Abstract
Objective
To compare the effectiveness of prolotherapy with dextrose concentrations of 5%, 10%, and 20% in patients diagnosed with knee osteoarthritis.
Methods
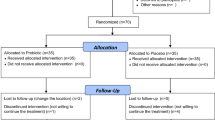
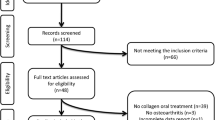
This study was planned as a prospective, randomized controlled interventional trial. Prolotherapy at 5% dextrose concentration in group 1, 10% in group 2, and 20% in group 3 was applied to the knee intra-articularly and periarticularly at 0, 3, and 6 weeks, and a home exercise program was given. Group 4 received a home exercise program. All groups received hotpack therapy at weeks 0, 3, and 6. Outcome measures included the visual analog scale (VAS), Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC), knee range of motion, timed up and go test, and Short Form-36.
Results
A total of 128 patients were divided into 4 groups. At the 6th and 12th weeks, VAS scores were significantly lower in groups 2 and 3 than in group 4 (p < 0.05). At the 12th week, the WOMAC pain score was significantly lower in group 3 than in group 4, and WOMAC physical function and WOMAC total scores were significantly lower in groups 1, 2, and 3 than in group 4 (p < 0.05). Week 6 active and passive knee flexion and week 12 passive knee flexion were significantly higher in group 3 than in group 4 (p < 0.05).
Conclusions
Although no significant difference was observed among the dextrose prolotherapy groups, higher dextrose concentrations demonstrated a greater improvement compared to the control group. Therefore, the use of 20% dextrose is recommended due to its significant superiority. Long-term follow-up and placebo-controlled studies are needed.
Trial registration
ClinicalTrial.gov Identifier: NCT05537077, registration date: 09.03.2022, retrospectively registered.
Key Points • The utilization of dextrose prolotherapy has gained popularity in the management of osteoarthritis, aiming to harness its regenerative and proliferative properties. However, the comparative efficacy of various concentrations of dextrose prolotherapy in treating knee osteoarthritis remains unexplored in the literature. This study aimed to address this gap by comparing different concentrations of dextrose prolotherapy in the treatment of knee osteoarthritis. The findings revealed no statistically significant difference among the various concentrations of dextrose prolotherapy for knee osteoarthritis treatment. |


Similar content being viewed by others
Data Availability
Authors may share data on reasonable request.
References
Cross M, Smith E, Hoy D, Nolte S, Ackerman I, Fransen M et al (2014) The global burden of hip and knee osteoarthritis: estimates from the global burden of disease 2010 study. Ann Rheum Dis 73(7):1323–1330. https://doi.org/10.1136/annrheumdis-2013-204763
Hsu H, Siwiec RM (2022) Knee Osteoarthritis. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan
Lespasio MJ, Piuzzi NS, Husni ME, Muschler GF, Guarino A, Mont MA (2017) Knee Osteoarthritis: A Primer. Perm J 21:16–183. https://doi.org/10.7812/TPP/16-183
Sharma L (2021) Osteoarthritis of the knee. N Engl J Med 384(1):51–59. https://doi.org/10.1056/NEJMcp1903768
Wu Y, Zhu F, Chen W, Zhang M (2022) Effects of transcutaneous electrical nerve stimulation (TENS) in people with knee osteoarthritis: a systematic review and meta-analysis. Clin Rehabil 36(4):472–485. https://doi.org/10.1177/02692155211065636
Bączkowicz D, Skiba G, Czerner M, Majorczyk E (2018) Gait and functional status analysis before and after total knee arthroplasty. Knee 25(5):888–896. https://doi.org/10.1016/j.knee.2018.06.004
Wee TC, Neo EJR, Tan YL (2021) Dextrose prolotherapy in knee osteoarthritis: a systematic review and meta-analysis. J Clin Orthop Trauma 19:108–117. https://doi.org/10.1016/j.jcot.2021.05.015
