Abstract
Background
To evaluate the activity and safety of bevacizumab when given with standard 5FU/leukovorin (LV) regimens in patients with metastatic colorectal cancer who have failed irinotecan and oxaliplatin-based treatments.
Methods
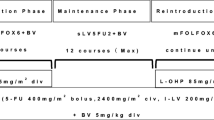
Bevacizumab was given at 5 mg/kg as an IV infusion every 2 weeks. Patients received 5FU according to Roswell Park or the de Gramont regimen.
Results
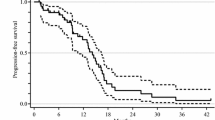
Nineteen patients enrolled, median age 60, median PS: 1. Most common toxicity attributable to bevacizumab was mild hypertension, epistaxis and mild proteinuria; 1 patient had a CNS haemorrhage. The median number of cycles was 1 (8 weeks). Clinical benefit as disease stabilisation lasting 2–6 months was noted in 9 patients, whereas 10 progressed (median f/u: 5 months). TTP was 16 weeks, and the overall survival has not been reached (24+ weeks).
Conclusions
Bevacizumab may result in growth arrest and clinical benefit in a substantial proportion of patients with colorectal cancer and no alternative treatment.
Similar content being viewed by others
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Emmanouilides, C., Pegram, M., Robinson, R. et al. Anti-VEGF antibody bevacizumab (Avastin) with 5FU/LV as third line treatment for colorectal cancer. Tech Coloproctol 8 (Suppl 1), s50–s52 (2004). https://doi.org/10.1007/s10151-004-0110-4
Issue Date:
DOI: https://doi.org/10.1007/s10151-004-0110-4