Abstract
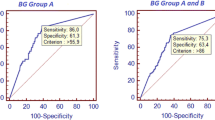
Detection of the fungal cell wall component beta-glucan (BG) in serum is increasingly used to diagnose invasive fungal infections (IFI), but its optimal use in hematology patients with high risk of IFI is not well defined. We retrospectively analyzed the diagnostic accuracy, optimal cut-off level, and potential confounding factors of BG reactivity. The inclusion criteria were: adult patients with hematologic disease who were admitted to the hematology ward during the 2-year study period and who had two or more consecutive BG assays performed. In total, 127 patients were enrolled. Thirteen patients with proven or probable IFI, as defined by the 2008 European Organization for Research and Treatment of Cancer/Mycoses Study Group (EORTC/MSG) criteria, were identified. Receiver operating characteristic (ROC) curve analysis showed a high overall diagnostic performance (area under the ROC curve = 0.98) and suggested an optimal cut-off level of 158 pg/ml, with a sensitivity and a specificity of 92 % and 96 %, respectively. Multiway analysis of variance indicated that treatment with pegylated asparaginase (p < 0.001), admission to the intensive care unit (ICU; p = 0.0007), and treatment with albumin, plasma, or coagulation factors (p = 0.01) are potential confounding factors of BG reactivity. We propose that a higher cut-off level than that recommended by the manufacturer should be used to monitor adult hematology patients at high risk for IFI. Our results also suggest that elevated BG levels in patients treated with pegylated asparaginase, albumin, plasma, or coagulation factors, or those admitted to the ICU should be interpreted with caution.


Similar content being viewed by others
References
Fukuda T, Boeckh M, Carter RA, Sandmaier BM, Maris MB, Maloney DG, Martin PJ, Storb RF, Marr KA (2003) Risks and outcomes of invasive fungal infections in recipients of allogeneic hematopoietic stem cell transplants after nonmyeloablative conditioning. Blood 102(3):827–833. doi:10.1182/blood-2003-02-0456
Kurosawa M, Yonezumi M, Hashino S, Tanaka J, Nishio M, Kaneda M, Ota S, Koda K, Suzuki N, Yoshida M, Hirayama Y, Takimoto R, Torimoto Y, Mori A, Takahashi T, Iizuka S, Ishida T, Kobayashi R, Oda T, Sakai H, Yamamoto S, Takahashi F, Fukuhara T (2012) Epidemiology and treatment outcome of invasive fungal infections in patients with hematological malignancies. Int J Hematol 96(6):748–757. doi:10.1007/s12185-012-1210-y
Nucci M, Garnica M, Gloria AB, Lehugeur DS, Dias VC, Palma LC, Cappellano P, Fertrin KY, Carlesse F, Simões B, Bergamasco MD, Cunha CA, Seber A, Ribeiro MP, Queiroz-Telles F, Lee ML, Chauffaille ML, Silla L, de Souza CA, Colombo AL (2013) Invasive fungal diseases in haematopoietic cell transplant recipients and in patients with acute myeloid leukaemia or myelodysplasia in Brazil. Clin Microbiol Infect 19(8):745–751. doi:10.1111/1469-0691.12002
Pagano L, Caira M, Candoni A, Offidani M, Fianchi L, Martino B, Pastore D, Picardi M, Bonini A, Chierichini A, Fanci R, Caramatti C, Invernizzi R, Mattei D, Mitra ME, Melillo L, Aversa F, Van Lint MT, Falcucci P, Valentini CG, Girmenia C, Nosari A (2006) The epidemiology of fungal infections in patients with hematologic malignancies: the SEIFEM-2004 study. Haematologica 91(8):1068–1075
