Abstract
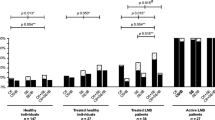
This retrospective case-control study assesses the sensitivity, specificity, and area under the curve of the ZEUS Borrelia VlsE1/pepC10 assay in comparison with the C6-ELISA in European patients with Lyme borreliosis, healthy blood donors, and potentially cross-reactive controls. We included a convenience series of 161 sera from patients with physician-confirmed early localized or disseminated Lyme borreliosis (n = 143), 400 sera from healthy blood donors and 44 sera with potentially cross-reactive antibodies, on which we performed the aforementioned serological assays and the recomLine immunoblot. Diagnostic parameters were compared in various single-tier and two-tier algorithms. The specificities of the C6-ELISA and the ZEUS Borrelia VlsE1/pepC10 were comparable in healthy blood donors (e.g., single-tier permissive: C6: 362/400, 90.5% [87.2–93.2]; VlsE1/pepC10: 361/400, 90.3% [86.9–93.0]). The C6-ELISA had an apparently higher sensitivity in EM sera (e.g., both time points combined: C6: 61/76, 80.3% [69.5–88.5]; VlsE1/pepC10: 54/76, 71.1% [59.5–80.9]), but these differences were all not-significant. Interestingly, the VlsE1/pepC10 assay had a significantly higher specificity in sera with potentially cross-reactive antibodies (e.g., single-tier permissive: C6: 34/44, 77.3% [62.2–88.5]; VlsE1/pepC10: 40/44, 90.9% [78.3–97.5]; p = 0.031). While the areas under the curve for both assays were excellent, that of the C6-ELISA exceeded that of the VlsE1/pepC10 (C6: AUC = 0.925; VlsE1/pepC10: AUC = 0.878; p = 0.003). The novel ZEUS Borrelia VlsE1/pepC10 assay has generally comparable diagnostic parameters to the C6-ELISA with potentially improved specificity in cross-reactive sera. Thus, it is a useful tool for the serodiagnosis of Lyme borreliosis in Europe.

Similar content being viewed by others
Availability of data and materials
De-identified data for this project can be obtained from the corresponding author upon reasonable request.
References
Waddell LA, Greig J, Mascarenhas M, Harding S, Lindsay R, Ogden N (2016) The accuracy of diagnostic tests for lyme disease in humans, a systematic review and meta-analysis of North American Research. PLoS One 11(12):e0168613
Leeflang MMG, Ang CW, Berkhout J, Bijlmer HA, Van Bortel W, Brandenburg AH et al (2016) The diagnostic accuracy of serological tests for Lyme borreliosis in Europe: a systematic review and meta-analysis. BMC Infect Dis 16:140
Liang FT, Alvarez AL, Gu Y, Nowling JM, Ramamoorthy R, Philipp MT (1999) An immunodominant conserved region within the variable domain of VlsE, the variable surface antigen of Borrelia burgdorferi. J Immunol 163(10):5566–5573
Baarsma ME, Schellekens J, Meijer BC, Brandenburg AH, Souilljee T, Hofhuis A et al (2020) Diagnostic parameters of modified two-tier testing in European patients with early Lyme disease. Eur J Clin Microbiol Infect Dis 39(11):2143–2152
Branda JA, Linskey K, Kim YA, Steere AC, Ferraro MJ (2011) Two-tiered antibody testing for Lyme disease with use of 2 enzyme immunoassays, a whole-cell sonicate enzyme immunoassay followed by a VlsE C6 peptide enzyme immunoassay. Clin Infect Dis 53(6):541–547
Branda JA, Strle K, Nigrovic LE, Lantos PM, Lepore TJ, Damle NS et al (2017) Evaluation of modified 2-tiered serodiagnostic testing algorithms for early Lyme disease. Clin Infect Dis 64(8):1074–1080
Lantos PM, Rumbaugh J, Bockenstedt LK, Falck-Ytter YT, Aguero-Rosenfeld ME, Auwaerter PG et al (2021) Clinical Practice Guidelines by the Infectious Diseases Society of America (IDSA), American Academy of Neurology (AAN), and American College of Rheumatology (ACR): 2020 guidelines for the prevention, diagnosis and treatment of Lyme disease. Clin Infect Dis 72(1):e1–e48
Wormser GP, Schriefer M, Aguero-Rosenfeld ME, Levin A, Steere AC, Nadelman RB et al (2013) Single-tier testing with the C6 peptide ELISA kit compared with two-tier testing for Lyme disease. Diagn Microbiol Infect Dis 75(1):9–15
Mathiesen MJ, Christiansen M, Hansen K, Holm A, Asbrink E, Theisen M (1998) Peptide-based OspC enzyme-linked immunosorbent assay for serodiagnosis of Lyme borreliosis. J Clin Microbiol 36(12):3474–3479
Porwancher RB, Hagerty CG, Fan J, Landsberg L, Johnson BJ, Kopnitsky M et al (2011) Multiplex immunoassay for Lyme disease using VlsE1-IgG and pepC10-IgM antibodies: improving test performance through bioinformatics. Clin Vaccine Immunol 18(5):851–859
Bacon RM, Biggerstaff BJ, Schriefer ME, Gilmore RD Jr, Philipp MT, Steere AC et al (2003) Serodiagnosis of Lyme disease by kinetic enzyme-linked immunosorbent assay using recombinant VlsE1 or peptide antigens of Borrelia burgdorferi compared with 2-tiered testing using whole-cell lysates. J Infect Dis 187(8):1187–1199
Rutjes AW, Reitsma JB, Vandenbroucke JP, Glas AS, Bossuyt PM (2005) Case-control and two-gate designs in diagnostic accuracy studies. Clin Chem 51(8):1335–1341
Baarsma ME, Ursinus J, Zaaijer HL, Kuiper H, Hovius JW (2021) Diagnostic performance of the novel BioPlex Lyme serological assays in European patients with Lyme disease. J Clin Microbiol 59(7):e0320520.
