Abstract
Background
Parkinson's disease (PD) is the second most prevalent degenerative disease globally. While observational studies have demonstrated a correlation between thyroid function and PD, the causal relationship between these two factors remains uncertain.
Methods
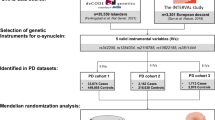
A bidirectional Mendelian randomization (MR) analysis was performed to explore the causal relationship between thyroid function (free thyroxine [FT4], thyroid-stimulating hormone [TSH], hyperthyroidism, and hypothyroidism) and PD. GWAS summary-level statistics of thyroid function and PD were obtained from publicly available GWAS databases. The inverse variance weighted method was the main MR approach to assess causal associations. In addition, two additional MR methods (MR-Egger regression and weighted median) were performed to supplement the IVW. Furthermore, various sensitivity tests were performed to verify the reliability of the MR findings: (i) Heterogeneity was examined by Cochrane's Q test. (ii) Horizontal pleiotropy was assessed by the MR-Egger intercept test and MR-PRESSO global test. (iii) The robustness of MR results was estimated using the leave-one-out method.
Results
Various MR results showed that FT4, TSH, hyperthyroidism, and hypothyroidism did not causally affect PD (P > 0.05). Likewise, PD did not causally affect FT4, TSH, hyperthyroidism, and hypothyroidism (P > 0.05). Cochrane's Q test indicated that MR analysis was not affected by significant heterogeneity (P > 0.05). MR-Egger intercept test and MR-PRESSO global test indicated that MR analysis was not affected by a remarkable horizontal pleiotropy (P > 0.05). The leave-one-out method demonstrated the stability of MR results.
Conclusion
MR analysis did not support a causal relationship between thyroid function and PD.

Similar content being viewed by others
Data availability
The original contributions presented in this study are included in the article/Supplementary material.
References
Poewe W, Seppi K, Tanner CM, Halliday GM, Brundin P, Volkmann J, Schrag AE, Lang AE (2017) Parkinson disease. Nat Rev Dis Primers 3:17013. https://doi.org/10.1038/nrdp.2017.13
Armstrong MJ, Okun MS (2020) Diagnosis and Treatment of Parkinson Disease: A Review. JAMA 323:548–560. https://doi.org/10.1001/jama.2019.22360
Ascherio A, Schwarzschild MA (2016) The epidemiology of Parkinson’s disease: risk factors and prevention. Lancet Neurol 15:1257–1272. https://doi.org/10.1016/s1474-4422(16)30230-7
Vázquez-Vélez GE, Zoghbi HY (2021) Parkinson’s Disease Genetics and Pathophysiology. Annu Rev Neurosci 44:87–108. https://doi.org/10.1146/annurev-neuro-100720-034518
Stepien BK, Huttner WB (2019) Transport, Metabolism, and Function of Thyroid Hormones in the Developing Mammalian Brain. Front Endocrinol (Lausanne) 10:209. https://doi.org/10.3389/fendo.2019.00209
Schroeder AC, Privalsky ML (2014) Thyroid hormones, t3 and t4, in the brain. Front Endocrinol (Lausanne) 5:40. https://doi.org/10.3389/fendo.2014.00040
Charoenngam N, Rittiphairoj T, Ponvilawan B, Prasongdee K (2022) Thyroid Dysfunction and Risk of Parkinson’s Disease: A Systematic Review and Meta-Analysis. Front Endocrinol (Lausanne) 13:863281. https://doi.org/10.3389/fendo.2022.863281
Lee EH, Kim SM, Kim CH, Pagire SH, Pagire HS, Chung HY, Ahn JH, Park CH (2019) Dopamine neuron induction and the neuroprotective effects of thyroid hormone derivatives. Sci Rep 9:13659. https://doi.org/10.1038/s41598-019-49876-6
Villanueva I, Alva-Sánchez C, Pacheco-Rosado J (2013) The role of thyroid hormones as inductors of oxidative stress and neurodegeneration. Oxid Med Cell Longev 2013:218145. https://doi.org/10.1155/2013/218145
Henchcliffe C, Beal MF (2008) Mitochondrial biology and oxidative stress in Parkinson disease pathogenesis. Nat Clin Pract Neurol 4:600–609. https://doi.org/10.1038/ncpneuro0924
Chen SF, Yang YC, Hsu CY, Shen YC (2020) Risk of Parkinson’s disease in patients with hypothyroidism: A nationwide population-based cohort study. Parkinsonism Relat Disord 74:28–32. https://doi.org/10.1016/j.parkreldis.2020.04.001
Lin SR, Chen SF, Yang YC, Hsu CY, Shen YC (2021) Association between hyperthyroidism and risk of incident in Parkinson’s disease. Endocr Connect 10:13–20. https://doi.org/10.1530/ec-20-0554
Paternoster L, Tilling K, Davey Smith G (2017) Genetic epidemiology and Mendelian randomization for informing disease therapeutics: Conceptual and methodological challenges. PLoS Genet 13:e1006944. https://doi.org/10.1371/journal.pgen.1006944
