Abstract
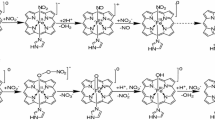
Many heme proteins undergo covalent attachment of the heme group to a protein side chain. Such posttranslational modifications alter the thermodynamic and chemical properties of the holoprotein. Their importance in biological processes makes them attractive targets for mechanistic studies. We have proposed a reductively driven mechanism for the covalent heme attachment in the monomeric hemoglobins produced by the cyanobacteria Synechococcus sp. PCC 7002 and Synechocystis sp. PCC 6803 (GlbN) (Nothnagel et al. in J Biol Inorg Chem 16:539–552, 2011). These GlbNs coordinate the heme iron with two axial histidines, a feature that distinguishes them from most hemoglobins and conditions their redox properties. Here, we uncovered evidence for an electron exchange chain reaction leading to complete heme modification upon substoichiometric reduction of GlbN prepared in the ferric state. The GlbN electron self-exchange rate constants measured by NMR spectroscopy were on the order of 102–103 M−1 s−1 and were consistent with the proposed autocatalytic process. NMR data on ferrous and ferric Synechococcus GlbN in solution indicated little dependence of the structure on the redox state of the iron or cross-link status of the heme group. This allowed the determination of lower bounds to the cross-exchange rate constants according to Marcus theory. The observations illustrate the ability of bishistidine hemoglobins to undergo facile interprotein electron transfer and the chemical relevance of such transfer for covalent heme attachment.









Similar content being viewed by others
Abbreviations
- ESE:
-
Electron self-exchange
- ET:
-
Electron transfer
- GlbN:
-
Hemoglobin produced by Synechococcus sp. PCC 7002 or Synechocystis sp. PCC 6803
- GlbN-A:
-
GlbN with covalently attached heme
- GlbN-R:
-
GlbN with noncovalently attached heme
- GODCAT:
-
Glucose oxidase/d-glucose/catalase
- HSQC:
-
Heteronuclear single quantum coherence
- PTM:
-
Posttranslational modification
References
Vinogradov SN, Moens L (2008) J Biol Chem 283:8773–8777
Kakar S, Sturms R, Tiffany A, Nix JC, DiSpirito AA, Hargrove MS (2011) Biochemistry 50:4273–4280
Scott NL, Falzone CJ, Vuletich DA, Zhao J, Bryant DA, Lecomte JTJ (2002) Biochemistry 41:6902–6910
Scott NL, Xu Y, Shen G, Vuletich DA, Falzone CJ, Li Z, Ludwig M, Pond MP, Preimesberger MR, Bryant DA, Lecomte JTJ (2010) Biochemistry 49:7000–7011
Couture M, Das TK, Savard PY, Ouellet Y, Wittenberg JB, Wittenberg BA, Rousseau DL, Guertin M (2000) Eur J Biochem 267:4770–4780
Scott NL, Lecomte JTJ (2000) Protein Sci 9:587–597
Vu BC, Jones AD, Lecomte JTJ (2002) J Am Chem Soc 124:8544–8545
Vu BC, Vuletich DA, Kuriakose SA, Falzone CJ, Lecomte JTJ (2004) J Biol Inorg Chem 9:183–194
Bowman SE, Bren KL (2008) Nat Prod Rep 25:1118–1130
Pearson AR, Elmore BO, Yang C, Ferrara JD, Hooper AB, Wilmot CM (2007) Biochemistry 46:8340–8349
Huang L, Wojciechowski G, Ortiz de Montellano PR (2006) J Biol Chem 281:18983–18988
Nothnagel HJ, Preimesberger MR, Pond MP, Winer BY, Adney EM, Lecomte JTJ (2011) J Biol Inorg Chem 16:539–552
Vuletich DA, Falzone CJ, Lecomte JTJ (2006) Biochemistry 45:14075–14084
Englander SW, Calhoun DB, Englander JJ (1987) Anal Biochem 161:300–306
Di Iorio EE (1981) Methods Enzymol 76:57–72
Johnson KA, Simpson ZB, Blom T (2009) Anal Biochem 387:30–41
Johnson KA (2009) Methods Enzymol 467:601–626
