Abstract
Introduction
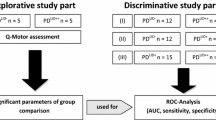
The acute levodopa challenge test (ALCT) is an important and valuable examination but there are still some shortcomings with it. We aimed to objectively assess ALCT based on a depth camera and filter out the best indicators.
Methods
Fifty-nine individuals with parkinsonism completed ALCT and the improvement rate (IR, which indicates the change in value before and after levodopa administration) of the Movement Disorder Society-Sponsored Revision of the Unified Parkinson’s Disease Rating Scale part III (MDS-UPDRS III) was calculated. The kinematic features of the patients’ movements in both the OFF and ON states were collected with an Azure Kinect depth camera.
Results
The IR of MDS-UPDRS III was significantly correlated with the IRs of many kinematic features for arising from a chair, pronation-supination movements of the hand, finger tapping, toe tapping, leg agility, and gait (rs = − 0.277 ~ − 0.672, P < 0.05). Moderate to high discriminative values were found in the selected features in identifying a clinically significant response to levodopa with sensitivity, specificity, and area under the curve (AUC) in the range of 50–100%, 47.22%–97.22%, and 0.673–0.915, respectively. The resulting classifier combining kinematic features of toe tapping showed an excellent performance with an AUC of 0.966 (95% CI = 0.922–1.000, P < 0.001). The optimal cut-off value was 21.24% with sensitivity and specificity of 94.44% and 87.18%, respectively.
Conclusion
This study demonstrated the feasibility of measuring the effect of levodopa and objectively assessing ALCT based on kinematic data derived from an Azure Kinect-based system.



Similar content being viewed by others
Data availability
The data used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Saranza G, Lang AE (2020) Levodopa challenge test: indications, protocol, and guide. J Neurol 268:3135–3143
Goetz CG, Tilley BC, Shaftman SR, Stebbins GT, Fahn S, Martinez-Martin P, Poewe W, Sampaio C, Stern MB, Dodel R, Dubois B, Holloway R, Jankovic J, Kulisevsky J, Lang AE, Lees A, Leurgans S, LeWitt PA, Nyenhuis D, Olanow CW, Rascol O, Schrag A, Teresi JA, van Hilten JJ, LaPelle N (2008) Movement disorder society-sponsored revision of the unified Parkinson’s disease rating scale (MDS-UPDRS): scale presentation and clinimetric testing results. Mov Disord: Off J Mov Disord Soc 23:2129–2170
Postuma RB, Berg D, Stern M, Poewe W, Olanow CW, Oertel W, Obeso J, Marek K, Litvan I, Lang AE, Halliday G, Goetz CG, Gasser T, Dubois B, Chan P, Bloem BR, Adler CH, Deuschl G (2015) MDS clinical diagnostic criteria for Parkinson’s disease. Mov Disord: Off J Mov Disord Soc 30:1591–1601
Lang AE, Widner H (2002) Deep brain stimulation for Parkinson’s disease: patient selection and evaluation. Mov Disord: Off J Mov Disord Soc 17(Suppl 3):S94-101
Zheng Z, Yin Z, Zhang B, Fan H, Liu D, Zhou Y, Duan J, Zhou D, Wu X, Lu G (2021) Levodopa challenge test predicts STN-DBS outcomes in various Parkinson’s disease motor subtypes: a more accurate judgment. Neural Plast 2021:4762027
Fabbri M, Coelho M, Abreu D, Guedes LC, Rosa MM, Costa N, Antonini A, Ferreira JJ (2016) Do patients with late-stage Parkinson’s disease still respond to levodopa? Parkinsonism Relat Disord 26:10–16
Fabbri M, Coelho M, Guedes LC, Chendo I, Sousa C, Rosa MM, Abreu D, Costa N, Godinho C, Antonini A, Ferreira JJ (2017) Response of non-motor symptoms to levodopa in late-stage Parkinson’s disease: results of a levodopa challenge test. Parkinsonism Relat Disord 39:37–43
Khodakarami H, Ricciardi L, Contarino MF, Pahwa R, Lyons KE, Geraedts VJ, Morgante F, Leake A, Paviour D, De Angelis A, Horne M (2019) Prediction of the levodopa challenge test in Parkinson’s disease using data from a wrist-worn sensor. Sensors (Basel) 19:5153
Lee WL, Sinclair NC, Jones M, Tan JL, Proud EL, Peppard R, McDermott HJ, Perera T (2019) Objective evaluation of bradykinesia in Parkinson’s disease using an inexpensive marker-less motion tracking system. Physiol Meas 40:014004
Defer GL, Widner H, Marié RM, Rémy P, Levivier M (1999) Core assessment program for surgical interventional therapies in Parkinson’s disease (CAPSIT-PD). Mov Disord: Off J Mov Disord Soc 14:572–584
