Abstract
Food toxins are produced as defense tools by microorganisms that use nutrients for their growth. Microorganisms thus spoil food, taste and can infect humans, sometimes leading to death. Food adulteration and brand protection are also major issues in the food industry. Here we review the use of nanomaterials for sensing food quality. Nanosensors can detect pathogenic bacteria, food-contaminating toxins, adulterant, vitamins, dyes, fertilizers, pesticides, taste and smell. Food freshness can be monitored using time–temperature and oxygen indicators. Product authenticity and brand protection can be assessed using invisible nanobarcodes. Overall, nanosensors with unique properties are improving food security.
Similar content being viewed by others
Introduction
Food products have high nutritional value that lead to their easy contamination by microorganisms. Microorganisms contaminate food and generate toxins in self-defense to cause several diseases in humans. Food adulteration also induces ill effect to human health. The toxin and adulterant can induce vomiting, diarrhea to paralysis and even death. Being primary and fundamental need of humans, the food safety is a global concern. The health problem is any fold worse in case of warfare use of such toxins or chemicals. Hence, there is urgent need to develop quick and sensitive approaches to detect the harmful chemicals, bacteria and related toxins (Franz et al. 1997; Leggiadro 2000; Zhu et al. 2009). Due to their unique properties nanoparticles are increasingly employed to develop detection techniques for sensing contamination, adulteration and freshness of food materials.
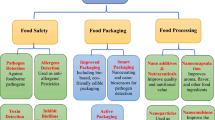
Proper packaging of food product prevents them from moisture, contamination and spoilage. Traditional passive packaging systems act as passive barrier against air, dust and moisture. However, the passive packaging systems fail to address increasing concern of food safety and bioterrorism (Vermeiren et al. 1999), whereas intelligent packaging system can sense the quality of food products and protect the shelf life and brand name of packed food. Intelligent packaging uses various smart packaging devices like barcodes, time–temperature indicators, gas indicators and biosensors. As the existing barcodes-based protection tags are very easy to manipulate, nanoparticles-based invisible and sophisticated tags would be very useful to verify the originality of food products in future (Banu et al. 2006; Birtwell et al. 2008). Sensing is an important part of intelligent packaging system (Farahi et al. 2012). Present article describes the food sensing application of nanoparticles (Fig. 1). This article is an abridged version of the chapter published by Kumar et al. (2016) in the series Sustainable Agriculture Reviews (http://www.springer.com/series/8380).
Nanosensors of microorganisms, toxins and adulterants
The food items are good to consume only if they are fresh and free from adulterants and contaminants. Detection of food adulterant and contaminants at low level using routine detection system is a challenging task. So, nanoparticles were explored to detect the toxic chemicals and microorganisms with high sensitivity (Table 1).
Detection of food pathogenic bacteria
Pathogenic bacterial detection in food materials is mainly achieved by identifying the bacterial genetic material or whole bacterial cell. Nanoparticles-assisted deoxyribonucleic acid (DNA) isolation and bacteria detection was less time-consuming and more sensitive than other conventional methods. Magnetic iron oxide nanoparticles have been used for isolating DNA of milk pathogenic bacterium Listeria monocytogenes. The DNA isolated from L. monocytogenes-contaminated milk sample was quantified using polymerase chain reaction (PCR) (Yang et al. 2007).
The 16s ribosomal ribonucleic acid is commonly used as a selective marker for PCR-based microbial detection. The PCR-based assay is costly and 16s ribosomal ribonucleic acid-based microarray method lacks sensitivity (Call et al. 2003). However, the nanoparticles-based detection of 16s ribosomal ribonucleic acid is easy and more sensitive (Joung et al. 2008). Various nanoparticles have been documented for detecting pathogenic bacteria in standard bacterial culture samples as well as complex food samples (Table 1).
Detection of food-contaminating toxins
Aflatoxins are a group of toxic and carcinogenic compounds found in food contaminated with Aspergillus flavus and Aspergillus parasiticus. Gold nanoparticles functionalized with anti-aflatoxin antibodies have been used for the detection of aflatoxin B1 (Table 2).
Likewise, superparamagnetic beads containing anti-aflatoxin M1 antibodies and gold nanoprobes have also been used for the detection of aflatoxin M1 in milk sample (Fig. 2).
Dynamic light scattering-based detection of aflatoxin M1 using gold nanoparticle-based probes. In aflatoxin M1-contaminated samples, the aflatoxin M1 displaced nanoprobes by competitive binding to antibodies. The concentration of free nanoprobes was measured using dynamic light scattering reading that was proportional to quantity of aflatoxin M1 in test samples. Figure adapted with permission from (Zhang et al. 2013a). Copyright (2013) American Chemical Society
Gold nanoparticles-based immunochromatographic strip method has been employed for the detection of aflatoxin M1 in milk. The aflatoxin M1-contaminated milk sample appears as colorless zone on the strip, while in the absence of aflatoxin M1 red color band appears (Wang et al. 2011). Contaminated seafood generally contains marine toxin, namely palytoxin. Carbon nanotubes-based electrochemiluminescent sensors have been designed for the ultrasensitive detection of palytoxin in mussel meat (Zamolo et al. 2012). Various nanoparticles have been reported for the detection of food-contaminating toxins (Table 2).
Detection of food-contaminating pesticides and chemicals
Nanoparticles have been used for the detection of pesticides, fertilizers and other toxic chemicals (Table 3). Among the various pesticides, organophosphates are the most common (Vamvakaki and Chaniotakis 2007). Gold nanoparticles have been used as colorimetric and fluorometric sensors for the detection of organophosphorus and carbamate pesticides (Liu et al. 2012). Cadmium selenide and cadmium selenide–zinc sulfide core–shell quantum dots have been explored for the pesticide paraoxon sensing (Ji et al. 2005). Selective binding of phosphate group containing pesticide parathion to zirconium dioxide/gold nanocomposite film electrode has been employed for developing voltammetric biosensors (Wang and Li 2008). MWCNT–silica nanocomposite-based potentiometer sensors have been documented for the detection of toxic cadmium ions (Bagheri et al. 2013).
Like pesticides, excessive use of fertilizers is also a big concern. Melamine is a fertilizer, and it is used as adulterant in protein-rich products such as egg, biscuits, candy and coffee drinks. Gold nanoparticles-based fluorescence sensors were able to detect even picomolar concentration of melamine in cow milk and infant formulas (Vasimalai and John 2013). Likewise, other nanoparticles have also been reported for melamine sensing (Table 3). Food dyes and preservatives are also toxic when used above permissible limit. Multi wall carbon nanotubes (MWCNT)–ionic liquid nanocomposites modified carbon–ceramic electrodes have been used for the detection of food dyes, sunset yellow and tartrazine (Majidi et al. 2013). Cobalt nitroprusside nanoparticles has been used for the detection of sulfite in sugar, dry fruits and wine (Devaramani and Malingappa 2012).
