Abstract
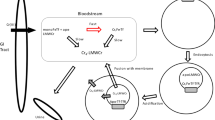
Iron release from human serum transferrin (hTF) has been studied extensively; however, the molecular details of the mechanism(s) remain incomplete. This is in part due to the complexity of this process, which is influenced by lobe–lobe interactions, the transferrin receptor (TFR), the salt effect, the presence of a chelator, and acidification within the endosome, resulting in iron release. The present work brings together many of the concepts and assertions derived from previous studies in a methodical, uniform, and visual manner. Examination of earlier work reveals some uncertainty due to sample and technical limitations. We have used a combination of steady-state fluorescence and urea gels to evaluate the effect of conformation, pH, time, and the soluble portion of the TFR (sTFR) on iron release from each lobe of hTF. The use of authentic recombinant monoferric and locked species removes any possibility of cross-contamination by acquisition of iron. Elimination of detergent by use of the sTFR provides a further technical advantage. We find that iron release from the N-lobe is very sensitive to the conformation of the C-lobe, but is insensitive to the presence of the sTFR or to changes in pH (between 5.6 and 6.4). Specifically, when the cleft of the C-lobe is locked, the urea gels indicate that only about half of the iron is completely removed from the cleft of the N-lobe. Iron release from the C-lobe is most affected by the presence of the sTFR and changes in pH, but is unaffected by the conformation of the N-lobe. A model for iron release from diferric hTF is provided to delineate our findings.








Similar content being viewed by others
Abbreviations
- BHK:
-
Baby hamster kidney cells
- DMEM-F12:
-
Dulbecco’s modified Eagle’s medium containing Ham F-12 nutrient mixture
- FeC hTF:
-
Nonglycosylated recombinant monoferric C-lobe human serum transferrin (mutations Y95F and Y188F preclude binding in the N-lobe) that contains an N-terminal His6 tag
- Fe2 hTF:
-
Nonglycosylated recombinant diferric human serum transferrin that contains an N-terminal His6 tag
- FeN hTF:
-
Nonglycosylated recombinant monoferric N-lobe human serum transferrin (mutations Y426F and Y517F preclude binding in the C-lobe) that contains an N-terminal His6 tag
- hTF:
-
Human serum transferrin
- LockC hTF:
-
Nonglycosylated recombinant diferric hTF that contains an N-terminal His6 tag and where mutation R632A locks iron in the C-lobe
- LockN hTF:
-
Nonglycosylated recombinant diferric hTF that contains an N-terminal His6 tag and where mutation K206E locks iron in the N-lobe
- MES:
-
2-Morpholinoethanesulfonic acid
- sTFR:
-
Soluble portion of the transferrin receptor (residues 121–760) expressed as a recombinant entity that contains an N-terminal His6 tag
- TBE:
-
Tris(hydroxymethyl)aminomethane–borate–EDTA
- TFR:
-
Human serum transferrin receptor
- Tris:
-
Tris(hydroxymethyl)aminomethane
References
Halbrooks PJ, He QY, Briggs SK, Everse SJ, Smith VC, MacGillivray RT, Mason AB (2003) Biochemistry 42:3701–3707
Williams J, Moreton K (1980) Biochem J 185:483–488
Huebers H, Josephson B, Huebers E, Csiba E, Finch C (1981) Proc Natl Acad Sci USA 78:2572–2576
Mason AB, Halbrooks PJ, James NG, Connolly SA, Larouche JR, Smith VC, MacGillivray RT, Chasteen ND (2005) Biochemistry 44:8013–8021
He QY, Mason AB (2002) In: Templeton DM (ed) Molecular and cellular iron transport. Dekker, Toronto
Dautry-Varsat A, Ciechanover A, Lodish HF (1983) Proc Natl Acad Sci USA 80:2258–2262
Bali PK, Harris WR (1989) J Am Chem Soc 111:4457–4461
