Abstract
Background
The mitochondrial protein frataxin regulates iron metabolism for heme and iron sulfur cluster synthesis in the mitochondria and could be associated with the regulation of oxidative stress. To clarify the expression of frataxin and its association with uremia, we evaluated the mRNA and protein levels of frataxin in the polymorphonuclear leukocytes (PMNLs) of patients on hemodialysis (HD).
Methods
Uremic patients on HD (n = 18) and healthy control subjects (n = 18) were investigated. PMNLs were isolated by differential centrifugation. The mRNA levels of frataxin in isolated leukocytes were quantified by TaqMan real-time polymerase chain reaction. Frataxin protein expression in the cell lysate was evaluated using SDS-polyacrylamide gel electrophoresis and Western blotting.
Results
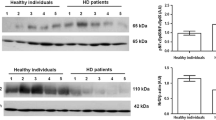
The frataxin/glyceraldehyde-3-phosphate dehydrogenase mRNA ratio in PMNLs from uremic patients was significantly lower than that in control subjects. Frataxin protein expression in uremic patients was also significantly lower than that in controls. Multiple regression analysis showed that frataxin mRNA levels were independently associated with the serum levels of both the oxidative stress marker malondialdehyde and the proinflammatory cytokine tumor necrosis factor-α.
Conclusion
The downregulation of frataxin seems to be linked with uremic status, which is usually associated with chronic inflammation and the acceleration of oxidative stress. Mitochondrial iron regulation may play a role in several comorbidities and in the poor prognosis in uremic patients. Further investigation is needed to elucidate whether reduced frataxin levels are linked to the pathological status of uremic patients and whether uremic substances affect frataxin expression.




Similar content being viewed by others
References
Gakh O, Park S, Liu G, et al. Mitochondrial iron detoxification is a primary function of frataxin that limits oxidative damage and preserves cell longevity. Hum Mol Genet. 2006;15(3):467–79.
Campuzano V, Montermini L, Lutz Y, et al. Frataxin is reduced in Friedreich ataxia patients and is associated with mitochondrial membranes. Hum Mol Genet. 1997;6(11):1771–80.
Wilson RB, Roof DM. Respiratory deficiency due to loss of mitochondrial DNA in yeast lacking the frataxin homologue. Nat Genet. 1997;16(4):352–7.
Wilson RB. Frataxin and frataxin deficiency in Friedreich’s ataxia. J Neurol Sci. 2003;207(1–2):103–5.
Campuzano V, Montermini L, Moltò MD, et al. Friedreich’s ataxia: autosomal recessive disease caused by an intronic GAA triplet repeat expansion. Science. 1996;271(5254):1423–7.
Rötig A, de Lonlay P, Chretien D, et al. Aconitase and mitochondrial iron-sulphur protein deficiency in Friedreich ataxia. Nat Genet. 1997;17(2):215–7.
Koutnikova H, Campuzano V, Foury F, Dollé P, Cazzalini O, Koenig M. Studies of human, mouse and yeast homologues indicate a mitochondrial function for frataxin. Nat Genet. 1997;16(4):345–51.
Otaki Y, Nakanishi T, Hasuike Y, et al. Defective regulation of iron transporters leading to iron excess in the polymorphonuclear leukocytes of patients on maintenance hemodialysis. Am J Kidney Dis. 2004;43(6):1030–9.
Moriguchi R, Otaki Y, Hazeki S, et al. High levels of tumor necrosis factor-α downregulate antimicrobial iron transport protein, Nramp1, in chronic hemodialysis patients: a key factor for infection risk. Am J Nephrol. 2012;35(4):372–8.
Yagi K. A simple fluorometric assay for lipoperoxide in blood plasma. Biochem Med. 1976;15(2):212–6.
Willis JH, Isaya G, Gakh O, Capaldi RA, Marusich MF. Lateral-flow immunoassay for the frataxin protein in Friedreich’s ataxia patients and carriers. Mol Genet Metab. 2008;94(4):491–7.
Cossée M, Dürr A, Schmitt M, et al. Friedreich’s ataxia: point mutations and clinical presentation of compound heterozygotes. Ann Neurol. 1999;45(2):200–6.
Boehm T, Scheiber-Mojdehkar B, Kluge B, Goldenberg H, Laccone F, Sturm B. Variations of frataxin protein levels in normal individuals. Neurol Sci. 2011;32(2):327–30.
Pandolfo M, Pastore A. The pathogenesis of Friedreich ataxia and the structure and function of frataxin. J Neurol. 2009;256(Suppl 1):9–17.
Pandolfo M. Frataxin deficiency and mitochondrial dysfunction. Mitochondrion. 2002;2(1–2):87–93.
Popescu BF, Pickering IJ, George GN, Nichol H. The chemical form of mitochondrial iron in Friedreich’s ataxia. J Inorg Biochem. 2007;101(6):957–66.
Kim W, Kim DW, Shin BN, et al. PEP-1-frataxin significantly increases cell proliferation and neuroblast differentiation by reducing lipid peroxidation in the mouse dentate gyrus. Neurochem Res. 2011;36(12):2452–8.
Lu C, Schoenfeld R, Shan Y, Tsai HJ, Hammock B, Cortopassi G. Frataxin deficiency induces Schwann cell inflammation and death. Biochim Biophys Acta. 2009;1792(11):1052–61.
Hasuike Y, Nonoguchi H, Tokuyama M, et al. Pathological role of aminolevulinate in uremic patients. Ther Apher Dial. 2011;15(1):28–33.
Hasuike Y, Nonoguchi H, Tokuyama M, et al. Serum ferritin predicts prognosis in hemodialysis patients: the Nishinomiya study. Clin Exp Nephrol. 2010;14(4):349–55.
Marmolino D. Friedreich’s ataxia: past, present and future. Brain Res Rev. 2011;67(1–2):311–30.
MacKenzie EL, Iwasaki K, Tsuji Y. Intracellular iron transport and storage: from molecular mechanisms to health implications. Antioxid Redox Signal. 2008;10(6):997–1030.
Zhao MM, Xu MJ, Cai Y, et al. Mitochondrial reactive oxygen species promote p65 nuclear translocation mediating high-phosphate-induced vascular calcification in vitro and in vivo. Kidney Int. 2011;79(10):1071–9.
Nachbauer W, Hering S, Seifert M, Steinkellner H, Sturm B, Scheiber-Mojdehkar B, Reindl M, Strasak A, Poewe W, Weiss G, Boesch S. Effects of erythropoietin on frataxin levels and mitochondrial function in Friedreich ataxia—a dose-response trial. Cerebellum. 2011;10(4):763–9.
Mariotti C, Fancellu R, Caldarazzo S, Nanetti L, Di Bella D, Plumari M, Lauria G, Cappellini MD, Duca L, Solari A, Taroni F. Erythropoietin in Friedreich ataxia: no effect on frataxin in a randomized controlled trial. Mov Disord. 2012;27(3):446–9.
Acknowledgments
The authors wish to thank Kiyo Maeda and Eiko Akabane for their technical assistance in preparing this paper.
Conflict of interest
The authors have declared that no conflict of interest exists.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Hasuike, Y., Nagai, T., Yorifuji, S. et al. The mitochondrial protein frataxin is downregulated in hemodialysis patients. Clin Exp Nephrol 17, 424–430 (2013). https://doi.org/10.1007/s10157-012-0737-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-012-0737-9