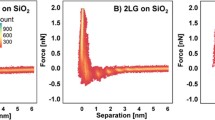
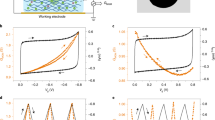
Abstract
Atomistic molecular dynamics simulations were used to investigate the processes of electrical double layer formation and electrolyte confinement in graphene-based supercapacitors. For both processes, free energy calculations were used to analyze the thermodynamics involved in the electrolyte confinement and its re-arrangement in a double layer on the electrode surface. The value of the free energy of the formation of the double electric layer was related to the energy required to charge the supercapacitor, i.e., the energy density stored, and compared with values obtained using Poisson’s electrostatic formalism, which is the conventionally employed approach. Both analyzes were consistent with each other, presenting compatible values for the stored energy.




Similar content being viewed by others
References
Zhou H, Rouha M, Feng G, Lee S, Docherty H, Fenter P, Cummings PT, Fulvio PF, Dai S, McDonough J, Presser V, Gogotsi Y (2012) Nanoscale perturbations of room temperature ionic liquid structure at charged and uncharged interfaces. ACS Nano 6(11):9818–9827. https://doi.org/10.1021/nn303355b
Péan C, Merlet C, Rotenberg B, Madden P, Taberna P-L, Daffos B, Salanne M, Simon P (2014) On the dynamics of charging in nanoporous carbon-based supercapacitors. ACS Nano 8(2):1576–1583. https://doi.org/10.1021/nn4058243
Kondrat S, Wu P, Qiao R, Kornyshev AA (2014) Accelerating charging dynamics in subnanometre pores. Nat. Mater. 13(4). https://doi.org/10.1038/nmat3916
Cl M, Limmer DT, Salanne M, van Roij R, Madden PA, Chandler D, Rotenberg B (2014) The electric double layer has a life of its own. J. Phys. Chem. C 118(32):18291–18298. https://doi.org/10.1021/jp503224w
Cl M, Péan C, Rotenberg B, Madden PA, Simon P, Salanne M (2013) Simulating supercapacitors: can we model electrodes as constant charge surfaces? The Journal of Physical Chemistry Letters 4(2):264–268. https://doi.org/10.1021/jz3019226
Hu Z, Vatamanu J, Borodin O, Bedrov D (2013) A molecular dynamics simulation study of the electric double layer and capacitance of [BMIM][PF 6] and [BMIM][BF 4] room temperature ionic liquids near charged surfaces. Phys. Chem. Chem. Phys. 15(34):14234–14247. https://doi.org/10.1039/C3CP51218E
Béguin F, Presser V, Balducci A, Frackowiak E (2014) Carbons and electrolytes for advanced supercapacitors. Adv. Mater. 26(14):2219–2251. https://doi.org/10.1002/adma.201304137
Jiang D-e WJ (2014) Unusual effects of solvent polarity on capacitance for organic electrolytes in a nanoporous electrode. Nanoscale 6(10):5545–5550. https://doi.org/10.1039/C4NR00046C
Chmiola J, Largeot C, Taberna PL, Simon P, Gogotsi Y (2008) Desolvation of ions in subnanometer pores and its effect on capacitance and double-layer theory. Angew. Chem. Int. Ed. 47(18):3392–3395. https://doi.org/10.1002/anie.200704894
Chabi S, Peng C, Hu D, Zhu Y (2014) Ideal three-dimensional electrode structures for electrochemical energy storage. Adv. Mater. 26(15):2440–2445. https://doi.org/10.1002/adma.201305095
Zhang C, Peng Z, Lin J, Zhu Y, Ruan G, Hwang C-C, Lu W, Hauge RH, Tour JM (2013) Splitting of a vertical multiwalled carbon nanotube carpet to a graphene nanoribbon carpet and its use in supercapacitors. ACS Nano 7(6):5151–5159. https://doi.org/10.1021/nn400750n
Krause A, Balducci A (2011) High voltage electrochemical double layer capacitor containing mixtures of ionic liquids and organic carbonate as electrolytes. Electrochem. Commun. 13(8):814–817. https://doi.org/10.1016/j.elecom.2011.05.010
Schütter C, Neale AR, Wilde P, Goodrich P, Hardacre C, Passerini S, Jacquemin J, Balducci A (2016) The use of binary mixtures of 1-butyl-1-methylpyrrolidinium bis{(trifluoromethyl)sulfonyl}imide and aliphatic nitrile solvents as electrolyte for supercapacitors. Electrochim. Acta 220:146–155. https://doi.org/10.1016/j.electacta.2016.10.088
Leach A (2001) Molecular modelling: principles and applications. Prentice Hall, New York
Darden T, York D, Pedersen L (1993) Particle mesh Ewald: an N·log(N) method for Ewald sums in large systems. J. Chem. Phys. 98:10089–10099
Chaban VV, Fileti EE, Prezhdo OV (2017) Exfoliation of graphene in ionic liquids: pyridinium versus pyrrolidinium. J. Phys. Chem. C 121(1):911–917. https://doi.org/10.1021/acs.jpcc.6b11003
Chaban VV, Fileti EE (2015) Graphene exfoliation in ionic liquids: unified methodology. RSC Adv. 5(99):81229–81234. https://doi.org/10.1039/c5ra16857k
