Abstract
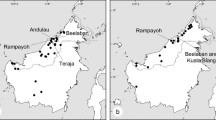
Several barriers contribute to reproductive isolation between plant species, which can be classified as pre- or post-pollination. Understanding the strength of these barriers could clarify the factors that maintain reproductive isolation and thus species integrity. In this study, we quantified reproductive isolation between two bat-pollinated co-occurring Bauhinia species (B. acuruana and B. pentandra) with similar flower morphology. Over the course of 18 months, we assessed reproductive isolation between these two Bauhinia species by quantifying the individual strengths and absolute contributions of five pre- and post- pollination barriers. Our data showed that both species are completely isolated in their reproduction by a combination of several barriers. Although they co-occur in a few populations, we found a high degree of geographic isolation between them. And although their flowering periods overlap, there is a significant difference in flowering peaks. Both species have the same pollinating bats, but the interspecific transfer of pollen between the plant species may be reduced due to the different length of the flower stamens, resulting in different pollen deposition on the bats’ bodies. We have documented complete incompatibility between taxa and conclude that pre- and post-pollination barriers are important factors in preventing gene flow, even in contact zones between these two species of Bauhinia. We highlight that our work is the first study to use methods to estimate the strength of reproductive isolation barriers between bat-pollinated species.





Similar content being viewed by others
References
Albuquerque-Lima S, Taylor NP, Zappi DC, Machado IC (2023) Floral specialization and bat pollination in subtribe Cereinae (Cactaceae): a morphological approach. Diversity 15:1–8. https://doi.org/10.3390/d15020207
Andrade-Lima D (1981) The Caatinga dominium. Rev Bras Bot 4:149–153
Athiê-Souza SM, Melo JIMD, Silva LP, Santos LL, Santos JS, Oliveira LSD, Sales MF (2019) Phanerogamic flora of the Catimbau National Park, Pernambuco, Brazil. Biota Neotrop 19:1–27. https://doi.org/10.1590/1676-0611-bn-2018-0622
Baack E, Melo MC, Rieseberg LH, Ortiz-Barrientos D (2015) The origins of reproductive isolation in plants. New Phytol 207:968–984. https://doi.org/10.1111/nph.13424
Barônio GJ, Maciel AA, Oliveira AC, Kobal RO, Meireles DA, Brito VL, Rech AR (2016) Plantas, polinizadores e algumas articulações da biologia da polinização com a teoria ecológica. Rodriguésia 67:275–293. https://doi.org/10.1590/2175-7860201667201
Bergallo HG (1990) Floral biology and pollination of Bauhinia bongardii Steud in Serra Dos Carajás, Pará. Braz J Bot 50:401–405
Christenhusz MJM, Fay MF, Chase MW (2017) Plants of the world: an illustrated encyclopedia of vascular plants. Kew Publishing, Royal Botanic Gardens, Kew; The University of Chicago Press, Richmond, Surrey: Chicago
Cordero-Schmidt E, Maruyama PK, Vargas‐Mena JC, Oliveira PP, Santos FAR, Medellín RA, Rodriguez-Herrera R, Venticinque EM (2021) Bat–flower interaction networks in Caatinga reveal generalized associations and temporal stability. Biotropica 53:1546–1557. https://doi.org/10.1111/btp.13007
Coughlan JM, Matute DR (2020) The importance of intrinsic postzygotic barriers throughout the speciation process. Phil Trans R Soc B 375:20190533. https://doi.org/10.1098/rstb.2019.0533
Coyne JA, Orr HA (2004) Speciation. Sinauer Associates, Sunderland
Cuevas E, Espino J, Marques I (2018) Reproductive isolation between Salvia elegans and S. fulgens, two hummingbird-pollinated sympatric sages. Plant Biol 20:1075–1082. https://doi.org/10.1111/plb.12874
Silva JMC, Leal IR, Tabarelli M (2017) Caatinga: the largest tropical dry forest region in South America. Springer
