Abstract
Objectives
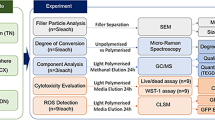
This study aims to evaluate the cytotoxicity and genotoxicity of three different extracts obtained from Filtek™ One Bulk Fill, Tetric Evoceram® Bulk Fill and Coltene Fill-Up! resins.
Materials and methods
The cytotoxicity was determined on 3T3 fibroblast cells using the MTT and crystal violet assays. The genotoxicity was determined using a cytokinesis-block micronucleus assay.
Results
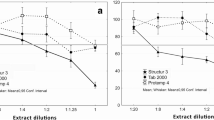
The cytotoxicity of the resin extracts on 3T3 mouse fibroblasts was found to be dose-dependent with both the MTT and crystal violet assays. Extracts concentrated above 1% were cytotoxic according to the MTT assay. The Filtek™ One Bulk Fill, Tetric Evoceram® Bulk Fill, and Coltene Fill-Up! resins reached the LD50 at concentrations of 60%, 50%, and 20%, respectively, and showed genotoxicity rates that were 2–5 times, 3–8 times, and 4–15 times higher than the negative control, respectively.
Conclusions
Coltene Fill-Up! resin extracts were the most cytotoxic and genotoxic, followed by Tetric Evoceram® Bulk Fill and Filtek™ One Bulk Fill.
Clinical relevance
The analyzed bulk-fill resins showed differences in in vitro biocompatibility, and the Filtek™ One Bulk Fill was found to be the safest for clinical use.




Similar content being viewed by others
References
Dionysopoulos D, Gerasimidou O (2021) Wear of contemporary dental composite resin restorations: a literature review. Restor Dent Endod 46(2):1–13. https://doi.org/10.5395/rde.2021.46.e18
Meereis CTW, Münchow EA, Rosa WLO, Silva AF, Piva E (2018) Polymerization shrinkage stress of resin-based dental materials: a systematic review and meta-analyses of composition strategies. J Mech Behav Biomed Mater 82:268–281. https://doi.org/10.1016/j.jmbbm.2018.03.019
Małkiewicz K, Wychowański P, Olkowska-Truchanowicz J, Tykarska M, Czerwiński M, Wilczko M, Owoc A (2017) Uncompleted polymerization and cytotoxicity of dental restorative materials as potential health risk factors. Ann Agric Environ Med 24(4):618–623. https://doi.org/10.5604/12321966.1235159
Cender EU, Guler C, Odabasi D (2021) The effects of polymerization mode and layer thickness on monomer released from bulk fill composite resins. Niger J Clin Pract 24(10):1442–1449
Rizzante FAP, Mondelli RFL, Furuse AY, Borges AFS, Mendonça G, Ishikiriama SK (2019) Shrinkage stress and elastic modulus assessment of bulk-fill composites. J Appl Oral Sci 27:1–9. https://doi.org/10.1590/1678-7757-2018-0132
Moura AGSM, Melo MGRR, Santos WTNP, Bezerra ALCA, Monteiro GQM, Durão MA (2021) An in vitro evaluation of the color stability of bulk-fill resin composites. Arch Health Invest 10(4):530–535. https://doi.org/10.21270/archi.v10i4.4973
Tauböck TT, Jäger F, Attin T (2018) Polymerization shrinkage and shrinkage force kinetics of high- and low-viscosity dimethacrylate- and ormocerbased bulk-fill resin composites. Odontology 107(1):103–110. https://doi.org/10.1007/s10266-018-0369-y
Gonçalves F, Campos LMP, Rodrigues-Júnior EC, Costa FV, Marques PA, Francci CE, Braga RR, Boaro LCC (2018) A comparative study of bulk-fill composites: degree of conversion, post-gel shrinkage and cytotoxicity. Braz Oral Res 32(17):1–9. https://doi.org/10.1590/1807-3107bor-2018.vol32.0017
Tauböck TT, Marovic D, Zeljezic D, Steingruber AD, Attin T, Tarle Z (2017) Genotoxic potential of dental bulk-fill resin composites. Dent Mater 33(7):788–795. https://doi.org/10.1016/j.dental.2017.04.011
Haugen HJ, Marovic D, Par M, Thieu MKL, Reseland JE, Johnsen GF (2020) Bulk fill composites have similar performance to conventional dental composites. Int J Mol Sci 21(14):1–21. https://doi.org/10.3390/ijms21145136
Tardem C, Albuquerque EG, Lopes LS, Marins SS, Calazans FS, Poubel LA, Barcelos R, Barceleiro MO (2019) Clinical time and postoperative sensitivity after use of bulk-fill (syringe and capsule) vs. incremental filling composites: a randomized clinical trial. Res Braz Oral 33(0):1–13. https://doi.org/10.1590/1807-3107bor-2019.vol33.0089
Huang B, Sadeghinejad L, Adebayo OIA, Ma D, Xiao Y, Siqueira WL, Cvitkovitch DG, Finer Y (2018) Gene expression and protein synthesis of esterase from Streptococcus mutans are affected by biodegradation by-product from methacrylate resin composites and adhesives. Acta Biomater 81:158–168. https://doi.org/10.1016/j.actbio.2018.09.050
Girão LB, Martins JOL, Lemos JVM, Pinto MR, Rolim JPML, Silva FCFA, Saboia VPA, Sousa FB, Silva PGB (2020) Influence of the degree of conversion and Bis-GMA residues of bulk fill resins on tissue toxicity in na subcutaneous model in rats. J Appl Biomater Funct Mater 18:1–10. https://doi.org/10.1177/2280800020947330