Chloramphenicol is a low-cost, but toxic broad-spectrum antibiotic. But it is still used to cure infections in bees, and as a result, honey gets contaminated with antibiotics. Poly(ethylene glycol dimethacrylate-N-methacryloyl-l-histidine methylester) nanoparticles have been used for the detection of chloramphenicol in honey samples (Kara et al. 2013). Sudan I is a carcinogenic red dye used as adulterant in chili powder. Multi-wall carbon nanotubes (MWCNTs) have been used for the detection of Sudan I adulteration in chili powder (Yang et al. 2010). Likewise, nanoparticles were used for the detection of Sudan I in chili powder, egg yolk, ketchup, tomato, chilli and strawberry sauce (Table 3). SWCNT and MWCNT–zinc oxide nanocomposite has been documented for the simultaneous detection of bisphenol A and Sudan I. Bisphenol A is a toxic contaminant released from plastic container (Sanchez-Acevedo et al. 2009; Najafi et al. 2014).
Nanosensors for food freshness detection
The food ingredients generally get spoiled on storage longer than shelf life and exposure of air and moisture. Laboratory-based food spoilage testing is not possible for individual packages. Alternatively, nanoparticles based on spot indicators are sensitive and can be easily labeled on individual packages (Realini and Marcos 2014; Jiang et al. 2015).
Food quality assessment due to improper storage
Besides adulteration and contamination by bacteria or toxin, some food products are perishable and prone to degradation. Some food items perish on change in storage time and temperature. Traditional time–temperature indicators are costly and lack flexibility in programming. So, nanoparticles-based time–temperature indicators have been developed to overcome these limitations. The change in color, size shape and spectral properties of nanoparticles has been explored for developing time–temperature indicator (Table 4).
Freshness of packed food items is spoiled on exposure to oxygen exposure. Oxygen promotes the oxidation of antioxidants and, hence, induces the growth of bacteria. Colorimetric oxygen indicators have been produced for this purpose using methylene blue/titanium dioxide hybrid nanocomposite material (Gutierrez-Tauste et al. 2007). Oxygen sensor was also developed using luminescent metal–ligand complex functionalized poly-(styrene-block-vinylpyrrolidone) nanobeads (Borisov and Klimant 2009). Trimethylamine is generally produced as a result of metabolism of animal proteins with passage of time. So, presence of trimethylamine is also an indicator of loss of freshness. Tin dioxide–zinc oxide nanocomposite sensors were effectively employed for the detection of trimethylamine in fish samples (Zhang and Zhang 2008). Like zinc oxide microrods, polyvinylpyrrolidone-capped zinc oxide nanoparticles and branched iron oxide–titanium dioxide heteronanostructure have also been used for trimethylamine sensing (Tang et al. 2006; Lou et al. 2013). Likewise, xanthine and hypoxanthine found in meat undergo degradation with time to release hydrogen peroxide. Hydrogen peroxide was detected by gold nanoparticles (Cubukcu et al. 2007).
Sensing the quality of unstable key food ingredients during food processing and storage
Vitamins and other antioxidant components present in food products are easily degraded. Nanoparticles have been used for the detection of vitamins in food items (Table 5). Deficiency of water-soluble vitamin folic acid can cause anemia, carcinogenesis and heart attack. MWCNT and SWCNT–ionic liquid nanocomposites have been reported for the detection of folic acid in wheat flour, fruit juices and milk samples (Wei et al. 2006; Xiao et al. 2008). Nickel oxide nanoparticles have been used for the detection of vitamin, ascorbic acid (Karimi-Maleh et al. 2014). Likewise, N-(3,4-dihydroxyphenethyl)-3,5-dinitrobenzamide modified MWCNT has been used for sensing ascorbic acid and essential amino acid, tryptophan (Ensafi et al. 2012).
The antioxidant value of red wine is due to its phenolic content. Tyrosinase enzyme immobilized gold nanoparticles was used to detect the quality of phenol in red and white wines (Liu et al. 2003; Sanz et al. 2005). Hydrogen peroxide is used as an antioxidant in food industry, but higher concentration of hydrogen peroxide can induce toxic effects in humans. Various nanoparticles were reported for the hydrogen peroxide sensing (Table 5). The level of glucose, fructose, sucrose, d-sorbitol, l-malic acid, citric acid, succinic acid, l-glutamic acid, hydrogen peroxide and alcohol during food processing and stored product is used as indicators of food quality (Verstrepen et al. 2004; Terry et al. 2005; Vermeir et al. 2007). Nanoparticles have also been used to sense the quality of such food component as shown in Table 5.
Nanobarcodes for product authenticity
Barcodes are globally used as product authentication labels. Commonly used two-dimensional barcodes can be easily located and, hence, are more prone to damage, alteration and falsification. However, at the same time, nanoparticle-based invisible barcodes are hard to manipulate (Wang et al. 2015b). Unique nanoparticle-based encoding system and nanodisk codes have been reported recently. Nanodisk code is a sequence of surface-enhanced Raman scattering producing disk pairs that can be scanned with Raman microscope (Qin et al. 2007). Authors have documented linear arrays of gold nanodisk, silver nanodisk and silver–gold heterodimer nanodisk codes (Table 6). This approach can be further improved by using nanodisk codes with disk pairs of different metal compositions and their functionalization with different type of chromophores.
Fluorescent poly(p-phenylene vinylene)-based barcode nanorods have been developed for individual packet labeling (Li et al. 2010). Invisible nanobarcode tags containing 7400 and 68,000 unique barcodes have been reported (Banu et al. 2006; Birtwell et al. 2008). Fluorescent DNA dendrimer nanobarcodes have been reported for the detection of E. coli, anthrax, Ebola and severe acute respiratory syndrome pathogens in food and biological samples (Li et al. 2005; Lin et al. 2012). So, nanoparticle-based robust nanobarcodes are better than traditional barcodes.
Electronic nose and electronic tongue for artificial smell and taste sensing
Artificial detection of smell and taste of food products with human-like efficiency helps in producing food of desired quality and taste. Nanoparticles have been explored for designing electronic nose and electronic tongue (Table 7).
Electronic nose
Electronic nose is an electronic device derived from aroma detection techniques. It can sense the smell like mammalian olfactory system. Electronic nose mainly consists of gas sensors that senses change in type, quality and quantity of odor/flavor. Nanoparticles help in better absorption of gas on sensor surface due to more surface area than macroscopic particles (Ranjan et al. 2014; Dasgupta et al. 2015). Electronic nose senses the characteristic volatile organic compounds present in food to ensure good quality, uniformity and consistency of raw material during mixing, cooking and of final product during packaging and storage processes (Wilson and Baietto 2009). Detection of ethylene gas level is useful for monitoring the harvesting, storage and processing of the fruits and vegetables. Excess exposure of ethylene gas deteriorates the quality of fruits and vegetables. Tungsten oxide–tin oxide nanocomposites have been employed for ethylene sensing (Pimtong-Ngam et al. 2007). Similarly, nanoparticles have been explored for the sensing of ethanol gas, aromas and other volatile organic compounds (Table 7).