Dobat K, Peikert-Holle T (1985) Blüten und fledermäuse: bestäubung durch fledermäuse und flughunde (Chiropterophilie), 1St edn. Verlag Waldemar Kramer, Frankfurt am Main
Domingos-Melo A, Albuquerque-Lima S, Diniz UM, Lopes AV, Machado IC (2023) Bat pollination in the Caatinga: a review of studies and peculiarities of the system in the New World’s largest and most diverse seasonally dry Tropical Forest. https://doi.org/10.1016/j.flora.2023.152332. Flora 152332
Endress PK (1994) Diversity and evolutionary biology of tropical flowers. Cambridge University Press, Cambridge
Fægri K, van der Pijl L (1979) The principles of pollination ecology. Pergamon Press, Oxford
Fernandes MF, Cardoso D, Queiroz LP (2020) An updated plant checklist of the Brazilian Caatinga seasonally dry forests and woodlands reveals high species richness and endemism. J Arid Environ 174:104079. https://doi.org/10.1016/j.jaridenv.2019.104079
Fischer EA (1992) Foraging of nectarivorous bats on Bauhinia ungulata. Biotropica 24:579–582. https://doi.org/10.2307/2389025
Fischer E, Leal IR (2006) Effect of nectar secretion rate on pollination success of Passiflora coccinea (Passifloraceae) in the Central Amazon. Braz J Biol 66:747–754. https://doi.org/10.1590/S1519-69842006000400019
Fleming TH, Geiselman C, Kress WJ (2009) The evolution of bat pollination: a phylogenetic perspective. Ann Bot 104:1017–1043. https://doi.org/10.1093/aob/mcp197
Galetto L, Bernardello G (2005) Rewards in flowers–nectar. In: Dafni A, Kevan PG, Husband BC (eds) Practical Pollination Biology. Enviroquest, Ltd. Cambridge, Ontario, pp 261–313
Ghazoul J (2006) Floral diversity and the facilitation of pollination. J Ecol 94:295–304. https://doi.org/10.1111/j.1365-2745.2006.01098.x
Grant V (1994) Modes and origins of mechanical and ethological isolation in angiosperms. Proc Natl Acad Sci 91:3–10. https://doi.org/10.1073/pnas.91.1.3
Grant KA, Grant V (1964) Mechanical isolation of Salvia apiana and Salvia mellifera. (Labiatae) Evol 18:196. https://doi.org/10.2307/2406392
Gribel R, Gibbs PE, Queiróz AL (1999) Flowering phenology and pollination biology of Ceiba pentandra (Bombacaceae) in Central Amazonia. J Trop Ecol 15:247–263. https://doi.org/10.1017/S0266467499000796
Grossenbacher DL, Veloz SD, Sexton JP (2014) Niche and range size patterns suggest that speciation begins in small, ecologically diverged populations in north American monkeyflowers (Mimulus spp.): niche and range size patterns. Evolution 68:1270–1280. https://doi.org/10.1111/evo.12355
Heithaus ER, Opler PA, Baker HG (1974) Bat activity and pollination of Bauhinia pauletia: plant-pollinator coevolution. Ecology 55:412–419. https://doi.org/10.2307/1935229
Hokche O, Ramirez N (1990) Pollination ecology of seven species of Bauhinia L. (Leguminosae: Caesalpinioideae). Ann Mo Bot Garden 77:559. https://doi.org/10.2307/2399520
Horner MA, Fleming TH, Sahey CT (1998) Foraging behaviour and energetics of a nectar-feeding bat, Leptonycteris curasoae (Chiroptera: Phyllostomidae). J Zool 244:575–586. https://doi.org/10.1111/j.1469-7998.1998.tb00062.x
Kay KM (2006) Reproductive isolation between two closely related hummingbird pollinated neotropical gingers. Evolution 60:538–552. https://doi.org/10.1111/j.0014-3820.2006.tb01135.x
Kay KM, Sargent RD (2009) The role of animal pollination in plant speciation: integrating Ecology, Geography, and Genetics. Annu Rev Ecol Evol Syst 40:637–656. https://doi.org/10.1146/annurev.ecolsys.110308.120310
Lemke TO (1984) Foraging ecology of the long-nosed bat, Glossophaga Soricina, with respect to resource availability. Ecology 65:538–548. https://doi.org/10.2307/1941416
Lowry DB, Modliszewski JL, Wright KM, Wu CA, Willis JH (2008) The strength and genetic basis of reproductive isolating barriers in flowering plants. Phil Trans R Soc B 363:3009–3021. https://doi.org/10.1098/rstb.2008.0064
