Abstract
Background
Axillary lymph node dissection (ALND) has been recommended to include levels I–III for melanoma patients who have evidence of metastasis in the axillary sentinel lymph node (SLN). The extent of the subsequent axillary dissection is in debate. The objective of this study was to determine the frequency of metastasis of level III nodes in addition to that of level II nodes in this setting.
Methods
A multi-institutional retrospective study was undertaken in 14 melanoma treatment centers in Japan.
Results
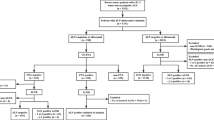
Between 2007 and 2012, 69 patients with involved axillary SLNs underwent a subsequent ALND and 55 underwent level I and II dissections. Level III metastatic nodes, which is our primary endpoint, were seen in only 1 patient (1.5 %). The level II metastatic rate was 4.4 %.
Conclusions
Our study sample size was small, but melanoma patients with positive SLN rarely had level III disease, suggesting that level III dissection may be unnecessary. We also found that level II metastasis was not so frequent. More evidence is needed to standardize the extent of ALND and to identify the patients who would have the most benefit with undergoing level II dissection for positive axillary SLNs.
Similar content being viewed by others
References
Morton DL, Thompson JF, Cochran AJ et al (2014) Final trial report of sentinel-node biopsy versus nodal observation in melanoma. N Engl J Med 370:599–609
Garbe C, Hauschild A, Volkenandt M et al (2008) Evidence-based and interdisciplinary consensus-based German guidelines: systemic medical treatment of melanoma in the adjuvant and palliative setting. Melanoma Res 18:152–160
Essner R (2003) Surgical treatment of malignant melanoma. Surg Clin N Am 83:109–156
NCCN (2015) Clinical Practical Guidelines in Oncology–Melanoma
Karakousis CP (1998) Therapeutic node dissections in malignant melanoma. Ann Surg Oncol 5:473–482
Meyer T, Merkel S, Gohl J et al (2002) Lymph node dissection for clinically evident lymph node metastases of malignant melanoma. Eur J Surg Oncol 28:424–430
Serpell JW, Carne PW, Bailey M (2003) Radical lymph node dissection for melanoma. ANZ J Surg 73:294–299
Sakorafas GH, Peros G, Cataliotti L et al (2006) Lymphedema following axillary lymph node dissection for breast cancer. Surg Oncol 15:153–165
Tominaga T, Takashima S, Danno M (2004) Randomized clinical trial comparing level II and level III node dissection in addition to mastectomy for breast cancer. Br J Surg 91:38–43
Namikawa K, Yamazaki N, Nakai Y et al (2012) Prediction of additional lymph node positivity and clinical outcome of micrometastases in sentinel lymph nodes in cutaneous melanoma: a multi-institutional study of 450 patients in Japan. J Dermatol 39:130–137
Namikawa K, Yamazaki N (2011) Sentinel lymph node biopsy guided by indocyanine green fluorescence for cutaneous melanoma. Eur J Dermatol 21:184–190
Namikawa K, Tsutsumida A, Tanaka R et al (2014) Limitation of indocyanine green fluorescence in identifying sentinel lymph node prior to skin incision in cutaneous melanoma. Int J Clin Oncol 19:198–203
Nessim C, Law C, McConnell Y et al (2013) How often do level III nodes bear melanoma metastases and does it affect patient outcomes? Ann Surg Oncol 20:2056–2064
Namm JP, Chang AE, Cimmino VM et al (2012) Is a level III dissection necessary for a positive sentinel lymph node in melanoma? J Surg Oncol 105:225–228
Curtin JA, Fridlyand J, Kageshita T et al (2005) Distinct sets of genetic alterations in melanoma. N Engl J Med 353:2135–2147
Broekaert SM, Roy R, Okamoto I et al (2010) Genetic and morphologic features for melanoma classification. Pigment Cell Melanoma Res 23:763–770
Ito T, Wada M, Nagae K et al (2015) Acral lentiginous melanoma: who benefits from sentinel lymph node biopsy? J Am Acad Dermatol 72:71–77
Acknowledgments
This work was partly supported by National Cancer Center Research and Development Fund (26-A-4). We thank Prof. Ryuhei Okuyama from Department of Dermatology, Shinshu University; Dr. Yasuhiro Nakamura and Prof. Akifumi Yamamoto from Department of Skin Oncology and Dermatology, Saitama Medical University International Medical Center; Prof. Hironobu Ihn from Department of Dermatology and Plastic Surgery, Kumamoto University; Dr. Yoshie Hasegawa and Prof. Masashi Akiyama from Department of Dermatology, Nagoya University; Prof. Akemi Yamamoto from Department of Dermatology, Asahikawa Medical University; Prof. Shinji Imafuku from Department of Dermatology, Fukuoka University; Dr. Kentaro Yonekura and Prof. Takuro Kanekura from Department of Dermatology, Kagoshima University; Dr. Takafumi Kadono from St. Marianna University School of Medicine; Prof. Shinichi Sato from Department of Dermatology, Graduate School of Medicine and Faculty of Medicine, The University of Tokyo; Dr. Takamichi Ito and Prof. Masutaka Furue from Department of Dermatology, Kyushu University; Prof. Manabu Fujimoto from Department of Dermatology, University of Tsukuba; and Prof. Tetsuya Tsuchida from Department of Dermatology, Saitama Medical University, for their cooperation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
About this article
Cite this article
Tsutsumida, A., Takahashi, A., Namikawa, K. et al. Frequency of level II and III axillary nodes metastases in patients with positive sentinel lymph nodes in melanoma: a multi-institutional study in Japan. Int J Clin Oncol 21, 796–800 (2016). https://doi.org/10.1007/s10147-015-0944-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-015-0944-y