Pagano L, Caira M, Nosari A, Van Lint MT, Candoni A, Offidani M, Aloisi T, Irrera G, Bonini A, Picardi M, Caramatti C, Invernizzi R, Mattei D, Melillo L, de Waure C, Reddiconto G, Fianchi L, Valentini CG, Girmenia C, Leone G, Aversa F (2007) Fungal infections in recipients of hematopoietic stem cell transplants: results of the SEIFEM B-2004 study—Sorveglianza Epidemiologica Infezioni Fungine Nelle Emopatie Maligne. Clin Infect Dis 45(9):1161–1170. doi:10.1086/522189
Neofytos D, Horn D, Anaissie E, Steinbach W, Olyaei A, Fishman J, Pfaller M, Chang C, Webster K, Marr K (2009) Epidemiology and outcome of invasive fungal infection in adult hematopoietic stem cell transplant recipients: analysis of Multicenter Prospective Antifungal Therapy (PATH) Alliance registry. Clin Infect Dis 48(3):265–273. doi:10.1086/595846
Nivoix Y, Velten M, Letscher-Bru V, Moghaddam A, Natarajan-Amé S, Fohrer C, Lioure B, Bilger K, Lutun P, Marcellin L, Launoy A, Freys G, Bergerat JP, Herbrecht R (2008) Factors associated with overall and attributable mortality in invasive aspergillosis. Clin Infect Dis 47(9):1176–1184. doi:10.1086/592255
Chryssanthou E, Kalin M, Engervall P, Petrini B, Björkholm M (1998) Low incidence of candidaemia among neutropenic patients treated for haematological diseases. Scand J Infect Dis 30(5):489–493
Simoneau E, Kelly M, Labbe AC, Roy J, Laverdière M (2005) What is the clinical significance of positive blood cultures with Aspergillus sp in hematopoietic stem cell transplant recipients? A 23 year experience. Bone Marrow Transplant 35(3):303–306. doi:10.1038/sj.bmt.1704793
Tarrand JJ, Lichterfeld M, Warraich I, Luna M, Han XY, May GS, Kontoyiannis DP (2003) Diagnosis of invasive septate mold infections. A correlation of microbiological culture and histologic or cytologic examination. Am J Clin Pathol 119(6):854–858. doi:10.1309/exbv-yaup-enbm-285y
De Pauw B, Walsh TJ, Donnelly JP, Stevens DA, Edwards JE, Calandra T, Pappas PG, Maertens J, Lortholary O, Kauffman CA, Denning DW, Patterson TF, Maschmeyer G, Bille J, Dismukes WE, Herbrecht R, Hope WW, Kibbler CC, Kullberg BJ, Marr KA, Muñoz P, Odds FC, Perfect JR, Restrepo A, Ruhnke M, Segal BH, Sobel JD, Sorrell TC, Viscoli C, Wingard JR, Zaoutis T, Bennett JE; European Organization for Research and Treatment of Cancer/Invasive Fungal Infections Cooperative Group; National Institute of Allergy and Infectious Diseases Mycoses Study Group (EORTC/MSG) (2008) Revised definitions of invasive fungal disease from the European Organization for Research and Treatment of Cancer/Invasive Fungal Infections Cooperative Group and the National Institute of Allergy and Infectious Diseases Mycoses Study Group (EORTC/MSG) Consensus Group. Clin Infect Dis 46(12):1813–1821. doi:10.1086/588660
Marchetti O, Lamoth F, Mikulska M, Viscoli C, Verweij P, Bretagne S; European Conference on Infections in Leukemia (ECIL) Laboratory Working Groups (2012) ECIL recommendations for the use of biological markers for the diagnosis of invasive fungal diseases in leukemic patients and hematopoietic SCT recipients. Bone Marrow Transplant 47(6):846–854. doi:10.1038/bmt.2011.178
Ellis M, Al-Ramadi B, Finkelman M, Hedstrom U, Kristensen J, Ali-Zadeh H, Klingspor L (2008) Assessment of the clinical utility of serial beta-D-glucan concentrations in patients with persistent neutropenic fever. J Med Microbiol 57(Pt 3):287–295. doi:10.1099/jmm.0.47479-0