Gold nanoparticles were used to modify an array of quartz crystal microbalance sensors to form electronic nose. So designed nose was used for the detection of extra virgin, virgin and non-edible lampante olive oil (Carlo et al. 2014). Likewise, zinc oxide nanoparticles have been used to scrutinize the quality of 17 commercially available Chinese vinegars (Zhang et al. 2006). Manganese dioxide-, titanium dioxide- and cobalt oxide-doped zinc oxide NPs have been used for the identification of five different types of Chinese alcoholic liquors, namely baiyunbian, Beijing erguotou, red star erguotou, zhijiangdaqu and jianliliangjiu (Zhang et al. 2005). Surface plasmon resonance-based immunosensor has been designed for the detection of characteristic fragrant compound, benzaldehyde in peach products (Gobi et al. 2008).
SWCNT field-effect transistor functionalized with human olfactory receptor 2AG1 protein has been employed for sensing fruit odorant amylbutyrate in apricot (Kim et al. 2009; Jin et al. 2012). Olfactory receptors-functionalized carbon nanotubes-based transistor has been documented for the selective detection of hexanal as olfactory indicator of spoiled milk and oxidized food (Park et al. 2012). SWCNT-based electronic nose has been used for sensing the femtomolar concentration of the seafood spoilage indicator, trimethylamine (Lim et al. 2013). SWCNT-based nanobioelectronic nose has also been used to sense other gaseous odorants selectively up to parts per trillion concentration (Lee et al. 2012a, b).
Electric tongue
Nanobioelectronic tongue sensor for bitter taste detection has been developed by functionalization of carboxylated polypyrrole nanotube field-effect transistor with human taste receptor protein, hTAS2R38. Interestingly, the nanotongue could selectively detect target bitterness compounds, phenylthiocarbamide and propylthiouracil with human-like efficiency (Song et al. 2013). Similarly, human bitter taste receptor protein was immobilized on SWCNT field-effect transistor to form tasters for bitter taste (Table 7). The bioelectronic tongue could discriminate between femtomolar concentration of bitter and non-bitter tastants (Kim et al. 2011). SWCNT field-effect transistor functionalized with nanovesicles containing heterodimeric G-protein-coupled human sweet taste receptors has also been used to develop bioelectronic tongue (Song et al. 2014). Floating electrode-based bioelectronic tongue has been designed for the detection of umami substances. In this study, carbon nanotube field-effect transistor with floating electrodes was hybridized with nanovesicles containing honeybee (Apis mellifera) umami taste receptor, gustatory receptor 10 (Fig. 3). As the umami taste substance, l-monosodium glutamate, binds receptor there is increase in flow of current to electrode. This system was successfully used for the detection of umami taste in chicken soup (Lee et al. 2015).
Image showing the basic design of floating electrode-based bioelectronic tongue for umami taste detection. Carbon nanotubes field-effect transistor with floating electrodes hybridized to nanovesicles containing honeybee umami taste receptor, AmGr10. Figure adapted with permission from (Lee et al. 2015). Copyright (2015) American Chemical Society
The nanoparticle-based electronic nose and electronic tongue can act as substitute for cell-based assays in order to better understand the mechanism of human taste (Song et al. 2013).
Conclusion
Nanosensors ensure fast and effective detection of microorganisms, toxins and adulterants as compared to the existing traditional sensors. Nanoparticles are also very useful for the detection of degradable food ingredients like vitamins and antioxidant materials. Individual pack quality indicator and smart robust packaging materials are some other areas of nanoparticles use. Invisible nanobarcodes protect brands and prevent adulteration. Use of nanoparticles in electronic nose and electronic tongue has lead to artificial sensing of smell and taste with human-like efficiency. So nanoparticles have huge significance in food industry.
References
Actis P, Jejelowo O, Pourmand N (2010) Ultrasensitive mycotoxin detection by STING sensors. Biosens Bioelectron 26:333–337. doi:10.1016/j.bios.2010.08.016
Afonso AS, Perez-Lopez B, Faria RC, Mattoso LHC, Hernandez-Herrero M, Roig-Sagues AX, Costa MM, Merkoci A (2013) Electrochemical detection of Salmonella using gold nanoparticles. Biosens Bioelectron 40:121–126. doi:10.1016/j.bios.2012.06.054
Amagliani G, Brandi G, Omiccioli E, Casiere A, Bruce I, Magnani M (2004) Direct detection of Listeria monocytogenes from milk by magnetic based DNA isolation and PCR. Food Microbiol 21:597–603. doi:10.1016/j.fm.2003.10.008
Ansari AA, Kaushik A, Solanki PR, Malhotra BD (2010) Nanostructured zinc oxide platform for mycotoxin detection. Bioelectrochemistry 77:75–81. doi:10.1016/j.bioelechem.2009.06.014
Bagheri H, Afkhami A, Shirzadmehr A, Khoshsafar H, Khoshsafar H, Ghaedi H (2013) Novel potentiometric sensor for the determination of Cd2Y based on a new nano-composite. Int J Environ Anal Chem 93:578–591. doi:10.1080/03067319.2011.649741
Baik JM, Zielke M, Kim MH, Turner KL, Wodtke AM, Moskovits M (2010) Tin-oxide-nanowire-based electronic nose using heterogeneous catalysis as a functionalization strategy. ACS Nano 4:3117–3122. doi:10.1021/nn100394a
Banholzer MJ, Osberg KD, Li S, Mangelson BF, Schatz GC, Mirkin CA (2010) Silver-based nanodisk codes. ACS Nano 4:5446–5452. doi:10.1021/nn101231u
Banu S, Birtwell S, Chen Y, Galitonov G, Morgan H, Zheludev N (2006) High capacity nano-optical diffraction barcode tagging for biological and chemical applications. Opt Express 14:1382–1387. doi:10.1364/OE.14.001382