Chasteen ND, Grady JK, Woodworth RC, Mason AB (1994) Adv Exp Med Biol 357:45–52
Hamilton DH, Turcot I, Stintzi A, Raymond KN (2004) J Biol Inorg Chem 9:936–944
Dewan JC, Mikami B, Hirose M, Sacchettini JC (1993) Biochemistry 32:11963–11968
He QY, Mason AB, Tam BM, MacGillivray RT, Woodworth RC (1999) Biochemistry 38:9704–9711
Halbrooks PJ, Giannetti AM, Klein JS, Bjorkman PJ, Larouche JR, Smith VC, MacGillivray RT, Everse SJ, Mason AB (2005) Biochemistry 44:15451–15460
Hall DR, Hadden JM, Leonard GA, Bailey S, Neu M, Winn M, Lindley PF (2002) Acta Crystallogr D Biol Crystallogr 58:70–80
Wally J, Halbrooks PJ, Vonrhein C, Rould MA, Everse SJ, Mason AB, Buchanan SK (2006) J Biol Chem 281:24934–24944
Egan TJ, Zak O, Aisen P (1993) Biochemistry 32:8162–8167
Lehrer SS (1969) J Biol Chem 244:3613–3617
Ross JA, Jameson DM (2008) Photochem Photobiol Sci 7:1301–1312
James NG, Berger CL, Byrne SL, Smith VC, Macgillivray RT, Mason AB (2007) Biochemistry 46:10603–10611
James NG, Byrne SL, Steere AN, Smith VC, MacGillivray RTA, Mason AB (2009) Biochemistry (in press)
Makey DG, Seal US (1976) Biochim Biophys Acta 453:250–256
Aisen P, Leibman A, Zweier J (1978) J Biol Chem 253:1930–1937
Evans RW, Williams J (1978) Biochem J 173:543–552
Leibman A, Aisen P (1979) Blood 53:1058–1065
Chasteen ND, Williams J (1981) Biochem J 193:717–727
Williams J, Chasteen ND, Moreton K (1982) Biochem J 201:527–532
Bali PK, Aisen P (1991) Biochemistry 30:9947–9952
Mason AB, Miller MK, Funk WD, Banfield DK, Savage KJ, Oliver RW, Green BN, MacGillivray RT, Woodworth RC (1993) Biochemistry 32:5472–5479
Turkewitz AP, Amatruda JF, Borhani D, Harrison SC, Schwartz AL (1988) J Biol Chem 263:8318–8325
Bali PK, Zak O, Aisen P (1991) Biochemistry 30:324–328
Mason AB, Halbrooks PJ, Larouche JR, Briggs SK, Moffett ML, Ramsey JE, Connolly SA, Smith VC, MacGillivray RT (2004) Protein Expr Purif 36:318–326
Byrne SL, Leverence R, Klein JS, Giannetti AM, Smith VC, MacGillivray RT, Kaltashov IA, Mason AB (2006) Biochemistry 45:6663–6673
Mason AB, He QY, Adams TE, Gumerov DR, Kaltashov IA, Nguyen V, MacGillivray RT (2001) Protein Expr Purif 23:142–150
James NG, Mason AB (2008) Anal Biochem 378:202–207
Bali PK, Aisen P (1992) Biochemistry 31:3963–3967
El Hage Chahine JM, Pakdaman R (1995) Eur J Biochem 230:1102–1110
Hemadi M, Ha-Duong NT, El Hage Chahine JM (2006) J Mol Biol 358:1125–1136
Grossmann JG, Crawley JB, Strange RW, Patel KJ, Murphy LM, Neu M, Evans RW, Hasnain SS (1998) J Mol Biol 279:461–472
Acknowledgments
This work was supported by USPHS Grant R01 (DK 21739) to A.B.M.. Support for S.L.B. came from Hemostasis and Thrombosis Training Grant (5T32HL007594), issued to K. G. Mann at the University of Vermont by the National Heart, Lung, and Blood Institute.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
775_2009_491_MOESM1_ESM.pdf
Maximum fluorescence emission from Fe2 hTF, FeN hTF, FeC hTF, LockC hTF and LockN hTF at pH 7.4 (100 mM HEPES) and pH 6.4–5.6 (100 mM MES, 300 mM KCl, 4 mM EDTA). All samples were incubated at least 15 minutes to try to assure complete iron removal. Samples were excited at 280 nm and emission was monitored between 300–400 nm using a 320 nm cut-on filter. (PDF 281 kb)
Rights and permissions
About this article
Cite this article
Byrne, S.L., Mason, A.B. Human serum transferrin: a tale of two lobes. Urea gel and steady state fluorescence analysis of recombinant transferrins as a function of pH, time, and the soluble portion of the transferrin receptor. J Biol Inorg Chem 14, 771–781 (2009). https://doi.org/10.1007/s00775-009-0491-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-009-0491-y