Hachem RY, Kontoyiannis DP, Chemaly RF, Jiang Y, Reitzel R, Raad I (2009) Utility of galactomannan enzyme immunoassay and (1,3) beta-D-glucan in diagnosis of invasive fungal infections: low sensitivity for Aspergillus fumigatus infection in hematologic malignancy patients. J Clin Microbiol 47(1):129–133. doi:10.1128/jcm.00506-08
Odabasi Z, Mattiuzzi G, Estey E, Kantarjian H, Saeki F, Ridge RJ, Ketchum PA, Finkelman MA, Rex JH, Ostrosky-Zeichner L (2004) Beta-D-glucan as a diagnostic adjunct for invasive fungal infections: validation, cutoff development, and performance in patients with acute myelogenous leukemia and myelodysplastic syndrome. Clin Infect Dis 39(2):199–205. doi:10.1086/421944
Pazos C, Pontón J, Del Palacio A (2005) Contribution of (1->3)-beta-D-glucan chromogenic assay to diagnosis and therapeutic monitoring of invasive aspergillosis in neutropenic adult patients: a comparison with serial screening for circulating galactomannan. J Clin Microbiol 43(1):299–305. doi:10.1128/jcm.43.1.299-305.2005
Ostrosky-Zeichner L, Alexander BD, Kett DH, Vazquez J, Pappas PG, Saeki F, Ketchum PA, Wingard J, Schiff R, Tamura H, Finkelman MA, Rex JH (2005) Multicenter clinical evaluation of the (1-->3) beta-D-glucan assay as an aid to diagnosis of fungal infections in humans. Clin Infect Dis 41(5):654–659. doi:10.1086/432470
Lamoth F, Cruciani M, Mengoli C, Castagnola E, Lortholary O, Richardson M, Marchetti O; Third European Conference on Infections in Leukemia (ECIL-3) (2012) beta-Glucan antigenemia assay for the diagnosis of invasive fungal infections in patients with hematological malignancies: a systematic review and meta-analysis of cohort studies from the Third European Conference on Infections in Leukemia (ECIL-3). Clin Infect Dis 54(5):633–643. doi:10.1093/cid/cir897
Tamura H, Tanaka S, Oda T, Uemura Y, Aketagawa J, Hashimoto Y (1996) Purification and characterization of a (1-->3)-beta-D-glucan-binding protein from horseshoe crab (Tachypleus tridentatus) amoebocytes. Carbohydr Res 295:103–116
Marty FM, Lowry CM, Lempitski SJ, Kubiak DW, Finkelman MA, Baden LR (2006) Reactivity of (1-->3)-beta-d-glucan assay with commonly used intravenous antimicrobials. Antimicrob Agents Chemother 50(10):3450–3453. doi:10.1128/aac.00658-06
Mennink-Kersten MA, Warris A, Verweij PE (2006) 1,3-beta-D-glucan in patients receiving intravenous amoxicillin–clavulanic acid. N Engl J Med 354(26):2834–2835. doi:10.1056/NEJMc053340
Ikemura K, Ikegami K, Shimazu T, Yoshioka T, Sugimoto T (1989) False-positive result in Limulus test caused by Limulus amebocyte lysate-reactive material in immunoglobulin products. J Clin Microbiol 27(9):1965–1968
Nagasawa K, Yano T, Kitabayashi G, Morimoto H, Yamada Y, Ohata A, Usami M, Horiuchi T (2003) Experimental proof of contamination of blood components by (1-->3)-beta-D-glucan caused by filtration with cellulose filters in the manufacturing process. J Artif Organs 6(1):49–54. doi:10.1007/s100470300008
Ogawa M, Hori H, Niiguchi S, Azuma E, Komada Y (2004) False-positive plasma (1-->3)-beta-D-glucan test following immunoglobulin product replacement in an adult bone marrow recipient. Int J Hematol 80(1):97–98
Ohata A, Usami M, Horiuchi T, Nagasawa K, Kinoshita K (2003) Release of (1-->3)-beta-D-glucan from depth-type membrane filters and their in vitro effects on proinflammatory cytokine production. Artif Organs 27(8):728–735
Usami M, Ohata A, Horiuchi T, Nagasawa K, Wakabayashi T, Tanaka S (2002) Positive (1-->3)-beta-D-glucan in blood components and release of (1-->3)-beta-D-glucan from depth-type membrane filters for blood processing. Transfusion 42(9):1189–1195