Clark RA, Mentiplay BF, Hough E, Pua YH (2019) Three-dimensional cameras and skeleton pose tracking for physical function assessment: a review of uses, validity, current developments and Kinect alternatives. Gait Posture 68:193–200
Williams S, Zhao Z, Hafeez A, Wong DC, Relton SD, Fang H, Alty JE (2020) The discerning eye of computer vision: can it measure Parkinson’s finger tap bradykinesia? J Neurol Sci 416:117003
Zhan A, Mohan S, Tarolli C, Schneider RB, Adams JL, Sharma S, Elson MJ, Spear KL, Glidden AM, Little MA, Terzis A, Dorsey ER, Saria S (2018) Using smartphones and machine learning to quantify Parkinson disease severity: the mobile Parkinson disease score. JAMA Neurol 75:876–880
Álvarez I, Latorre J, Aguilar M, Pastor P, Llorens R (2020) Validity and sensitivity of instrumented postural and gait assessment using low-cost devices in Parkinson’s disease. J NeuroEng Rehab 17:149
Dranca L, de Abetxuko Ruiz de Mendarozketa L, Goñi A, Illarramendi A, Gomez IN, Alvarado MD, Rodríguez-Oroz MC (2018) Using Kinect to classify Parkinson’s disease stages related to severity of gait impairment. BMC Bioinforma 19:471
Tolgyessy M, Dekan M, Chovanec L, Hubinsky P (2021) Evaluation of the Azure Kinect and its comparison to Kinect V1 and Kinect V2. Sensors (Basel) 21:413
Zhu Y, Lu W, Gan W, Hou W (2021) A contactless method to measure real-time finger motion using depth-based pose estimation. Comput Biol Med 131:104282
Wu Z, Gu H, Hong R, Xing Z, Zhang Z, Peng K, He Y, Xie L, Zhang J, Gao Y, Jin Y, Su X, Zhi H, Guan Q, Pan L, Jin L (2023) Kinect-based objective evaluation of bradykinesia in patients with Parkinson’s disease. Digit Health 9:20552076231176652
Albanese A, Bonuccelli U, Brefel C, Chaudhuri KR, Colosimo C, Eichhorn T, Melamed E, Pollak P, Van Laar T, Zappia M (2001) Consensus statement on the role of acute dopaminergic challenge in Parkinson’s disease. Mov Disord: Off J Mov Disord Soc 16:197–201
Schade S, Sixel-Döring F, Ebentheuer J, Schulz X, Trenkwalder C, Mollenhauer B (2017) Acute levodopa challenge test in patients with de novo Parkinson’s disease: data from the DeNoPa cohort. Mov Disord Clin Pract 4:755–762
Wu Z, Hong R, Li S, Peng K, Lin A, Gao Y, Jin Y, Su X, Zhi H, Guan Q, Pan L, Jin L (2022) Technology-based therapy-response evaluation of axial motor symptoms under daily drug regimen of patients with Parkinson’s disease. Front Aging Neurosci 14:901090
Aghanavesi S, Bergquist F, Nyholm D, Senek M, Memedi M (2020) Motion sensor-based assessment of Parkinson’s disease motor symptoms during leg agility tests: results from levodopa challenge. IEEE J Biomed Health Inform 24:111–119
ZhuParris A, Thijssen E, Elzinga WO, Makai-Bölöni S, Kraaij W, Groeneveld GJ, Doll RJ (2023) Treatment detection and movement disorder society-unified Parkinson’s disease rating scale, part III estimation using finger tapping tasks. Mov Disord: Off J Mov Disord Soc 38:1795–1805
Thomas I, Westin J, Alam M, Bergquist F, Nyholm D, Senek M, Memedi M (2018) A treatment-response index from wearable sensors for quantifying Parkinson’s disease motor states. IEEE J Biomed Health Inform 22:1341–1349
Vaillancourt DE, Prodoehl J, Sturman MM, Bakay RA, Metman LV, Corcos DM (2006) Effects of deep brain stimulation and medication on strength, bradykinesia, and electromyographic patterns of the ankle joint in Parkinson’s disease. Mov Disord: Off J Mov Disord Soc 21:50–58
Buongiorno D, Bortone I, Cascarano GD, Trotta GF, Brunetti A, Bevilacqua V (2019) A low-cost vision system based on the analysis of motor features for recognition and severity rating of Parkinson’s disease. BMC Med Inform Decis Mak 19:243
Rovini E, Maremmani C, Cavallo F (2020) A wearable system to objectify assessment of motor tasks for supporting Parkinson’s disease diagnosis. Sensors (Basel, Switzerland) 20:2630
Di Lazzaro G, Ricci M, Al-Wardat M, Schirinzi T, Scalise S, Giannini F, Mercuri NB, Saggio G, Pisani A (2020) Technology-based objective measures detect subclinical axial signs in untreated, de novo Parkinson’s disease. J Parkinson’s Dis 10:113–122
Li MH, Mestre TA, Fox SH, Taati B (2018) Vision-based assessment of parkinsonism and levodopa-induced dyskinesia with pose estimation. J Neuroeng Rehabil 15:97