Hassan F, Trebinjac S, Murrell WD, Maffulli N (2017) The effectiveness of prolotherapy in treating knee osteoarthritis in adults: a systematic review. Br Med Bull 122(1):91–108. https://doi.org/10.1093/bmb/ldx006
Bernetti A, Agostini F, Alviti F, Giordan N, Martella F, Santilli V et al (2021) New viscoelastic hydrogel hymovis MO. RE. Single intra-articular injection for the treatment of knee osteoarthritis in sportsmen: safety and efficacy study results. Front Pharmacol 12:673988. https://doi.org/10.3389/fphar.2021.673988
Bae G, Kim S, Lee S, Lee WY, Lim Y (2021) Prolotherapy for the patients with chronic musculoskeletal pain: systematic review and meta-analysis. Anesth Pain Med 16(1):81–95. https://doi.org/10.17085/apm.20078
Rhatomy S, Margaretha E, Rahmadian R (2020) Dextrose prolotherapy for muscle, tendon and ligament injury or pathology: a systematic review. Ann Res Rev Biol 35(10):43–62. https://doi.org/10.9734/arrb/2020/v35i1030288
Akpancar S, Murat Seven M, Yasin Tuzun H, Gurer L, Ekinci S (2017) Current concepts of prolotherapy in orthopedic surgery. Arch Trauma Res 6(2):1–9. https://doi.org/10.5812/ATR.40447
Hauser RA, Lackner JB, Steilen-Matias D, Harris DK (2016) A systematic review of dextrose prolotherapy for chronic musculoskeletal pain. Clin Med Insights: Arthritis Musculoskelet Disord 9:CMAMD. S39160. https://doi.org/10.4137/CMAMD.S39160
Sit RWS, Wu RWK, Reeves KD, Rabago D, Chan DCC, Yip BHK et al (2018) Efficacy of intra-articular hypertonic dextrose prolotherapy versus normal saline for knee osteoarthritis: a protocol for a triple-blinded randomized controlled trial. BMC Complement Altern Med 18:1–8. https://doi.org/10.1186/s12906-018-2226-5
Dalton JA, McNaull F (1998) A call for standardizing the clinical rating of pain intensity using a 0 to 10 rating scale. Cancer Nurs 21(1):46–49. https://doi.org/10.1097/00002820-199802000-00006
Basaran S, Guzel R, Seydaoglu G, Guler-Uysal F (2010) Validity, reliability, and comparison of the WOMAC osteoarthritis index and Lequesne algofunctional index in Turkish patients with hip or knee osteoarthritis. Clin Rheumatol 29:749–756. https://doi.org/10.1007/s10067-010-1398-2
Podsiadlo D, Richardson S (1991) The timed “Up & Go”: a test of basic functional mobility for frail elderly persons. J Am Geriatr Soc 39(2):142–148. https://doi.org/10.1111/j.1532-5415.1991.tb01616.x
Ware Jr JE, Sherbourne CD (1992) The MOS 36-item short-form health survey (SF-36): I. Conceptual framework and item selection. Med Care 30(6):473–83
Güran Ş, Çoban ZD, Karasimav Ö, Demirhan S, Karaağaç N, Örsçelik A et al (2018) Dextrose solution used for prolotherapy decreases cell viability and increases gene expressions of angiogenic and apopitotic factors. Gulhane Med J 60(2):42–46
Billesberger LM, Fisher KM, Qadri YJ, Boortz-Marx RL (2020) Procedural treatments for knee osteoarthritis: a review of current injectable therapies. Pain Res Manag 3873098. https://doi.org/10.1155/2020/3873098
Farpour HR, Fereydooni F (2017) Comparative effectiveness of intra-articular prolotherapy versus peri-articular prolotherapy on pain reduction and improving function in patients with knee osteoarthritis: a randomized clinical trial. Electron Physician 9(11):5663. https://doi.org/10.19082/5663
Rezasoltani Z, Taheri M, Mofrad MK, Mohajerani SA (2017) Periarticular dextrose prolotherapy instead of intra-articular injection for pain and functional improvement in knee osteoarthritis. J Pain Res 10:1179–87. https://doi.org/10.2147/JPR.S127633
Chen Y-W, Lin Y-N, Chen H-C, Liou T-H, Liao C-D, Huang S-W (2022) Effectiveness, compliance, and safety of dextrose prolotherapy for knee osteoarthritis: a meta-analysis and metaregression of randomized controlled trials. Clin Rehabil 36(6):740–752. https://doi.org/10.1177/02692155221086213
Sit RW, Chung VC, Reeves KD, Rabago D, Chan KK, Chan DC et al (2016) Hypertonic dextrose injections (prolotherapy) in the treatment of symptomatic knee osteoarthritis: a systematic review and meta-analysis. Sci Rep 6(1):25247. https://doi.org/10.1038/srep25247