Beitollahi H, Ardakania MM, Ganjipour B, Naeimi H (2008) Novel 2,2′-[1,2-ethanediylbis(nitriloethylidyne)]-bis-hydroquinone double-wall carbon nanotube paste electrode for simultaneous determination of epinephrine, uric acid and folic acid. Biosens Bioelectron 24(2008):362–368. doi:10.1016/j.bios.2008.04.009
Birtwell SW, Galitonov GS, Morgan H, Zheludev NI (2008) Superimposed nanostructured diffraction gratings as high capacity barcodes for biological and chemical applications. Opt Commun 281:1789–1795. doi:10.1016/j.optcom.2007.04.066
Borisov SM, Klimant I (2009) Luminescent nanobeads for optical sensing and imaging of dissolved oxygen. Microchim Acta 164:7–15. doi:10.1007/s00604-008-0047-9
Call DR, Borucki MK, Loge FJ (2003) Detection of bacterial pathogens in environmental samples using DNA microarrays. J Microbiol Meth 53:235–243. doi:10.1016/S0167-7012(03)00027-7
Carlo MD, Fusella GC, Pepe A, Sergi M, Martino MD, Mascini M, Martino G, Cichelli A, Natale CD, Compagnone D (2014) Novel oligopeptides based e-nose for food quality control: application to extra-virgin olive samples. Qual Assur Saf Crop 6:307–317. doi:10.3920/QAS2013.0377
Chen S, Ma L, Yuan R, Chai Y, Xiang Y, Wang C (2011) Electrochemical sensor based on Prussian blue nanorods and gold nanochains for the determination of H2O2. Eur Food Res Technol 232:87–95. doi:10.1007/s00217-010-1364-x
Cheng Y, Liu Y, Huang J, Li K, Zhang W, Xian Y, Xin L (2009) Combining biofunctional magnetic nanoparticles and ATP bioluminescence for rapid detection of Escherichia coli. Talanta 77:1332–1336. doi:10.1016/j.talanta.2008.09.014
Chiao DJ, Shyu RH, Hu CS, Chiang HY, Tang SS (2004) Colloidal gold-based immunochromatographic assay for detection of botulinum neurotoxin type B. J Chromatogr B 809:37–41. doi:10.1016/j.jchromb.2004.05.033
Cho CE, Choi JW, Lee M, Koo K (2008) Fabrication of an electrochemical immunosensor with self-assembled peptide nanotubes. Colloid Surf A 313–314:95–99. doi:10.1016/j.colsurfa.2007.04.154
Constantine C, Gattas-Asfura K, Mello S, Crespo G, Rastogi V, Cheng T, DeFrank J, Leblanc R (2003) Layer-by-layer films of chitosan, organophosphorus hydrolase and thioglycolic acid-capped CdSe quantum dots for the detection of paraoxon. J Phys Chem B 107:13762–13764. doi:10.1021/jp036381v
Cubukcu M, Timur S, Anik U (2007) Examination of performance of glassy carbon paste electrode modified with gold nanoparticle and xanthine oxidase for xanthine and hypoxanthine detection. Talanta 74:434–439. doi:10.1016/j.talanta.2007.07.039
Dasgupta N, Ranjan S, Mundekkad D, Ramalingam C, Shanker R, Kumar A (2015) Nanotechnology in agro-food: from field to plate. Food Res Int 69:381–400. doi:10.1016/j.foodres.2015.01.005
Devaramani S, Malingappa P (2012) Synthesis and characterization of cobalt nitroprusside nano particles: application to sulfite sensing in food and water samples. Electrochim Acta 85:579–587. doi:10.1016/j.electacta.2012.08.105
Elyasi M, Khalilzadeh MA, Karimi-Maleh H (2013) High sensitive voltammetric sensor based on Pt/CNTs nanocomposite modified ionic liquid carbon paste electrode for determination of Sudan I in food samples. Food Chem 141:4311–4317. doi:10.1016/j.foodchem.2013.07.020
Ensafi AA, Karimi-Maleh H, Mallakpour S (2012) Simultaneous determination of ascorbic acid, acetaminophen and tryptophan by square wave voltammetry using N-(3,4-dihydroxyphenethyl)-3,5-dinitrobenzamide-modified carbon nanotubes paste electrode. Electroanal 24:666–675. doi:10.1002/elan.201100465
Farahi RH, Passian A, Tetard L, Thundat T (2012) Critical issues in sensor science to aid food and water safety. ACS Nano 6:4548–4556. doi:10.1021/nn204999j
Filippo E, Serra A, Manno D (2009) Poly(vinyl alcohol) capped silver nanoparticles as localized surface plasmon resonance-based hydrogen peroxide sensor. Sens Actuators B-Chem 138:625–630. doi:10.1016/j.snb.2009.02.056
Franz DR, Jahrling PB, Friedlander AM, McClain DJ, Hoover DL, Bryne WR, Avlin JA, Christopher GW, Eitzen EM (1997) Clinical recognition and management of patients exposed to biological warfare agents. J Am Med Assoc 278:399–411. doi:10.1001/jama.1997.03550050061035
Gan T, Li K, Wu K (2008) Multi-wall carbon nanotube-based electrochemical sensor for sensitive determination of Sudan I. Sens Actuators B-Chem 132:134–139. doi:10.1016/j.snb.2008.01.013
Garcia-Aljaro C, Bangar MA, Baldrich E, Munoz FJ, Mulchandani A (2010) Conducting polymer nanowire-based chemiresistive biosensor for the detection of bacterial spores. Biosens Bioelectron 25:2309–2312. doi:10.1016/j.bios.2010.03.021
Gobi KV, Matsumoto K, Toko K, Miura N (2008) Highly regenerable and storageable all-chemical based PEG-immunosensor chip for SPR detection of ppt levels of fragrant compounds from beverage samples. Sens Instrum Food Qual 2:225–233. doi:10.1007/s11694-008-9033-5
Gutierrez-Tauste D, Domenech X, Casan N, Ayllon J (2007) Characterization of methylene blue/TiO2 hybrid thin films prepared by the liquid phase deposition (LPD) method: application for fabrication of light-activated colorimetric oxygen indicators. J Photochem Photobiol A 187:45–52. doi:10.1016/j.jphotochem.2006.09.011
Han C, Li H (2010) Visual detection of melamine in infant formula at 0.1 ppm level based on silver nanoparticles. Analyst 135:583–588. doi:10.1039/b923424a
Huang H, Li L, Zhou G, Liu Z, Ma Q, Feng Y, Zeng G, Tinnefeld P, He Z (2011) Visual detection of melamine in milk samples based on label-free and labeled gold nanoparticles. Talanta 85:1013–1019. doi:10.1016/j.talanta.2011.05.006
Ivanov P, Llobet E, Vilanova X, Brezmes J, Hubalek J, Correig X (2004) Development of high sensitivity ethanol gas sensors based on Pt-doped SnO2 surfaces. Sens Actuators B-Chem 99:201–206. doi:10.1016/j.snb.2003.11.012