Digby J, Kalbfleisch J, Glenn A, Larsen A, Browder W, Williams D (2003) Serum glucan levels are not specific for presence of fungal infections in intensive care unit patients. Clin Diagn Lab Immunol 10(5):882–885
Mennink-Kersten MA, Ruegebrink D, Verweij PE (2008) Pseudomonas aeruginosa as a cause of 1,3-beta-D-glucan assay reactivity. Clin Infect Dis 46(12):1930–1931. doi:10.1086/588563
Kimura Y, Nakao A, Tamura H, Tanaka S, Takagi H (1995) Clinical and experimental studies of the limulus test after digestive surgery. Surg Today 25(9):790–794
Nakao A, Yasui M, Kawagoe T, Tamura H, Tanaka S, Takagi H (1997) False-positive endotoxemia derives from gauze glucan after hepatectomy for hepatocellular carcinoma with cirrhosis. Hepatogastroenterology 44(17):1413–1418
Kanda H, Kubo K, Hamasaki K, Kanda Y, Nakao A, Kitamura T, Fujita T, Yamamoto K, Mimura T (2001) Influence of various hemodialysis membranes on the plasma (1-->3)-beta-D-glucan level. Kidney Int 60(1):319–323. doi:10.1046/j.1523-1755.2001.00802.x
Kato A, Takita T, Furuhashi M, Takahashi T, Maruyama Y, Hishida A (2001) Elevation of blood (1-->3)-beta-D-glucan concentrations in hemodialysis patients. Nephron 89(1):15–19. doi:10.1159/000046037
Yoshioka T, Ikegami K, Ikemura K, Shiono S, Uenishi M, Sugimoto H, Sugimoto T (1989) A study on limulus amebocyte lysate (LAL) reactive material derived from dialyzers. Jpn J Surg 19(1):38–41
Metan G, Ağkuş C, Buldu H, Koç AN (2010) The interaction between piperacillin/tazobactam and assays for Aspergillus galactomannan and 1,3-beta-D-glucan in patients without risk factors for invasive fungal infections. Infection 38(3):217–221. doi:10.1007/s15010-010-0003-6
Metan G, Agkus C, Nedret Koc A, Elmali F, Finkelman MA (2012) Does ampicillin–sulbactam cause false positivity of (1,3)-beta-D-glucan assay? A prospective evaluation of 15 patients without invasive fungal infections. Mycoses 55(4):366–371. doi:10.1111/j.1439-0507.2011.02131.x
Metan G, Koc AN, Ağkuş Ç, Kaynar LG, Alp E, Eser B (2012) Can bacteraemia lead to false positive results in 1,3-beta-D-glucan test? Analysis of 83 bacteraemia episodes in high-risk patients for invasive fungal infections. Rev Iberoam Micol 29(3):169–171. doi:10.1016/j.riam.2011.07.003
Racil Z, Kocmanova I, Toskova M, Winterova J, Lengerova M, Timilsina S, Mayer J (2013) Reactivity of the 1,3-beta-D-glucan assay during bacteraemia: limited evidence from a prospective study. Mycoses 56(2):101–104. doi:10.1111/j.1439-0507.2012.02210.x
Racil Z, Kocmanova I, Lengerova M, Weinbergerova B, Buresova L, Toskova M, Winterova J, Timilsina S, Rodriguez I, Mayer J (2010) Difficulties in using 1,3-{beta}-D-glucan as the screening test for the early diagnosis of invasive fungal infections in patients with haematological malignancies—high frequency of false-positive results and their analysis. J Med Microbiol 59(Pt 9):1016–1022. doi:10.1099/jmm.0.019299-0
Odabasi Z, Paetznick VL, Rodriguez JR, Chen E, McGinnis MR, Ostrosky-Zeichner L (2006) Differences in beta-glucan levels in culture supernatants of a variety of fungi. Med Mycol 44(3):267–272. doi:10.1080/13693780500474327
