Abstract
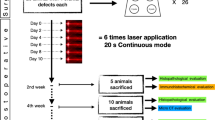
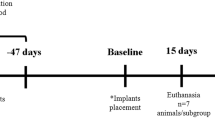
A new low-level laser therapy (LLLT) protocol is proposed and compared to another previously studied, in animal models, aiming to establish a more practical LLLT protocol. Protocol 1, the same used in other works and based on the clinical LLLT protocol for bone regeneration, consists of punctual transcutaneous applications in the defect region with fluence of 16 J/cm2 every 48 h for 15 days. Protocol 2, proposed in this work, consists of three sessions: the first application directly on the defect site with fluency of 3.7 J/cm2, during the surgical procedure, followed by two transcutaneous applications, 48 and 120 h postoperatively. The Thera Lase® (λ = 830 nm) was used, and the dosimetry of the first application of protocol 2 was calculated based on in vitro studies. Forty-five male rats were used, in which critical-size bone defects with 8 mm of diameter were surgically created in calvaria. The animals were randomly divided into three groups of 15 animals, named group 1 (protocol 1), group 2 (protocol 2), and control, which was not submitted to laser treatment. After 7, 15, and 45 days, five animals of each group were euthanized, and the pieces of calvarial bone were collected for microscopic and immunohistochemistry for vascular endothelial growth factor (VEGF), osteocalcin (OC), and osteopontin (OP) analysis. Histomorphometry showed that newly formed bone of 15-day samples from group 2 is higher than the control group (p < 0.05, ANOVA, Tukey). At 7 days, in the central part of the defect, VEGF expression was the same for all groups, OC was higher for protocol 2, and OP for protocol 1. The results suggest LLLT using the protocol 2 hastened the bone healing process in the early periods after surgery.




Similar content being viewed by others
References
Buser D (2009) 20 Years of guided bone regeneration in implant dentistry, 2nd edn. Quintessence book, London
Paiva-Oliveira EL, Lima NC, Silva PH, Sousa NTA, Barbosa FS, Orsini M, Silva JG (2012) Low-level laser therapy (LLLT) reduces inflammatory infiltrate and enhances skeletal muscle repair: histomorphometric parameters. Laser Phys 22(9):1425–1430
Pretel H, Lizarelli RFZ, Ramalho LTO (2007) Effect of low-level laser therapy on bone repair: histological study in rats. Lasers Surg Med 39(10):788–796
Poppi R, Silva A, Nacer R, Vieira R, Oliveira L, Faria-Junior NS, Tarso Camilo Carvalho P (2011) Evaluation of the osteogenic effect of low-level laser therapy (808 nm and 660 nm) on bone defects induced in the femurs of female rats submitted to ovariectomy. Lasers Med Sci 26(4):515–522
Jayasree RS, Gupta AK, Rathinam K, Mohanan PV, Mohanty M (2001) The influence of photodynamic therapy on the wound healing process in rats. J Appl Biomater 15(3):176–186
Tiphlova O, Karu T (1989) Role of primary photoacceptors in low-power laser effects: action of He-Ne laser radiation on bacteriophage T4-Escherichia coli interaction. Lasers Surg Med 9(1):67–69
Karu T, Pyatibrat L, Kalendo G (1995) Irradiation with He-Ne laser increases ATP level in cells cultivated in vitro. J Photochem Photobiol B: Biol 27(3):219–223
Pinheiro AB, Soares LP, Barbosa AS, Ramalho LP, Santos J (2012) Does LED phototherapy influence the repair of bone defects grafted with MTA, bone morphogenetic proteins, and guided bone regeneration? A description of the repair process on rodents. Lasers Med Sci 27(5):1013–1024
Ribeiro TP, Nascimento SB, Cardoso C, Alessandra D, Hage R, Almeida JD, Arisawa EAL (2012) Low-level laser therapy and calcitonin in bone repair: densitometric analysis. Int J Photoenergy. doi:10.1155/2012/829587
Omasa S, Motoyoshi M, Arai Y, Ejima K, Shimizu N (2012) Low-level laser therapy enhances the stability of orthodontic mini-implants via bone formation related to BMP-2 expression in a rat model. Photomed Laser Surg 30(5):255–261
Kawasaki K, Shimizu N (2000) Effects of low-energy laser irradiation on bone remodeling during experimental tooth movement in rats. Lasers Surg Med 26(3):282–291
Ninomiya T, Hosoya A, Nakamura H, Sano K, Nishisaka T, Ozawa H (2007) Increase of bone volume by a nanosecond pulsed laser irradiation is caused by a decreased osteoclast number and an activated osteoblasts. Bone 40(1):140–148
Kim YD, Kim SS, Hwang DS, Kim SG, Kwon YH, Shin SH, Kim UK, Kim JR, Chung IK (2007) Effect of low-level laser treatment after installation of dental titanium implant-immunohistochemical study of RANKL, RANK, OPG: an experimental study in rats. Lasers Surg Med 39(5):441–450
Fujita S, Yamaguchi M, Utsunomiya T, Yamamoto H, Kasai K (2008) Low-energy laser stimulates tooth movement velocity via expression of RANK and RANKL. Orthod Craniofac Res 11(3):143–155
Matsumoto MA, Ferino RV, Monteleone GF, Ribeiro DA (2009) Low-level laser therapy modulates cyclo-oxygenase-2 expression during bone repair in rats. Lasers Med Sci 24(2):195–201
Ribeiro DA, Matsumoto MA (2008) Low-level laser therapy improves bone repair in rats treated with anti-inflammatory drugs. J Oral Rehabil 35(12):925–933
Fukuhara E, Goto T, Matayoshi T, Kobayashi S, Takahashi T (2006) Optimal low-energy laser irradiation causes temporal G2/M arrest on rat calvarial osteoblasts. Calcif Tissue Int 79(6):443–450
Ozawa Y, Shimizu N, Kariya G, Abiko Y (1998) Low-energy laser irradiation stimulates bone nodule formation at early stages of cell culture in rat calvarial cells. Bone 22(4):347–354
Barbosa D, Souza R, Xavier M, Silva F, Arisawa E, Villaverde A (2013) Effects of low-level laser therapy (LLLT) on bone repair in rats: optical densitometry analysis. Lasers Med Sci 28(2):651–656
Janet CG, Barbee RW, Bielitzki JT, Clayton LA, Donovan JC, Hendriksen CFM, Kohn DF, Lipman NS, Locke PA, Melcher J, Quimby FW, Turner PV, Wood GA, Würbel H (2011) Guide for the care and use of laboratory animals, 8th edn. National Academies Press, Washington DC
Nagata M, Messora M, Okamoto R, Campos N, Pola N, Esper L, Sbrana M, Fucini S, Garcia V, Bosco A (2009) Influence of the proportion of particulate autogenous bone graft/platelet-rich plasma on bone healing in critical-size defects: an immunohistochemical analysis in rat calvaria. Bone 5(2):339–345
Acknowledgement
The authors would like to thank CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico) for the fellowship to LM and Wilson Orcini and Maira Couto for the technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Marques, L., Holgado, L.A., Francischone, L.A. et al. New LLLT protocol to speed up the bone healing process—histometric and immunohistochemical analysis in rat calvarial bone defect. Lasers Med Sci 30, 1225–1230 (2015). https://doi.org/10.1007/s10103-014-1580-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10103-014-1580-x