Jamali T, Karimi-Maleh H, Khalilzadeh MA (2014) A novel nanosensor based on Pt:Co nanoalloy ionic liquid carbon paste electrode for voltammetric determination of vitamin B9 in food samples. LWT Food Sci Technol 57:679–685. doi:10.1016/j.lwt.2014.01.023
Ji X, Zheng J, Xu J, Rastogi V, Cheng T, DeFrank J, Leblanc R (2005) (CdSe)ZnS quantum dots and organophosphorus hydrolase bioconjugate as biosensors for detection of paraoxon. J Phys Chem B 109:3793–3799. doi:10.1021/jp044928f
Jiang X, Valdeperez D, Nazarenus M, Wang Z, Stellacci F, Parak WJ, Pino PD (2015) Future perspectives towards the use of nanomaterials for smart food packaging and quality control. Part Part Syst Charact 32:408–416. doi:10.1002/ppsc.201400192
Jin HJ, Lee SH, Kim TH, Park J, Song HS, Park TH, Hong S (2012) Nanovesicle-based bioelectronic nose platform mimicking human olfactory signal transduction. Biosens Bioelectron 35:335–341. doi:10.1016/j.bios.2012.03.012
Joung HA, Lee NR, Lee SK, Ahn J, Shin YB, Choi HS, Lee CS, Kim S, Kim MG (2008) High sensitivity detection of 16s rRNA using peptide nucleic acid probes and a surface plasmon resonance biosensor. Anal Chim Acta 630:168–173. doi:10.1016/j.aca.2008.10.001
Kara M, Uzun L, Kolayli S, Denizli A (2013) Combining molecular imprinted nanoparticles with surface plasmon resonance nanosensor for chloramphenicol detection in honey. J Appl Polym Sci 129:2273–2279. doi:10.1002/APP.38936
Karimi-Maleh H, Moazampour M, Yoosefian M, Sanati AL, Javazmi FT, Mahani M (2014) An electrochemical nanosensor for simultaneous voltammetric determination of ascorbic acid and Sudan I in food samples. Food Anal Methods 7:2169–2176. doi:10.1007/s12161-014-9867-x
Kaushik A, Solanki PR, Ansari AA, Ahmad S, Malhotra BD (2009) A nanostructured cerium oxide film-based immunosensor for mycotoxin detection. Nanotechnology 20:55105. doi:10.1088/0957-4484/20/5/055105
Kim TH, Lee SH, Lee J, Song HS, Oh EH, Park HT, Hong S (2009) Single-carbon-atomic-resolution detection of odorant molecules using a human olfactory receptor-based bioelectronic nose. Adv Mater 21:91–94. doi:10.1002/adma.200801435
Kim TH, Song HS, Jin HJ, Lee SH, Namgung S, Kim UK, Park TH, Hong S (2011) Bioelectronic super-taster device based on taste receptor-carbon nanotube hybrid structures. Lab Chip 11:2262–2267. doi:10.1039/c0lc00648c
Ko W, Jung N, Lee M, Yun M, Jeon S (2013) Electronic nose based on multipatterns of ZnO nanorods on a quartz resonator with remote electrodes. ACS Nano 7:6685–6690. doi:10.1021/nn4027245
Kuang H, Chen W, Yan W, Xu L, Zhu Y, Liu L, Chu H, Peng C, Wang L, Kotov NA, Xu C (2011) Crown ether assembly of gold nanoparticles: melamine sensor. Biosens Bioelectron 26:2032–2037. doi:10.1016/j.bios.2010.08.081
Kumar V, Guleria P, Mehta SK (2016) Nanoparticles to sense food quality. In: Ranjan S, Dasgupta N, Lichtfouse E (eds) Nanoscience in food and agriculture 3. Springer, Berlin, pp 145–182. doi:10.1007/978-3-319-48009-1_6
Kwak CH, Woo HS, Hady FA, Wazzan AA, Lee JH (2016) Vapor-phase growth of urchin-like Mg-doped ZnO nanowire networks and their application to highly sensitive and selective detection of ethanol. Sens Actuators B-Chem 223:527–534. doi:10.1016/j.snb.2015.09.120
Lee SH, Jin HJ, Song HS, Hong S, Parka TH (2012a) Bioelectronic nose with high sensitivity and selectivity using chemically functionalized carbon nanotube combined with human olfactory receptor. J Biotechnol 157:467–472. doi:10.1016/j.jbiotec.2011.09.011
Lee SH, Kwon OS, Song HS, Park SJ, Sung JH, Jang J, Park TH (2012b) Mimicking the human smell sensing mechanism with an artificial nose platform. Biomaterials 33:1722–1729. doi:10.1016/j.biomaterials.2011.11.044
Lee M, Jung JW, Kim D, Ahn YJ, Hong S, Kwon HW (2015) Discrimination of umami tastants using floating electrode-based bioelectronic tongue mimicking insect taste systems. ACS Nano 9:11728–11736. doi:10.1021/acsnano.5b03031
Leggiadro RJ (2000) The threat of biological terrorism: a public health and infection control reality. Infect Control Hosp Epidemiol 21:53–56. doi:10.1086/501700
Li Y, Cu HYT, Luo D (2005) Multiplexed detection of pathogen DNA with DNA-based fluorescence nanobarcodes. Nat Biotechnol 23:885–889. doi:10.1038/nbt110
Li JW, Yu JJ, Zhao FQ, Zeng BZ (2007) Direct electrochemistry of glucose oxidase entrapped in nanogold particles–ionic liquid-N,N-dimethylformamide composite film on glassy carbon electrode and glucose sensing. Anal Chim Acta 587:33–40. doi:10.1016/j.aca.2007.01.014
Li X, Wang T, Zhang J, Zhu D, Zhang X, Ning Y, Zhang H, Yang B (2010) Controlled fabrication of fluorescent barcode nanorods. ACS Nano 4:4350–4360. doi:10.1021/nn9017137
Li L, Liu M, He S, Chen W (2014) Freestanding 3D mesoporous Co3O4@carbon foam nanostructures for ethanol gas sensing. Anal Chem 86:7996–8002. doi:10.1021/ac5021613
Liang KZ, Mu WZ (2008) ZrO2/DNA-derivated polyion hybrid complex membrane for the determination of hydrogen peroxide in milk. Ionics 14:533–539. doi:10.1007/s11581-008-0213-4
Lim JH, Park J, Ahn JH, Jin HJ, Hong S, Park TH (2013) A peptide receptor-based bioelectronic nose for the real-time determination of seafood quality. Biosens Bioelectron 39:244–249. doi:10.1016/j.bios.2012.07.054
Lin C, Jungmann R, Leifer AM, Li C, Levner D, Church GM, Shih WM, Yin P (2012) Submicrometre geometrically encoded fluorescent barcodes self-assembled from DNA. Nat Chem 4:832–839. doi:10.1038/nchem.1451
Liu S, Yu J, Ju H (2003) Renewable phenol biosensor based on a tyrosinase-colloidal gold modified carbon paste electrode. J Electroanal Chem 540:61–67. doi:10.1016/S0022-0728(02)01276-7
Liu Y, Yao D, Chang H, Liu C, Chen C (2008) Magnetic bead based DNA detection with multi-layers quantum dots labeling for rapid detection of Escherichia coli O157:H7. Biosens Bioelectron 24:558–565. doi:10.1016/j.bios.2008.06.019