Acosta J, Catalan M, del Palacio-Peréz-Medel A, Lora D, Montejo JC, Cuetara MS, Moragues MD, Ponton J, del Palacio A (2011) A prospective comparison of galactomannan in bronchoalveolar lavage fluid for the diagnosis of pulmonary invasive aspergillosis in medical patients under intensive care: comparison with the diagnostic performance of galactomannan and of (1-->3)-beta-d-glucan chromogenic assay in serum sample. Clin Microbiol Infect 17(7):1053–1060. doi:10.1111/j.1469-0691.2010.03357.x
Alam FF, Mustafa AS, Khan ZU (2007) Comparative evaluation of (1, 3)-beta-D-glucan, mannan and anti-mannan antibodies, and Candida species-specific snPCR in patients with candidemia. BMC Infect Dis 7:103. doi:10.1186/1471-2334-7-103
Persat F, Ranque S, Derouin F, Michel-Nguyen A, Picot S, Sulahian A (2008) Contribution of the (1-->3)-beta-D-glucan assay for diagnosis of invasive fungal infections. J Clin Microbiol 46(3):1009–1013. doi:10.1128/jcm.02091-07
Pickering JW, Sant HW, Bowles CA, Roberts WL, Woods GL (2005) Evaluation of a (1->3)-beta-D-glucan assay for diagnosis of invasive fungal infections. J Clin Microbiol 43(12):5957–5962. doi:10.1128/jcm.43.12.5957-5962.2005
De Vlieger G, Lagrou K, Maertens J, Verbeken E, Meersseman W, Van Wijngaerden E (2011) Beta-D-glucan detection as a diagnostic test for invasive aspergillosis in immunocompromised critically ill patients with symptoms of respiratory infection: an autopsy-based study. J Clin Microbiol 49(11):3783–3787. doi:10.1128/jcm.00879-11
Koo S, Bryar JM, Page JH, Baden LR, Marty FM (2009) Diagnostic performance of the (1-->3)-beta-D-glucan assay for invasive fungal disease. Clin Infect Dis 49(11):1650–1659. doi:10.1086/647942
Narta UK, Kanwar SS, Azmi W (2007) Pharmacological and clinical evaluation of L-asparaginase in the treatment of leukemia. Crit Rev Oncol Hematol 61(3):208–221. doi:10.1016/j.critrevonc.2006.07.009
Oettgen HF, Stephenson PA, Schwartz MK, Leeper RD, Tallai L, Tan CC, Clarkson BD, Golbey RB, Krakoff IH, Karnofsky DA, Murphy ML, Burchenal JH (1970) Toxicity of E. coli L-asparaginase in man. Cancer 25(2):253–278
Parsons SK, Skapek SX, Neufeld EJ, Kuhlman C, Young ML, Donnelly M, Brunzell JD, Otvos JD, Sallan SE, Rifai N (1997) Asparaginase-associated lipid abnormalities in children with acute lymphoblastic leukemia. Blood 89(6):1886–1895
Sendid B, Francois N, Decool V, Poissy J, Poulain D (2013) Strategy for overcoming serum interferences in detection of serum (1,3)-beta-D-glucans. J Clin Microbiol 51(1):375–376. doi:10.1128/jcm.02356-12
Acknowledgments
This study was financed by Västra Götaland Health Care Region ALF project grants (ALF 71580 and ALF 74860), Strategic ALF Transplantation project grant (ALF 74080), and the Cancer and Allergy Foundation.
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical standards
The study was approved by the Regional Ethics Committee of Göteborg and has been performed in accordance with the ethical standards of the 1964 Declaration of Helsinki and its later amendments. The Ethics Committee assessed that there was no requirement of informed consent by the patients due to the structure of the study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hammarström, H., Kondori, N., Friman, V. et al. How to interpret serum levels of beta-glucan for the diagnosis of invasive fungal infections in adult high-risk hematology patients: optimal cut-off levels and confounding factors. Eur J Clin Microbiol Infect Dis 34, 917–925 (2015). https://doi.org/10.1007/s10096-014-2302-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-014-2302-9