Hu X, Zhao J, Lin Z, Wang Y, Peng H, Zhao H, Wan X, Yang C (2022) Mendelian randomization for causal inference accounting for pleiotropy and sample structure using genome-wide summary statistics. Proc Natl Acad Sci U S A 119:e2106858119. https://doi.org/10.1073/pnas.2106858119
Teumer A, Chaker L, Groeneweg S, Li Y, Di Munno C, Barbieri C, Schultheiss UT, Traglia M, Ahluwalia TS, Akiyama M et al (2018) Genome-wide analyses identify a role for SLC17A4 and AADAT in thyroid hormone regulation. Nat Commun 9:4455. https://doi.org/10.1038/s41467-018-06356-1
Nalls MA, Blauwendraat C, Vallerga CL, Heilbron K, Bandres-Ciga S, Chang D, Tan M, Kia DA, Noyce AJ, Xue A et al (2019) Identification of novel risk loci, causal insights, and heritable risk for Parkinson’s disease: a meta-analysis of genome-wide association studies. Lancet Neurol 18:1091–1102. https://doi.org/10.1016/s1474-4422(19)30320-5
VanderWeele TJ, TchetgenTchetgen EJ, Cornelis M, Kraft P (2014) Methodological challenges in mendelian randomization. Epidemiology 25:427–435. https://doi.org/10.1097/ede.0000000000000081
Pierce BL, Ahsan H, Vanderweele TJ (2011) Power and instrument strength requirements for Mendelian randomization studies using multiple genetic variants. Int J Epidemiol 40:740–752. https://doi.org/10.1093/ije/dyq151
Pagoni P, Dimou NL, Murphy N, Stergiakouli E (2019) Using Mendelian randomisation to assess causality in observational studies. Evid Based Ment Health 22:67–71. https://doi.org/10.1136/ebmental-2019-300085
Bowden J, Davey Smith G, Haycock PC, Burgess S (2016) Consistent Estimation in Mendelian Randomization with Some Invalid Instruments Using a Weighted Median Estimator. Genet Epidemiol 40:304–314. https://doi.org/10.1002/gepi.21965
Burgess S, Thompson SG (2017) Interpreting findings from Mendelian randomization using the MR-Egger method. Eur J Epidemiol 32:377–389. https://doi.org/10.1007/s10654-017-0255-x
Hemani, G, Zheng, J, Elsworth, B, Wade, KH, Haberland, V, Baird, D, Laurin, C, Burgess, S, Bowden, J, Langdon, R et al. (2018) The MR-Base platform supports systematic causal inference across the human phenome. Elife 7. https://doi.org/10.7554/eLife.34408
Verbanck M, Chen CY, Neale B, Do R (2018) Detection of widespread horizontal pleiotropy in causal relationships inferred from Mendelian randomization between complex traits and diseases. Nat Genet 50:693–698. https://doi.org/10.1038/s41588-018-0099-7
Kim JH, Lee HS, Ahn JH, Oh JK, Chang IB, Song JH, Wee JH, Min CY, Yoo DM, Choi HG (2021) Association Between Thyroid Diseases and Parkinson’s Disease: A Nested Case-Control Study Using a National Health Screening Cohort. J Parkinsons Dis 11:211–220. https://doi.org/10.3233/jpd-202265
Lee K, Lim CY (2019) Mendelian Randomization Analysis in Observational Epidemiology. J Lipid Atherosclerosis 8:67–77. https://doi.org/10.12997/jla.2019.8.2.67
Xu J, Zhao C, Liu Y, Xu C, Qin B, Liang H (2022) Genetic correlation between thyroid hormones and Parkinson’s disease. Clin Exp Immunol 208:372–379. https://doi.org/10.1093/cei/uxac044
Acknowledgements
We thank the ThyroidOmics consortium, International Parkinson Disease Genomics Consortium, and IEU OpenGWAS project database for providing public access to GWAS summary statistics.
Funding
This research was funded by the National Natural Science Foundation of Hunan Province (2022JJ70069).
Author information
Authors and Affiliations
Contributions
Youjie Zeng: Conceptualization, Methodology, Validation, Investigation, Data Curation, Writing—Original Draft, Writing—Review & Editing, Visualization; Si Cao: Conceptualization, Methodology, Validation, Formal analysis, Investigation, Resources, Data Curation, Writing—Review & Editing; Heng Yang: Conceptualization, Methodology, Software, Validation, Formal analysis, Investigation, Supervision, Project administration, Funding acquisition.
Corresponding author
Ethics declarations
Ethical approval and Informed consent
The study utilized publicly available de-identified data from participant studies that had previously been approved by an ethics committee, thereby obviating the need for additional ethical approval and informed consent for this research.
Competing Interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zeng, Y., Cao, S. & Yang, H. No causal relationship between thyroid function and Parkinson’s disease: A bidirectional Mendelian randomization study. Neurol Sci 45, 1481–1487 (2024). https://doi.org/10.1007/s10072-023-07176-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-023-07176-y