LPWG (2017) A new subfamily classification of the Leguminosae based on a taxonomically comprehensive phylogeny. Taxon 66:44–77. https://doi.org/10.12705/661.3
Machado IC, Lopes AV (2004) Floral traits and pollination systems in the Caatinga, a Brazilian tropical dry forest. Ann Bot 94:365–376. https://doi.org/10.1093/aob/mch152
Machado IC, Vogel S (2004) The North-East-brazilian liana, Adenocalymna dichilum (Bignoniaceae) pollinated by bats. Ann Bot 93:609–613. https://doi.org/10.1093/aob/mch069
Machado IC, Lopes AV, Leite AV, Neves CB (2006) Cleome spinosa (Capparaceae): polygamodioecy and pollination by bats in urban and caatinga areas, northeastern Brazil. Botanis Jahr 127:69–82. https://doi.org/10.1127/0006-8152/2006/0127-0069
Marques I, Rossello-Graell A, Draper D, Iriondo JM (2007) Pollination patterns limit hybridization between two sympatric species of Narcissus (Amaryllidaceae). Am J Bot 94:1352–1359. https://doi.org/10.3732/ajb.94.8.1352
Martin NH, Willis JH (2007) Ecological divergence associated with mating system causes nearly complete reproductive isolation between sympatric Mimulus species. Evolution 61:68–82. https://doi.org/10.1111/j.1558-5646.2007.00006.x
Mayr E (1982) Systematics and the origin of species. Columbia University Press, New York
Minnaar C, de Jager ML, Anderson B (2019) Intraspecific divergence in floral-tube length promotes asymmetric pollen movement and reproductive isolation. New Phytol 224:1160–1170. https://doi.org/10.1111/nph.15971
Moeller DA (2004) Facilitative interactions among plants via shared pollinators. Ecology 85:3289–3301. https://doi.org/10.1890/03-0810
Morales CL, Traveset A (2008) Interspecific pollen transfer: magnitude, prevalence and consequences for plant fitness. Crit Rev Plant Sci 27:221–238. https://doi.org/10.1080/07352680802205631
Muchhala N (2008) Functional significance of interspecific variation in Burmeistera flower morphology: evidence from nectar bat captures in Ecuador. Biotropica 40:332–337. https://doi.org/10.1111/j.1744-7429.2007.00381.x
Munin RL, Teixeira RC, Sigrist MR (2008) Esfingofilia E sistema de reprodução de Bauhinia curvula Benth. (Leguminosae: Caesalpinioideae) em cerrado no centro-oeste brasileiro. Rev bras Bot 31. https://doi.org/10.1590/S0100-84042008000100003
Nassar JM, Ramírez N, Linares O (1997) Comparative pollination biology of Venezuelan columnar cacti and the role of nectar-feeding bats in their sexual reproduction. Am J Bot 84:918–927. https://doi.org/10.2307/2446282
Paudel BR, Burd M, Shrestha M et al (2018) Reproductive isolation in alpine gingers: how do coexisting Roscoea (R. Purpurea and R. tumjensis) conserve species integrity? Evolution 72:1840–1850. https://doi.org/10.1111/evo.13546
Paulino-Neto HF (2013) Floral biology and breeding system of Bauhinia forficata (Leguminosae: Caesalpinioideae), a moth-pollinated tree in southeastern Brazil. Braz J Bot 36:55–64. https://doi.org/10.1007/s40415-013-0011-8
Peel MC, Finlayson BL, McMahon TA (2007) Updated world map of the Köppen-Geiger climate classification. Hydrol Earth Syst Sci 11:1633–1644. https://doi.org/10.5194/hess-11-1633-2007
Queiroz JA, Quirino ZGM, Machado IC (2015) Floral traits driving reproductive isolation of two co-flowering taxa that share vertebrate pollinators. AoB Plants 7:plv127. https://doi.org/10.1093/aobpla/plv127
Queiroz JA, Quirino ZGM, Lopes AV, Machado IC (2016) Vertebrate mixed pollination system in Encholirium spectabile: a bromeliad pollinated by bats, opossum and hummingbirds in a tropical dry forest. J Arid Environ 125:21–30. https://doi.org/10.1016/j.jaridenv.2015.09.015
Queiroz JA, Diniz UM, Vázquez DP, Quirino ZM, Santos FAR, Mello MAR, Machado IC (2021) Bats and hawkmoths form mixed modules with flowering plants in a nocturnal interaction network. Biotropica 53:596–607. https://doi.org/10.1111/btp.12902