Liu H, Wen M, Zhang F, Liu D, Tian Y (2010) Electrochemical sensing platform for hydrogen peroxide using amorphous FeNiPt nanostructures. Anal Methods 2:143–148. doi:10.1039/b9ay00209j
Liu D, Chen W, Wei J, Li X, Wang Z, Jiang X (2012) A highly sensitive, dual-readout assay based on gold nanoparticles for organophosphorus and carbamate pesticides. Anal Chem 84:4185–4191. doi:10.1021/ac300545p
Liusman C, Li H, Lu G, Wu J, Boey F, Li S, Zhang H (2012) Surface-enhanced Raman scattering of Ag–Au nanodisk heterodimers. J Phys Chem C 116:10390–10395. doi:10.1021/jp301739a
Lou Z, Li F, Deng J, Wang LL, Zhang T (2013) Branch-like hierarchical heterostructure (α-Fe2O3/TiO2): a novel sensing material for trimethylamine gas sensor. ACS Appl Mater Interfaces 5:12310–12316. doi:10.1021/am402532v
Lu H, Yu S, Fan Y, Yang C, Xu D (2013) Nonenzymatic hydrogen peroxide electrochemical sensor based on carbon-coated SnO2 supported Pt nanoparticles. Colloid Surf B 101:106–110. doi:10.1166/sl.2015.3414
Ma X, Chao M, Wang Z (2013) Electrochemical determination of Sudan I in food samples at graphene modified glassy carbon electrode based on the enhancement effect of sodium dodecyl sulphonate. Food Chem 138:739–744. doi:10.1016/j.foodchem.2012.11.004
Majidi MR, Baj RFB, Naseri A (2013) Carbon nanotube–ionic liquid (CNT–IL) nanocomposite modified sol–gel derived carbon–ceramic electrode for simultaneous determination of sunset yellow and tartrazine in food samples. Food Anal Methods 6:1388–1397. doi:10.1007/s12161-012-9556-6
Mak AC, Osterfeld SJ, Yu H, Wang SX, Davis RW, Jejelowo OA, Pourmand N (2010) Sensitive giant magnetoresistive-based immunoassay for multiplex mycotoxin detection. Biosens Bioelectron 25:1635–1639. doi:10.1016/j.bios.2009.11.028
Najafi M, Khalilzadeh MA, Karimi-Maleh H (2014) A new strategy for determination of bisphenol A in the presence of Sudan I using a ZnO/CNTs/ionic liquid paste electrode in food samples. Food Chem 158:125–131. doi:10.1016/j.foodchem.2014.02.082
Nopwinyuwong A, Kaisone T, Hanthanon P, Nandhivajrin C, Boonsupthip W, Pechyen C, Suppakul P (2014) Effects of nanoparticle concentration and plasticizer type on colorimetric behavior of polydiacetylene/silica nanocomposite as time–temperature indicator. Energy Proc 56:423–430. doi:10.1016/j.egypro.2014.07.175
Norouzi P, Larijani B, Ganjali MR (2012) Ochratoxin A sensor based on nanocomposite hybrid film of ionic liquid-graphene nano-sheets using coulometric FFT cyclic voltammetry. Int J Electrochem Sci 7:7313–7324
Pan J, Ganesan R, Shen H, Mathur S (2010) Plasma-modified SnO2 nanowires for enhanced gas sensing. J Phys Chem C 114:8245–8250. doi:10.1021/jp101072f
Park J, Lim JH, Jin HJ, Namgung S, Lee SH, Park TH, Hong S (2012) A bioelectronic sensor based on canine olfactory nanovesicle-carbon nanotube hybrid structures for the fast assessment of food quality. Analyst 137:3249–3254. doi:10.1039/c2an16274a
Pimtong-Ngam Y, Jiemsirilers S, Supothina S (2007) Preparation of tungsten oxide–tin oxide nanocomposites and their ethylene sensing characteristics. Sensor Actuators A-Phys 139:7–11. doi:10.1016/j.sna.2006.10.032
Ping H, Zhang M, Li H, Li S, Chen Q, Sun C, Zhang T (2012) Visual detection of melamine in raw milk by label-free silver nanoparticles. Food Control 23:191–197. doi:10.1016/j.foodcont.2011.07.009
Qin L, Zou S, Xue C, Atkinson A, Schatz GC, Mirkin CA (2006) Designing, fabricating and imaging Raman hot spots. Proc Natl Acad Sci USA 103:13300–13303. doi:10.1073/pnas.0605889103
Qin L, Banholzer MJ, Millstone JE, Mirkin CA (2007) Nanodisk codes. Nano Lett 7:3849–3853. doi:10.1021/nl072606s
Radoi A, Targa M, Simon BP, Marty JL (2008) Enzyme-linked immunosorbent assay (ELISA) based on superparamagnetic nanoparticles for aflatoxin M1 detection. Talanta 77:138–143. doi:10.1016/j.talanta.2008.05.048
Ranjan S, Dasgupta N, Chakraborty AR, Samuel SM, Ramalingam C, Shanker R, Kumar A (2014) Nanoscience and nanotechnologies in food industries: opportunities and research trends. J Nanopart Res 16:2464. doi:10.1007/s11051-014-2464-5
Ravindranath SP, Mauer LJ, Roy CD, Irudayaraj J (2009) Biofunctionalized magnetic nanoparticle integrated mid-infrared pathogen sensor for food matrixes. Anal Chem 81:2840–2846. doi:10.1021/ac802158y
Realini CE, Marcos B (2014) Active and intelligent packaging systems for a modern society. Meat Sci 98:404–419. doi:10.1016/j.meatsci.2014.06.031
Sanchez-Acevedo ZC, Riu J, Rius FX (2009) Fast picomolar selective detection of bisphenol A in water using a carbon nanotube field effect transistor functionalized with estrogen receptor-α. Biosens Bioelectron 24:2842–2846. doi:10.1016/j.bios.2009.02.019
Sanz VC, Mena ML, Gonazalez AC, Sedeno PY, Pingarron JM (2005) Development of tyrosinase biosensor based on gold nanoparticles-modified glassy carbon electrodes application to the measurement of a bioelectrochemical polyphenols index in wines. Anal Chim Acta 528:1–8. doi:10.1016/j.aca.2004.10.007
Sberveglieri G, Concina I, Comini E, Falasconi M, Ferroni M, Sberveglieri V (2012) Synthesis and integration of tin oxide nanowires into an electronic nose. Vacuum 86:532–535. doi:10.1016/j.vacuum.2011.10.004
Scandurra G, Arena A, Ciofi C, Saitta G (2013) Electrical characterization and hydrogen peroxide sensing properties of gold/nafion: polypyrrole/MWCNTs electrochemical devices. Sensors 13:3878–3888. doi:10.3390/s130303878
Sharma A, Matharu Z, Sumana G, Solanki PR, Kim CG, Malhotra BD (2010) Antibody immobilized cysteamine functionalized-gold nanoparticles for aflatoxin detection. Thin Solid Film 519:1213–1218. doi:10.1016/j.tsf.2010.08.071
Song HS, Kwon OS, Lee SH, Park SJ, Kim UK, Jang J, Park TH (2013) Human taste receptor-functionalized field effect transistor as a human-like nanobioelectronic tongue. Nano Lett 13:172–178. doi:10.1021/nl3038147
