives. Renew Sustain Energy Rev. https://doi.org/10.1016/j.rser.2016.11.187
Zhong X, Cui C, Yu S (2017) Identification of oxidation intermediates in humic acid oxidation. Ozone Sci Eng. https://doi.org/10.1080/01919512.2017.1392845
Zhou L, Sleiman M, Ferronato C, Chovelon J, Richard C (2017) Reactivity of sulfate radicals with natural organic matters. Environ Chem Lett 15:733–737. https://doi.org/10.1007/s10311-017-0646-y
Acknowledgements
This work was supported by the National Key Research and Development Program of China (2016YFC0402103) and Science and Technology Research Project of Chongqing Municipal Education Committee (KJ1600527, CY 160704).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Zhang, X., Ding, Z., Yang, J. et al. Efficient microwave degradation of humic acids in water using persulfate and activated carbon. Environ Chem Lett 16, 1069–1075 (2018). https://doi.org/10.1007/s10311-018-0721-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10311-018-0721-z