Queiroz LP, Cardoso D, Fernandes MF, Moro MF (2017) Diversity and evolution of flowering plants of the Caatinga Domain. In: Silva JMC, Leal IR, Tabarelli M (eds) Caatinga. Springer, pp 23–63
Ramirez N, Sobrevila C, de Enrech NX, Ruiz-Zapata T (1984) Floral biology and breeding system of Bauhinia Benthamiana Taub. (Leguminosae), a bat-pollinated tree in Venezuelan Llanos. Am J Bot 71:273–280. https://doi.org/10.1002/j.1537-2197.1984.tb12514.x
Ramsey J, Bradshaw HD, Schemske DW (2003) Components of reproductive isolation between the monkeyflowers Mimulus lewisii and M. cardinalis (Phrymaceae). Evolution 57:1520–1534. https://doi.org/10.1111/j.0014-3820.2003.tb00360.x
Rao CB (2020) Carpenter bee pollination in the purple orchid tree, Bauhinia purpurea L. (sub-family Cercidoideae: Family Fabaceae). Species 21:244–248
Reis NR, Peracchi AL, Pedro WA, Lima IP (2007) Morcegos do Brasil. Edição dos Editores, Londrina
Reis NR, Fregonezi MN, Peracchi AL, Shibatta OA (2013) Morcegos do Brasil: guia de campo. Technical Books Editora, Rio de Janeiro
Rito KF, Arroyo-Rodríguez V, Queiroz RT, Leal IR, Tabarelli M (2017) Precipitation mediates the effect of human disturbance on the Brazilian caatinga vegetation. J Ecol 105:828–838. https://doi.org/10.1111/1365-2745.12712
Rocha EA, Machado IC, Zappi DC (2007) Floral biology of Pilosocereus tuberculatus (Werderm.) Byles & Rowley: a bat pollinated cactus endemic from the Caatinga in northeastern Brazil. Bradleya 25:129–144. https://doi.org/10.25223/brad.n25.2007.a10
Rocha EA, Domingos-Melo A, Zappi DC, Machado IC (2019) Reproductive biology of columnar cacti: are bats the only protagonists in the pollination of Pilosocereus, a typical chiropterophilous genus? Folia Geobot 54:239–256. https://doi.org/10.1007/s12224-019-09357-0
Sampaio EVSB (1995) Overview of the Brazilian Caatinga. In: Bullock SH, Mooney HA, Medina E (eds) Seasonally dry forests. Cambridge University Press, Cambridge, pp 35–58
Schemske DW, Bradshaw HD (1999) Pollinator preference and the evolution of floral traits in monkeyflowers (Mimulus). Proc Natl Acad Sci USA 96:11910–11915. https://doi.org/10.1073/pnas.96.21.11910
Schiestl FP, Schlüter PM (2009) Floral isolation, specialized pollination, and pollinator behavior in orchids. Annu Rev Entomol 54:425–446. https://doi.org/10.1146/annurev.ento.54.110807.090603
Sinou C, Forest F, Lewis GP, Bruneau A (2009) The genus Bauhinia s.l. (Leguminosae): a phylogeny based on the plastid trn L– trn F region. Botany 87:947–960. https://doi.org/10.1139/B09-065
Sobel JM, Chen GF (2014) Unification of methods for estimating the strength of reproductive isolation: estimating the strength of isolation. Evolution 68:1511–1522. https://doi.org/10.1111/evo.12362
Sokal RR, Rohlf FJ (1995) Biometry: the principles and practice of statistics in biological research, 3rd edn. W.H. Freeman, New York
Soltis PS, Soltis DE (2009) The role of hybridization in plant speciation. Annu Rev Plant Biol 60:561–588. https://doi.org/10.1146/annurev.arplant.043008.092039
Souza DG, Sfair JC, De Paula AS, Barros MF, Rito KF, Tabarelli M (2019) Multiple drivers of aboveground biomass in a human-modified landscape of the Caatinga dry forest. For Ecol Manag 435:57–65. https://doi.org/10.1016/j.foreco.2018.12.042
Stiles FG (1977) Coadapted competitors: the flowering seasons of hummingbird-pollinated plants in a tropical forest. Science 198:1177–1178. https://doi.org/10.1126/science.198.4322.1177
Sun M, Schlüter PM, Gross K, Schiestl FP (2015) Floral isolation is the major reproductive barrier between a pair of rewarding orchid sister species. J Evol Biol 28:117–129. https://doi.org/10.1111/jeb.12544
Tang J, Sun S, Huang S (2023) Experimental sympatry suggests geographic isolation as an essential reproductive barrier between two sister species of Pedicularis. J Syt Evol 61:428–439. https://doi.org/10.1111/jse.12835