Bilsel O, Zitzewitz JA, Bowers KE, Matthews CR (1999) Biochemistry 38:1018–1029
Falzone CJ, Vu BC, Scott NL, Lecomte JTJ (2002) J Mol Biol 324:1015–1029
Delaglio F, Grzesiek S, Vuister GW, Zhu G, Pfeifer J, Bax A (1995) J Biomol NMR 6:277–293
Goddard TD, Kneller DG (2006) Sparky 3. University of California, San Francisco
Falzone CJ, Lecomte JTJ (2002) J Biomol NMR 23:71–72
Pond MP, Vuletich DA, Falzone CJ, Majumdar A, Lecomte JTJ (2009) Biomol NMR Assign 3:211–214
Geen H, Freeman R (1991) J Magn Reson 93:93–141
Farrow NA, Zhang O, Forman-Kay JD, Kay LE (1994) J Biomol NMR 4:727–734
Emsley L, Bodenhausen G (1990) Chem Phys Lett 165:469–476
Kay LE, Torchia DA, Bax A (1989) Biochemistry 28:8972–8979
Piotto M, Saudek V, Sklenár V (1992) J Biomol NMR 2:661–665
Shen Y, Delaglio F, Cornilescu G, Bax A (2009) J Biomol NMR 44:213–223
Emerson SD, La Mar GN (1990) Biochemistry 29:1556–1566
Brünger AT (1992) X-PLOR, version 3.1. A system for X-ray crystallography and NMR. Yale University Press, New Haven
Banci L, Bertini I, Cavallaro G, Giachetti A, Luchinat C, Parigi G (2004) J Biomol NMR 28:249–261
Schwieters CD, Kuszewski JJ, Clore GM (2006) Prog NMR Spectrosc 619(48):47–62
Schmitz C, Stanton-Cook MJ, Su XC, Otting G, Huber T (2008) J Biomol NMR 621(41):179–189
Jeener J, Meier BH, Bachmann P, Ernst RR (1979) J Chem Phys 71:4546–4553
Vu BC, Nothnagel HJ, Vuletich DA, Falzone CJ, Lecomte JTJ (2004) Biochemistry 43:12622–12633
Simonneaux G, Bondon A (2005) Chem Rev 105:2627–2646
Marcus RA, Sutin N (1985) Biochim Biophys Acta 811:265–322
Hoy JA, Smagghe BJ, Halder P, Hargrove MS (2007) Protein Sci 16:250–260
Davidson VL (2000) Acc Chem Res 33:87–93
Bonamore A, Boffi A (2008) IUBMB Life 60:19–28
Gardner PR (2005) J Inorg Biochem 99:247–266
Gardner PR, Gardner AM, Brashear WT, Suzuki T, Hvitved AN, Setchell KD, Olson JS (2006) J Inorg Biochem 100:542–550
Ouellet H, Ouellet Y, Richard C, Labarre M, Wittenberg B, Wittenberg J, Guertin M (2002) Proc Natl Acad Sci USA 99:5902–5907
Fago A, Mathews AJ, Moens L, Dewilde S, Brittain T (2006) FEBS Lett 580:4884–4888
Kiger L, Tilleman L, Geuens E, Hoogewijs D, Lechauve C, Moens L, Dewilde S, Marden MC (2011) PLoS ONE 6:e20478
Kakar S, Hoffman FG, Storz JF, Fabian M, Hargrove MS (2010) Biophys Chem 152:1–14
Nothnagel HJ, Love N, Lecomte JT (2009) J Inorg Biochem 103:107–116
Kraulis P (1991) J Appl Crystallogr 24:946–950
Acknowledgments
This study was supported by National Science Foundation grant MCB-0349409. NMR facilities and resources at Johns Hopkins University were provided by the Biomolecular NMR Center. The authors thank Selena Rice for assistance with the optical measurements, Richard Himes, Ryan Peterson, and Kenneth Karlin for the use of their stopped-flow equipment, and Christopher Falzone for useful discussions and careful reading of the manuscript. Henry Nothnagel’s insight into heme chemistry was essential in the initial phases of the work. Figure 1a was prepared with Molscript [49].
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Preimesberger, M.R., Pond, M.P., Majumdar, A. et al. Electron self-exchange and self-amplified posttranslational modification in the hemoglobins from Synechocystis sp. PCC 6803 and Synechococcus sp. PCC 7002. J Biol Inorg Chem 17, 599–609 (2012). https://doi.org/10.1007/s00775-012-0880-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-012-0880-5