Parisi F, Ferrari G, Giuberti M, Contin L, Cimolin V, Azzaro C, Albani G, Mauro A (2015) Body-sensor-network-based kinematic characterization and comparative outlook of UPDRS scoring in leg agility, sit-to-stand, and gait tasks in Parkinson’s disease. IEEE J Biomed Health Inform 19:1777–1793
Richards M, Marder K, Cote L, Mayeux R (1994) Interrater reliability of the unified Parkinson’s disease rating scale motor examination. Mov Disord: Off J Mov Disord Soc 9:89–91
Goetz CG, Stebbins GT (2004) Assuring interrater reliability for the UPDRS motor section: utility of the UPDRS teaching tape. Mov Disord: Off J Mov Disord Soc 19:1453–1456
Fling BW, Cohen RG, Mancini M, Nutt JG, Fair DA, Horak FB (2013) Asymmetric pedunculopontine network connectivity in parkinsonian patients with freezing of gait. Brain: J Neurol 136:2405–2418
Maillet A, Pollak P, Debu B (2012) Imaging gait disorders in parkinsonism: a review. J Neurol Neurosurg Psychiatry 83:986–993
Nobrega-Sousa P, Gobbi LTB, Orcioli-Silva D, Conceicao NRD, Beretta VS, Vitorio R (2020) Prefrontal cortex activity during walking: effects of aging and associations with gait and executive function. Neurorehabil Neural Repair 34:915–924
Cavallieri F, Budriesi C, Gessani A, Contardi S, Fioravanti V, Menozzi E, Pinto S, Moro E, Valzania F, Antonelli F (2020) Dopaminergic treatment effects on dysarthric speech: acoustic analysis in a cohort of patients with advanced Parkinson’s disease. Front Neurol 11:616062
Curtze C, Nutt JG, Carlson-Kuhta P, Mancini M, Horak FB (2015) Levodopa Is a double-edged sword for balance and gait in people with Parkinson’s disease. Mov Disord: Off J Mov Disord Soc 30:1361–1370
Fabbri M, Guimaraes I, Cardoso R, Coelho M, Guedes LC, Rosa MM, Godinho C, Abreu D, Goncalves N, Antonini A, Ferreira JJ (2017) Speech and voice response to a levodopa challenge in late-stage Parkinson’s disease. Front Neurol 8:432
Sethi K (2008) Levodopa unresponsive symptoms in Parkinson disease. Mov Disord: Off J Mov Disord Soc 23(Suppl 3):S521-533
Rabel C, Le Goff F, Lefaucheur R, Ozel G, Fetter D, Rouille A, Maltete D (2016) Subjective perceived motor improvement after acute levodopa challenge in Parkinson’s disease. J Parkinson’s Dis 6:779–785
Davidson MB, McGhee DJ, Counsell CE (2012) Comparison of patient rated treatment response with measured improvement in Parkinson’s disease. J Neurol Neurosurg Psychiatry 83:1001–1005
Acknowledgements
We would like to thank all the participants for their understanding and participation.
Funding
This study was supported by the (1) Clinical Technology Innovation Project of Shanghai Shenkang Hospital Development Center (SHDC12020119); (2) Shanghai outstanding academic leaders’ plan of Shanghai Municipal Science and Technology Commission (20XD1403400); (3) National Clinical Key Specialty Construction Project of China (Z155080000004); (4) Shanghai Rehabilitation Medical Research Center (Top Priority Research Center of Shanghai) (2023ZZ02027); (5) Shanghai Clinical Research Ward (SHDC2023CRW018B); and (6) Shanghai Hospital Development Center Foundation—Shanghai Municipal Hospital Rehabilitation Medicine Specialty Alliance (SHDC22023304).
Author information
Authors and Affiliations
Contributions
LJJ and LZP conceived and designed the study with the help of QG. XYS, YJ, YCG, and HPZ developed the algorithms. Data acquisition and analysis were done by KWP, JXZ, YJH, ZYZ, RHH, and ZW. RHH and ZW were major contributors in writing the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethical approval
All procedures performed in this study involving human participants were in accordance with the Ethical Standards of the Institutional Research Committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The study was approved by the Ethics Committee of Shanghai Tongji Hospital (IRB no. 2019–061).
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent for publication
The participants have consented to the submission of their data to the journal.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Ronghua Hong and Zhuang Wu contributed equally to this work.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hong, R., Wu, Z., Peng, K. et al. Kinect-based objective assessment of the acute levodopa challenge test in parkinsonism: a feasibility study. Neurol Sci 45, 2661–2670 (2024). https://doi.org/10.1007/s10072-023-07296-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-023-07296-5