Temeles EJ, Liang J, Levy MC, Fan Y-L (2019) Floral isolation and pollination in two hummingbird-pollinated plants: the roles of exploitation barriers and pollinator competition. Evol Ecol 33:481–497. https://doi.org/10.1007/s10682-019-09992-1
Tschapka M, Dressler S (2002) Chiropterophily: on bat-flowers and flower-bats. Curtis’s Bot Mag 19:114–125. https://doi.org/10.1111/1467-8748.00340
Vaz AMSF (2001) Taxonomia de Bauhinia sect. Pauletia (Leguminosae: Caesalpinioideae: Cercideae) no Brasil. Dissertation, Universidade Estadual de Campinas, Campinas, SP
Vaz AMSF, Azevedo Tozzi AMG (2003) Bauhinia ser. Cansenia (Leguminosae: Caesalpinioideae) no Brasil. Rodriguésia 54:55–143. https://doi.org/10.1590/2175-78602003548305
Vaz AMSF, Tozzi AMGA (2005) Sinopse de Bauhinia sect. Pauletia (Cav.) DC. (Leguminosae: Caesalpinioideae: Cercideae) no Brasil. Braz J Bot 28:477–491. https://doi.org/10.1590/S0100-84042005000300006
Vogel S, Machado IC, Lopes AV (2004) Harpochilus neesianus and other novel cases of chiropterophily in neotropical acanthaceae. Taxon 53:55–60. https://doi.org/10.2307/4135488
Vogel S, Lopes AV, Machado IC (2005) Bat pollination in the NE Brazilian endemic Mimosa lewisii: an unusual case and first report for the genus. Taxon 54:693–700. https://doi.org/10.2307/25065426
Willmer P (2011) Pollination and floral ecology. Princeton University Press, Princeton, N.J
Wunderlin RP (1983) Revision of the arborescent bauhinias (Fabaceae: Caesalpinioideae: Cercideae) native to Middle America. Ann Mo Bot Gard 70:95. https://doi.org/10.2307/2399009
Xu S, Schlüter PM, Scopece G, Breitkopf H, Gross K, Cozzolino S, Schiestl FP (2011) Floral isolation is the main reproductive barrier among closely related sexually deceptive orchids: reproductive isolation in Ophrys. Evolution 65:2606–2620. https://doi.org/10.1111/j.1558-5646.2011.01323.x
Yang C-F, Gituru RW, Guo Y-H (2007) Reproductive isolation of two sympatric louseworts, Pedicularis rhinanthoides and Pedicularis longiflora (Orobanchaceae): how does the same pollinator type avoid interspecific pollen transfer? Biol J Linn Soc 90:37–48. https://doi.org/10.1111/j.1095-8312.2007.00709.x
Zar JH (2010) Biostatistical analysis. Prentice Hall, Upper Saddle River, N.J
Acknowledgements
We thank Ugo Diniz for his careful review of the semi-final version of the manuscript, and for his help with some statistical analyses; Arthur Domingos-Melo for the drawings of the Fig. 5c and help in the field; Ana Jardelino for identifying the bats; Sofia Chalegre and Haymée Alencar for assistance in the field. Daniela Zappi and Nigel Taylor for their careful review and for improving the text language in the final version. The Long Term Ecological Project (LTER) PELD-Catimbau, coordinated by Dr. Marcelo Tabarelli, for logistical support.
Funding
This work was supported by the CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior), Brazil (PROEX-0487, 88887.898666/2023-00 awarded to S-AL and 001 to all authors) and CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico (Chamada Universal 459485/2014-8, 437424/2018-9, LTER-Catimbau 403770/2012-2; and research grants to ICM: 311021/2014-0 and 310508/2019-3, and AVL: 309505/20186 and 306286/2022-0).
Author information
Authors and Affiliations
Contributions
SA-L and ICM conceived the study and designed the experiment, SA-L conducted the field experiment, SA-L, AVL and ICM analysed the data, and wrote the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Albuquerque-Lima, S., Lopes, A.V. & Machado, I.C. Reproductive isolation between two sympatric bat-pollinated Bauhinia (Leguminosae). J Plant Res 137, 65–77 (2024). https://doi.org/10.1007/s10265-023-01508-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10265-023-01508-x