Stanek G, Fingerle V, Hunfeld KP, Jaulhac B, Kaiser R, Krause A et al (2011) Lyme borreliosis: clinical case definitions for diagnosis and management in Europe. Clin Microbiol Infect 17(1):69–79
Borrelia VlsE1/pepC10 IgG/IgM Test System: ZEUS Scientific. Available from: https://www.zeusscientific.com/products/zeus-elisa-test-systems/zeus-elisa-borrelia-vlse1-pepc10-igg-igm-test-system. Accessed 3 Sept 2021
FDA clears new indications for existing Lyme disease tests that may help streamline diagnoses [press release]. Food and Drug Administration, 29 July, 2019. Available from:https://www.fda.gov/news-events/press-announcements/fda-clears-new-indications-existing-lyme-disease-testsmay-help-streamline-diagnoses. Accessed 1 Sept 2021
Acknowledgements
The authors thank the following colleagues for their assistance in performing and interpreting the immunoblots: Dieuwertje Hoornstra, MD; Nienke Verhaar, BSc; Jacqueline van Eck, BSc; and Alex Wagemakers, MD, PhD. They also thank Alexis Burnham, MSc, for her assistance in performing the ELISAs; Aradna Balak and Titia Moshe-Sterk for their assistance in selecting the healthy blood donors; Anneke Oei and Herman Kuiper, MD, for the inclusion of LB patients and development of the AMLC serum biobank; Jeroen Coumou, MD, PhD, for his assistance in providing an overview of the clinical information of the LB patients; Anita Buskermolen and other colleagues at the clinical serological laboratory of Amsterdam UMC for performing the C6-ELISAs and immunoblots for routine clinical diagnostics; and Alexander Platonov, PhD and Joris Koetsveld, MD, PhD for collecting and supplying the BMD sera.
Funding
The assays for this project were supplied free of charge by ITK Diagnostics (Uithoorn, The Netherlands) on behalf of ZEUS Scientific. Neither company had any role in the acquisition of the study data, analyses thereof or manuscript preparation. This study was supported by a grant to JWH and AV from the European Union through the European Regional Development Fund and the Interreg North Sea Region Programme 2014–2020 as part of the NorthTick project (reference number J-No: 38–2-7–19).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
This project was submitted to the Medical Ethics Committee of Amsterdam UMC, which found it to be outside the scope of the Dutch Medical Research Involving Human Subjects Act (MEC decision W20_455).
Consent to participate
As this study used only de-identified leftover patient materials, informed consent from patients was not required. Healthy blood donors consented specifically to their materials being used for scientific purposes.
Consent to publish
Not applicable.
Conflict of interest
None of the authors have received any personal financial compensation from ZEUS Scientific or any of the other assays’ manufacturers. MEB and JWH have in the past collaborated with Bio-Rad Laboratories (Hercules, CA, USA), QIAGEN (Germantown, MD, USA), Oxford Immunotec (Oxford, UK), Invitalabs (Neuss, Germany) and AID (Strassberg, Germany) on unrelated projects on LB diagnostics. They have not received any personal financial compensation from any of the aforementioned companies. MEB, APvD and JWH collaborate with Pfizer (New York City, NY, USA) on unrelated projects on LD. They have not received any personal financial compensation from Pfizer or any of the other companies for these projects. The other authors report no potential conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Baarsma, M.E., Vrijlandt, A., Ursinus, J. et al. Diagnostic performance of the ZEUS Borrelia VlsE1/pepC10 assay in European LB patients: a case–control study. Eur J Clin Microbiol Infect Dis 41, 387–393 (2022). https://doi.org/10.1007/s10096-021-04372-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-021-04372-6