Song HS, Jin HJ, Ahn SR, Kim D, Lee SH, Kim UK, Simons CT, Hong S, Park TH (2014) Bioelectronic tongue using heterodimeric human taste receptor for the discrimination of sweeteners with human-like performance. ACS Nano 8:9781–9789. doi:10.1021/nn502926x
Stoycheva T, Vallejos S, Blackman C, Moniz SJA, Calderer J, Correig X (2012) Important considerations for effective gas sensors based on metal oxide nanoneedles films. Sens Actuators B-Chem 161:406–413. doi:10.1016/j.snb.2011.10.052
Su X, Li Y (2004) Quantum dot biolabeling coupled with immunomagnetic separation for detection of Escherichia coli O157:H7. Anal Chem 76:4806–4810. doi:10.1021/ac049442
Su H, Fan H, Ai S, Wu N, Fan H, Bian P, Liu J (2011) Selective determination of melamine in milk samples using 3-mercapto-1-propanesulfonate-modified gold nanoparticles as colorimetric probe. Talanta 85:1338–1343. doi:10.1016/j.talanta.2011.06.017
Sysoev VV, Strelcov E, Sommer M, Bruns M, Kiselev I, Habicht W, Kar S, Gregoratti L, Kiskinova M, Kolmakov A (2010) Single-nanobelt electronic nose: engineering and tests of the simplest analytical element. ACS Nano 4:4487–4494. doi:10.1021/nn100435h
Tang H, Yan M, Ma X, Zhang H, Wang M, Yang D (2006) Gas sensing behavior of polyvinylpyrrolidone-modified ZnO nanoparticles for trimethylamine. Sens Actuators B-Chem 113:324–328. doi:10.1016/j.snb.2005.03.024
Terry LA, White SF, Tigwell LJ (2005) The application of biosensors to fresh produce and the wider food industry. J Agric Food Chem 53:1309–1316. doi:10.1021/jf040319t
Vamvakaki V, Chaniotakis N (2007) Pesticide detection with a liposome-based nano-biosensor. Biosens Bioelectron 22:2848–2853. doi:10.1016/j.bios.2006.11.024
Varshney M, Li Y, Srinivasan B, Tung S (2007) A label-free, microfluidics and interdigitated array microelectrode-based impedance biosensor in combination with nanoparticles immunoseparation for detection of Escherichia coli O157:H7 in food samples. Sens Actuators B-Chem 128:99–107. doi:10.1016/j.snb.2007.03.045
Vasimalai N, John SA (2013) Picomolar melamine enhanced the fluorescence of gold nanoparticles: spectrofluorimetric determination of melamine in milk and infant formulas using functionalized triazole capped gold nanoparticles. Biosens Bioelectron 42:267–272. doi:10.1016/j.bios.2012.10.023
Vermeir S, Nicolai BM, Jans K, Maes G, Lammertyn J (2007) High-throughput microplate enzymatic assays for fast sugar and acid quantification in apple and tomato. J Agric Food Chem 55:3240–3248. doi:10.1021/jf0637022
Vermeiren L, Devlieghere F, Beest M, Kruijf N, Debevere J (1999) Developments in the active packaging of foods. Trends Food Sci Technol 10:77–86. doi:10.1016/S0924-2244(99)00032-1
Verstrepen KJ, Iserentant D, Malcorps P, Derdelinckx G, Dijck PV, Winderickx J, Pretorius IS, Thevelein JM, Delvaux FR (2004) Glucose and sucrose: hazardous fast-food for industrial yeast? Trends Biotechnol 22:531–537. doi:10.1016/j.tibtech.2004.08.001
Villamizar RA, Maroto A, Rius FX, Inza I, Figueras MJ (2008) Fast detection of Salmonella Infantis with carbon nanotube field effect transistors. Biosens Bioelectron 24:279–283. doi:10.1016/j.bios.2008.03.046
Wang M, Li Z (2008) Nano-composite ZrO2/Au film electrode for voltammetric detection of parathion. Sens Actuators B-Chem 133:607–612. doi:10.1016/j.snb.2008.03.023
Wang SG, Zhang Q, Wang R, Yoon SF (2003) A novel multi-walled carbon nanotube-based biosensor for glucose detection. Biochem Biophys Res Commun 311:572–576. doi:10.1016/j.bbrc.2003.10.031
Wang Y, Wei W, Zeng J, Liu X, Zeng X (2008) Fabrication of a copper nanoparticle/chitosan/carbon nanotube-modified glassy carbon electrode for electrochemical sensing of hydrogen peroxide and glucose. Microchim Acta 160:253–260. doi:10.1007/s00604-007-0844-6
Wang L, Chen W, Xu D, Shim BS, Zhu Y, Sun F, Liu L, Peng C, Jin Z, Xu C, Kotov NA (2009) Simple, rapid, sensitive and versatile SWNT-paper sensor for environmental toxin detection competitive with ELISA. Nano Lett 9:4147–4152. doi:10.1021/nl902368r
Wang JJ, Liu BH, Hsu YT, Yu FY (2011) Sensitive competitive direct enzyme-linked immunosorbent assay and gold nanoparticle immunochromatographic strip for detecting aflatoxin M1 in milk. Food Control 22:964–969. doi:10.1016/j.foodcont.2010.12.003
Wang L, Gao P, Bao D, Wang Y, Chen Y, Chang C, Li G, Yang P (2014) Synthesis of crystalline/amorphous core/shell MoO3 composites through a controlled dehydration route and their enhanced ethanol sensing properties. Cryst Growth Des 14:569–575. doi:10.1021/cg401384t
Wang M, Duong B, Fenniria H, Su M (2015a) Nanomaterial-based barcodes. Nanoscale 7:11240–11247. doi:10.1039/C5NR01948F
Wang YC, Lu L, Gunasekaran S (2015b) Gold nanoparticle-based thermal history indicator for monitoring low-temperature storage. Microchim Acta 182:1305–1311. doi:10.1007/s00604-015-1451-6
Wei SH, Zhao FQ, Xu ZY, Zeng BZ (2006) Voltammetric determination of folic acid with a multi-walled carbon nanotube-modified gold electrode. Microchim Acta 152:285–290. doi:10.1007/s00604-005-0437-1
Welch CM, Banks CE, Simm AO, Compton RG (2005) Silver nanoparticle assemblies supported on glassy-carbon electrodes for the electron-analytical detection of hydrogen peroxide. Anal Bioanal Chem 382:12–21. doi:10.1007/s00216-005-3205-5
Wilson AD, Baietto M (2009) Applications and advances in electronic-nose technologies. Sensors 9:5099–5148. doi:10.1088/0031-8949/85/03/035701
Wu M, Tang W, Gu J, Wang Q, He P, Fang Y (2013a) Electrochemical detection of Sudan I using a multi-walled carbon nanotube/chitosan composite modified glassy carbon electrode. Am J Anal Chem 4:1–6. doi:10.4236/ajac.2013.46A001