Voroshylova IV, Chaban VV (2014) Atomistic force field for pyridinium-based ionic liquids: reliable transport properties. J. Phys. Chem. B 118(36):10716–10724. https://doi.org/10.1021/jp5054875
Canongia Lopes JN, Deschamps J, Pádua AAH (2004) Modeling ionic liquids using a systematic all-atom force field. J. Phys. Chem. B 108(6):2038–2047. https://doi.org/10.1021/jp0362133
Beutler TC, Mark AE, van Schaik RC, Greber PR, van Gunsteren WF (1994) Avoiding singularities and numerical instabilities in free energy calculations based on molecular simulations. Chem Phys Lett:222
Abraham MJ, van der Spoel D, Lindahl E, Hess B, and the Gromacs Development Team (2018) Gromacs user manual , http://www.gromacs.org
Martinez L, Andrade R, Birgin EG, Martinez JM (2009) PACKMOL: a package for building initial configurations for molecular dynamics simulations. J. Comput. Chem. 30(13):2157–2164. https://doi.org/10.1002/jcc.21224
Humphrey W, Dalke A, Schulten K (1996) VMD: visual molecular dynamics. J. Mol. Graph. 14(1):33–38. https://doi.org/10.1016/0263-7855(96)00018-5
Salanne M (2017) Ionic liquids for supercapacitor applications. Top. Curr. Chem. 375(3):63. https://doi.org/10.1007/s41061-017-0150-7
Xia L, Yu L, Hu D, Chen GZ (2017) Electrolytes for electrochemical energy storage. Materials Chemistry Frontiers 1(4):584–618. https://doi.org/10.1039/C6QM00169F
Zhan C, Lian C, Zhang Y, Thompson MW, Xie Y, Wu J, Kent PRC, Cummings PT, De J, Wesolowski DJ (2017) Computational insights into materials and interfaces for capacitive energy storage. Advanced Science 4(7):1700059. https://doi.org/10.1002/advs.201700059
Ke Q, Wang J (2016) Graphene-based materials for supercapacitor electrodes – a review. J. Mater. 2(1):37–54. https://doi.org/10.1016/j.jmat.2016.01.001
Theodoor J, Overbeek G (1990) The role of energy and entropy in the electrical double layer. Colloids and Surfaces 51:61–75. https://doi.org/10.1016/0166-6622(90)80132-N
Manciu M, Ruckenstein E (2003) On the chemical free energy of the electrical double layer. Langmuir 19(4):1114–1120. https://doi.org/10.1021/la0266132
Kornyshev AA (2007) Double-layer in ionic liquids: paradigm change? J. Phys. Chem. B 111(20):5545–5557. https://doi.org/10.1021/jp067857o
Burt R, Birkett G, Zhao XS (2014) A review of molecular modelling of electric double layer capacitors. Phys. Chem. Chem. Phys. 16(14):6519–6538. https://doi.org/10.1039/C3CP55186E
Bedrov D, Vatamanu J, Hu Z (2015) Ionic liquids at charged surfaces: insight from molecular simulations. J. Non-Cryst. Solids 407:339–348. https://doi.org/10.1016/j.jnoncrysol.2014.08.007
Haskins JB, Wu JJ, Lawson JW (2016) Computational and experimental study of li-doped ionic liquids at electrified interfaces. J. Phys. Chem. C 120(22):11993–12011. https://doi.org/10.1021/acs.jpcc.6b02449
Paek E, Pak AJ, Hwang GS (2013) A computational study of the interfacial structure and capacitance of graphene in [BMIM][PF6] ionic liquid. J. Electrochem. Soc. 160(1). https://doi.org/10.1149/2.019301jes
Lazzari M, Soavi F, Mastragostino M (2010) Mesoporous carbon design for ionic liquid-Based, double-layer supercapacitors. Fuel Cells 10(5):840–847. https://doi.org/10.1002/fuce.200900198
Lazzari M, Mastragostino M, Pandolfo AG, Ruiz V, Soavi F (2011) Role of carbon porosity and ion size in the development of ionic liquid based supercapacitors. J. Electrochem. Soc. 158(1). https://doi.org/10.1149/1.3514694
Acknowledgments
The author gratefully acknowledges support from FAPESP (São Paulo Research Foundation, grant number 2017/11631-2), Shell, the strategic importance of the support given by ANP (Brazil’s National Oil, Natural Gas and Biofuels Agency) through the R&D levy regulation and CNPq (National Council for Scientific and Technological Development).
Funding
This study is supported by FAPESP (São Paulo Research Foundation, grant number 2017/11631-2), Shell, the strategic importance of the support given by ANP (Brazil’s National Oil, Natural Gas and Biofuels Agency) through the R&D levy regulation and CNPq (National Council for Scientific and Technological Development).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This paper belongs to Topical Collection XX - Brazilian Symposium of Theoretical Chemistry (SBQT2019)
Rights and permissions
About this article
Cite this article
Fileti, E.E. Electric double layer formation and storing energy processes on graphene-based supercapacitors from electrical and thermodynamic perspectives. J Mol Model 26, 159 (2020). https://doi.org/10.1007/s00894-020-04428-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-020-04428-y