Maalekipour M, Safari M, Barekatain M, Fathi A (2021) Effect of adhesive resin as a modeling liquid on elution of resin composite restorations. Int J Dent 3178536:1–9. https://doi.org/10.1155/2021/3178536
Paolone G, Madurino M, Scotti N, Cantatore G, Blatz MB (2023) Color stability of bulk-fill compared to conventional resin-based composites: a scoping review. J Esthet Restor Dent 35(4):657–676. https://doi.org/10.1111/jerd.13017
Demirel G, Gür G, Demirsoy FF, Altuntas EG, Yener-Ílce B, Kiliçarslan MA (2020) Cytotoxic effects of contemporary bulk-fill dental composites: a real-time cell analysis. Dent Mater J 39(1):101–110. https://doi.org/10.4012/dmj.2018-336
Nascimento AS, Lima DB, Fook MVL, Albuquerque MS, Lima EA, Sabino MA, Borges SMP, Filgueira PTD, Sousa YC, Braz R (2018) Physicomechanical characterization and biological evaluation of bulk-fill composite resin. Braz Oral Res 32(107):1–14. https://doi.org/10.1590/1807-3107bor-2018.vol32.0107
Schweikl H, Spagnuolo G, Schmalz G (2006) Genetic and cellular toxicology of dental resin monomers. J Dent Res 85(10):870–877. https://doi.org/10.1177/154405910608501001
International Organization for Standardization (ISO) (2009) 10993-5: biological evaluation of medical devices. In: Part 5: Tests for in vitro cytotoxicity. Geneva: International Organization for Standardization. https://www.iso.org/standard/36406.html/#stages
International Organization for Standardization (ISO) (2021) 10993-12: biological evaluation of medical devices. In: Part 12: Sample preparation and reference materials. Geneva: International Organization for Standardization. https://www.iso.org/standard/75769.html
Bandarra S, Mascarenhas P, Luís AR, Catrau M, Bekman E, Ribeiro AC, Félix S, Caldeira J, Barahona I (2020) In vitro and in silico evaluations of resin-based dental restorative material toxicity. Clin Oral Investig 24(8):2691–2700. https://doi.org/10.1007/s00784-019-03131-4
Bandarra S, Fernandes AS, Magro I, Guerreiro PS, Pingarilho M, Churchwell MI, Gil OM, Batinic-Haberle I, Goncalves S, Rueff J, Miranda JP, Marques MM, Beland FA, Castro M, Gaspar JF, Oliveira NG (2013) Mechanistic insights into the cytotoxicity and genotoxicity induced by glycidamide in human mammary cells. Mutagenesis 28(6):721–729. https://doi.org/10.1093/mutage/get052
Lee SM, Kim SY, Kim JH, Jun SK, Kim HW, Lee JH, Lee HH (2019) Depth-dependent cellular response from dental bulk-fill resins in human dental pulp stem cells. Stem Cells Int 1251536:111. https://doi.org/10.1155/2019/1251536
Chaharom MEE, Bahari M, Safyari L, Safarvand H, Shafaei H, Navimipour EJ, Oskoee PA, Ajami AA, Kahnamouei MA (2020) Effect of preheating on the cytotoxicity of bulk-fill composite resins. J Dent Res Dent Clin Dent Prospects 14(1):19–25. https://doi.org/10.34172/joddd.2020.003
Kamalak H, Kamalak A, Taghizadehghalehjoughi A, Hacımüftüoğlu A, Nalci KA (2018) Cytotoxic and biological effects of bulk fill composites on rat cortical neuron cells. Odontology 106(4):377–388. https://doi.org/10.1007/s10266-018-0354-5
Lempel E, Czibulya Z, Kovács B, Szalma J, Tóth Á, Kunsági-Máté S, Varga Z, Böddi K (2016) Degree of conversion and BisGMA, TEGDMA, UDMA elution from flowable bulk fill compo-sites. Int J Mol Sci 17(5):1–13. https://doi.org/10.3390/ijms17050732
Rincón G, Sánchez C (2021) Short assay design for micronucleus detection in human lymphocytes. BioMed Res Int 322257:1–6. https://doi.org/10.1155/2021/2322257
Sommer S, Buraczewska I, Kruszewski M (2020) Micronucleus assay: the state of art, and future directions. Int J Mol Sci 21(4):1–19. https://doi.org/10.3390/ijms21041534
Bandarra S, Neves J, Paraíso A, Mascarenhas P, Ribeiro AC, Barahona I (2021) Biocompatibility of self-adhesive resin cement with fibroblast cells. J Prosthet Dent 125(4):705.e1-705.e7. https://doi.org/10.1016/j.prosdent.2021.01.0022
Funding
Egas Moniz CRL funded this research.
Author information
Authors and Affiliations
Contributions
C. J. and I. B. wrote the main manuscript and P. M. and M. A. prepared figures. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Informed consent
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Junqueira, C., Mascarenhas, P., Avelar, M. et al. Biocompatibility of bulk-fill resins in vitro. Clin Oral Invest 27, 7851–7858 (2023). https://doi.org/10.1007/s00784-023-05376-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-023-05376-6