Wu RJ, Lin DJ, Yu MR, Chen MH, Lai HF (2013b) Ag@SnO2 core–shell material for use in fast-response ethanol sensor at room operating temperature. Sens Actuators B-Chem 178:185–191. doi:10.1016/j.snb.2012.12.052
Xiao F, Ruan C, Liu L, Yan R, Zhao F, Zeng B (2008) Single-walled carbon nanotube–ionic liquid paste electrode for the sensitive voltammetric determination of folic acid. Sens Actuators B-Chem 134:895–901. doi:10.1016/j.snb.2008.06.037
Xiulan S, Xiaolian Z, Jian T, Zhou J, Chu FS (2005) Preparation of gold-labeled antibody probe and its use in immunochromatography assay for detection of aflatoxin B1. Int J Food Microbiol 99:185–194. doi:10.1016/j.ijfoodmicro.2004.07.021
Xu S, Lu H (2015) One-pot synthesis of mesoporous structured ratiometric fluorescence molecularly imprinted sensor for highly sensitive detection of melamine from milk samples. Biosens Bioelectron 73:160–166. doi:10.1016/j.bios.2015.05.064
Yan Z, Zhao J, Qin L, Mu F, Wang P, Feng X (2013) Non-enzymatic hydrogen peroxide sensor based on a gold electrode modified with granular cuprous oxide nanowires. Microchim Acta 180:145–150. doi:10.1007/s00604-012-0916-0
Yang L, Li Y (2006) Simultaneous detection of Escherichia coli O157:H7 and Salmonella typhimurium using quantum dots as fluorescence labels. Analyst 131:394–401. doi:10.1039/B510888H
Yang H, Qu L, Wimbrow A, Jiang X, Sun Y (2007) Rapid detection of Listeria monocytogenes by nanoparticle-based immunomagnetic separation and real-time PCR. Int J Food Microbiol 118:132–138. doi:10.1016/j.ijfoodmicro.2007.06.019
Yang D, Zhu L, Jiang X (2010) Electrochemical reaction mechanism and determination of Sudan I at a multi wall carbon nanotubes modified glassy carbon electrode. J Electroanal Chem 640:17–22. doi:10.1016/j.jelechem.2009.12.022
Ye JS, Wen Y, Zhang WD, Gan LM, Xu GQ, Sheu FS (2004) Nonenzymatic glucose detection using multi-walled carbon nanotube electrodes. Electrochem Commun 6:66–70. doi:10.1016/j.elecom.2003.10.013
Yemini M, Reches M, Gazit E, Rishpon J (2005) Peptide nanotube-modified electrodes for enzyme-biosensor applications. Anal Chem 77:5155–5159. doi:10.1021/ac050414g
Yin H, Zhou Y, Meng X, Tang T, Aia S, Zhu L (2011) Electrochemical behaviour of Sudan I at Fe3O4 nanoparticles modified glassy carbon electrode and its determination in food samples. Food Chem 127:1348–1353. doi:10.1016/j.foodchem.2011.01.097
Zamolo VA, Valenti G, Venturelli E, Chaloin O, Marcaccio M, Boscolo S, Castagnola V, Sosa S, Berti F, Fontanive G, Poli M, Tubaro A, Bianco A, Paolucci F, Prato M (2012) Highly sensitive electrochemiluminescent nanobiosensor for the detection of palytoxin. ACS Nano 6:7989–7997. doi:10.1021/nn302573c
Zeng J, Roberts S, Xia Y (2010) Nanocrystal-based time–temperature indicators. Chem Eur J 16:12559–12563. doi:10.1002/chem.201002665
Zhang WH, Zhang WD (2008) Fabrication of SnO2–ZnO nanocomposite sensor for selective sensing of trimethylamine and the freshness of fishes. Sens Actuators B-Chem 134:403–408. doi:10.1016/j.snb.2008.05.015
Zhang Q, Xie C, Zhang S, Wang A, Zhu B, Wang L, Yang Z (2005) Identification and pattern recognition analysis of Chinese liquors by doped nano ZnO gas sensor array. Sens Actuators B-Chem 110:370–376. doi:10.1016/j.snb.2005.02.017
Zhang Q, Zhang S, Xie C, Zeng D, Fan C, Li D, Bai Z (2006) Characterization of Chinese vinegars by electronic nose. Sens Actuators B-Chem 119:538–546. doi:10.1016/j.snb.2006.01.007
Zhang W, Tang H, Geng P, Wang Q, Jin L, Wu Z (2007) Amperometric method for rapid detection of Escherichia coli by flow injection analysis using a bismuth nano-film modified glassy carbon electrode. Electrochem Commun 9:833–838. doi:10.1016/j.elecom.2006.11.019
Zhang C, Yin AX, Jiang R, Rong J, Dong L, Zhao T, Sun LD, Wang J, Chen X, Yan CH (2013a) Time temperature indicator for perishable products based on kinetically programmable Ag overgrowth on Au nanorods. ACS Nano 7:4561–4568. doi:10.1021/nn401266u
Zhang Z, Lin M, Zhang S, Vardhanabhuti B (2013b) Detection of aflatoxin M1 in milk by dynamic light scattering coupled with superparamagnetic beads and gold nanoprobes. J Agric Food Chem 61:4520–4525. doi:10.1021/jf400043z
Zhao X, Hilliard LR, Mechery SJ, Wang Y, Bagwe RP, Jin S, Tan W (2004) A rapid bioassay for single bacterial cell quantitation using bioconjugated nanoparticles. Proc Natl Acad Sci USA 101:15027–15032. doi:10.1073/pnas.0404806101
Zhao G, Xing F, Deng S (2007) A disposable amperometric enzyme immunosensor for rapid detection of Vibrio parahaemolyticus in food based on agarose/nano-Au membrane and screen-printed electrode. Electrochem Commun 9:1263–1268. doi:10.1016/j.elecom.2007.01.036
Zhou Y, Pan FG, Li YS, Zhang YY, Zhang JH, Lu SY, Ren HL, Liu ZS (2009) Colloidal gold probe-based immunochromatographic assay for the rapid detection of brevetoxins in fishery product samples. Biosens Bioelectron 24:2744–2747. doi:10.1016/j.bios.2009.01.034
Zhou Z, Li T, Huang H, Chen Y, Liu F, Huang C, Li N (2014) A dual amplification strategy for DNA detection combining bio-barcode assay and metal-enhanced fluorescence modality. Chem Commun 50:13373–13376. doi:10.1039/c4cc05554c
Zhu S, Dua C, Fu Y (2009) Localized surface plasmon resonance-based hybrid Au–Ag nanoparticles for detection of Staphylococcus aureus enterotoxin B. Opt Mater 31:1608–1613. doi:10.1016/j.optmat.2009.03.009
Acknowledgements
V. K. would like to thank University Grant Commission-GOI for fellowship as UGC-DSK Postdoctoral fellowship. V. K. and P. G. are thankful to vice-chancellor and chancellor DAV University Jalandhar for encouragement and support.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Kumar, V., Guleria, P. & Mehta, S.K. Nanosensors for food quality and safety assessment. Environ Chem Lett 15, 165–177 (2017). https://doi.org/10.1007/s10311-017-0616-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10311-017-0616-4