Abstract
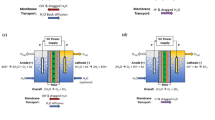
Nanoporous anatase xerogels were prepared via particulate sol–gel processes. The calcined xerogels were mesoporous, with a BET surface area of 121 m2/g, an average pore diameter of 5.8 nm and a pore volume of 0.236 cc/g. Proton conductivity of the membranes was measured as a function of temperature and relative humidity (RH). When anatase membranes are treated at pH 1.5, the proton conductivity increased in the whole range of temperature and RH. It indicates that the surface site density (number of water molecules per square nanometer) of these materials has a strong effect on conductivity. The proton conductivity of the studied anatase xerogels followed an Arrhenius-like dependence on the temperature (from room temperature to 90°C), in both treated and untreated membranes. A sigmoidal dependence of the conductivity on the RH was observed with the greatest increase noted between 58% and 81% RH in both treated and untreated anatase membranes. The highest value of proton conductivity was found to be 0.015 S/cm at 90°C and 81% RH, for treated anatase ceramic membranes. An increase in the conductivity could be achieved by means of longer times of treatment. According to the activation energy values, proton migration in this kind of materials could be dominated by the Grotthuss mechanism in the whole range of RH. The similar values of proton conductivity, lower cost and higher hydrophilicity of these membranes make them potential substitutes for perfluorosulfonic polymeric membranes in proton exchange membrane fuel cells (PEMFCs).







Similar content being viewed by others
References
Kreuer KD (1997) Solid State Ionics 97:1
Gruger A, Régis A, Schmatko T, Colomban Ph (2001) Vibrational Spectrosc 26:215
Nogami M, Nagao R, Wong C (1998) J Phys Chem B 102:5772
Colomer MT, Anderson MA (2001) J Non Cryst Solids 290:93
Matsuda A, Kanzaki T, Tadanaga K, Tatsumisago M, Minami T (2001) Electrochim Acta 47:939
Anappara AA, Rajeshkumar S, Mukundan P, Warrier PRS, Ghosh S, Warrier KGK (2004) Acta Mater 52:369
Mioc UB, Milonjic SK, Stamenkovic V, Radojevic M, Colomban Ph, Mitrovic MM, Dimitrijevic R (1999) Solid State Ionics 125:417
Mioc UB, Milonjic SK, Malovic D, Stamenkovic V, Colomban Ph, Mitrovic MM, Dimitrijevic R (1997) Solid State Ionics 97:239
Lowell S, Shields JE (1991) Powder surface area and porosity. Chapman and Hall, London
Sing KSW, Everett DH, Haul RAW, Moscou L, Pierotti RA, Rouquerol J, Siemieniewska T (1985) Pure Appl Chem 57:603
Barret EP, Joyney LG, Halenda PP (1951) J Am Chem Soc 73:373
Seaton NA (1991) Chem Eng Sci 46:1895
Nogami M, Matsushita H, Kasuga T, Hayakawa T (1999) Electrochem Solid State Lett 2:415
Lide DR (ed-in-Chief) (2003–2004) Handbook of chemistry and physics, 84th edn. CRC, Boca Raton
Boukamp B, Equivalent circuit (EQUIVCRT.PAS) (1988–89) University of Twente, Twente
Gregg SJ, Sing KSW (1982) Adsorption, surface area porosity. Academic, London
Vendange V, Colomban Ph (1996) J Porous Mater 3:193
Salame II, Baagrev A, Bandosz TJ (1999) J Phys Chem B 103:3877
McCallum C-L, Bandosz TJ, McGrother SC, Müller EA, Gubbins KE (1996) Langmuir 12:533
Dubinin MM, Serpinsky W (1966) J Colloid Interface Sci 21:378
Colomban P, Novak A (1992) In: Colomban P (ed) Proton conductors. Cambridge University Press, Cambridge, pp 38–55, 384–387
Kreuer KD, Stoll I, Rabenau A (1983) Solid State Ionics 9–10:1061
Zawodzisnki TA Jr, Gottesfeld S (1992) The electrochemical society extended abstracts, Toronto, pp 11–16 (Abstract 94)
Marrink SJ, Berkowitz M, Berendsen HJC (1993) Langmuir 9:3122
Agmon NJ (1996) Chim Phys et de Physiquo Chim Biolog 93:1714
Acknowledgments
This work is in the frame of the contracts CAM 07N/0102/2002 and CICYT MAT2002-00250.
Author information
Authors and Affiliations
Corresponding author
Additional information
Presented at the conference Solid State Chemistry 2004, September 13–17, Prague, Czech Republic
Rights and permissions
About this article
Cite this article
Colomer, M.T. Proton conductivity of nanoporous anatase xerogels prepared by a particulate sol–gel method. J Solid State Electrochem 10, 54–59 (2006). https://doi.org/10.1007/s10008-005-0662-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-005-0662-x