Maniquis-Smigel L, Dean Reeves K, Jeffrey Rosen H, Lyftogt J, Graham-Coleman C, Cheng AL et al (2017) Short term analgesic effects of 5% dextrose epidural injections for chronic low back pain: a randomized controlled trial. Anesth Pain Med 7(1):e42550. https://doi.org/10.5812/aapm.42550
Reeves KD, Sit RW, Rabago DP (2016) Dextrose prolotherapy: a narrative review of basic science, clinical research, and best treatment recommendations. Phys Med Rehabil Clin 27(4):783–823. https://doi.org/10.1016/j.pmr.2016.06.001
Topol GA, Podesta LA, Reeves KD, Giraldo MM, Johnson LL, Grasso R et al (2016) Chondrogenic effect of intra-articular hypertonic-dextrose (prolotherapy) in severe knee osteoarthritis. PM&R 8(11):1072–1082. https://doi.org/10.1016/j.pmrj.2016.03.008
Sert AT, Sen EI, Esmaeilzadeh S, Ozcan E (2020) The effects of dextrose prolotherapy in symptomatic knee osteoarthritis: a randomized controlled study. J Altern Complement Med 26(5):409–417. https://doi.org/10.1089/acm.2019.0335
Rabago D, Patterson JJ, Mundt M, Kijowski R, Grettie J, Segal NA et al (2013) Dextrose prolotherapy for knee osteoarthritis: a randomized controlled trial. Ann Family Med 11(3):229–237. https://doi.org/10.1370/afm.1504
Dumais R, Benoit C, Dumais A, Babin L, Bordage R, de Arcos C et al (2012) Effect of regenerative injection therapy on function and pain in patients with knee osteoarthritis: a randomized crossover study. Pain Med 13(8):990–999. https://doi.org/10.1111/j.1526-4637.2012.01422.x
Reeves KD, Hassanein K (2000) Randomized prospective double-blind placebo-controlled study of dextrose prolotherapy for knee osteoarthritis with or without ACL laxity. Altern Ther Health Med 6(2):68–80
Soliman D, Sherif N, Omar O, El Zohiery A (2016) Healing effects of prolotherapy in treatment of knee osteoarthritis healing effects of prolotherapy in treatment of knee osteoarthritis. Egypt Rheumatol Rehabil 43:47–52. https://doi.org/10.4103/1110-161X.181858
Mustafa R, Güngörmüs M, Mollaoglu N (2018) Evaluation of the efficacy of different concentrations of dextrose prolotherapy in temporomandibular joint hypermobility treatment. J Craniofac Surg 29(5):e461–e465. https://doi.org/10.1097/SCS.0000000000004480
Ciftci YGD, Tuncay F, Kocak FA, Okcu M (2023) Is low-dose dextrose prolotherapy as effective as high-dose dextrose prolotherapy in the treatment of lateral epicondylitis? A double-blind, ultrasound guided, randomized controlled study. Arch Phys Med Rehabil 104(2):179–187. https://doi.org/10.1016/j.apmr.2022.09.017
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Conceptualization, data curation, funding acquisition, methodology, investigation, project administration, resources, visualization, and writing—original draft were performed by Muhammet Uğur Öztürk. Formal analysis, supervision, software, validation, writing—review, and editing were performed by Fatih Baygutalp. The first draft of the manuscript was written by Muhammet Uğur Öztürk. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical statement
The Atatürk University Faculty of Medicine Ethics Committee approved this study on February 27, 2020 (No: B.30.2ATA.0.01.00/104). The study was uploaded to Clinicaltrials.gov and received the ID code NCT05537077. This study was conducted in accordance with the principles of the Declaration of Helsinki.
Informed consent
After being informed about the study, written informed consent forms were acquired from each patient.
Disclosures
None.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Öztürk, M.U., Baygutalp, F. A comparative analysis of prolotherapy efficacy in patients with knee osteoarthritis across varied dextrose concentrations. Clin Rheumatol 42, 3321–3331 (2023). https://doi.org/10.1007/s10067-023-06723-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-023-06723-4