Abstract
Introduction
This study aimed to assess the association between pharmacotherapy and secondary hip fracture incidence.
Materials and methods
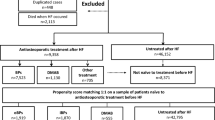
The correlation between secondary hip fracture incidence and the presence, type, and medication possession ratio (MPR) of pharmacotherapy was investigated using medical insurance data acquired from the National Database of Health Insurance Claims and Specific Health Checkups of Japan.
Results
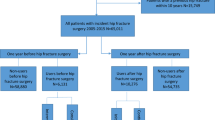
Data collected from female patients (n = 1,435,347) were analyzed. The 2-year secondary hip fracture incidence was 3.48% (n = 49,921). Secondary hip fracture was significantly more common in patients without medications (3.80%) than in those with medications (3.00%). Patients receiving selective estrogen receptor modulators (SERMs) had the lowest average age. The crude incidence of secondary hip fracture was the lowest in patients receiving SERMs (n = 2088 [2.52%]), followed by those taking bisphosphonates (n = 11,355 [2.88%]), denosumab (n = 1118 [2.90%]), no medications (n = 32,747 [3.80%]), and parathyroid hormone (PTH: n = 2163 [4.55%]), whereas the age-adjusted incidence was the lowest in patients administered denosumab (2.27%), followed by those taking bisphosphonates (2.47%), SERMs (2.55%), PTH (3.67%), and no medications (3.80%). The mean MPR was the highest in patients taking denosumab (64.9%), followed by those receiving bisphosphonates (58.7%), SERMs (58.2%), and PTH (40.6%) in the no hip fracture group.
Conclusion
Secondary hip fractures were less likely to occur with medication versus no medication. Differences in the crude incidence of secondary hip fracture based on medications usage might be attributed to background characteristics.


Similar content being viewed by others
Availability of data and materials
Permission from the Ministry of Health, Labour and Welfare is required for the use of all data and materials.
References
Klotzbuecher CM, Ross PD, Landsman PB, Abbott TA 3rd, Berger M (2000) Patients with prior fractures have an increased risk of future fractures: a summary of the literature and statistical synthesis. J Bone Miner Res 15:721–739
Frederiksen A, Abrahamsen B, Johansen PB, Sørensen HA (2018) Danish, national cross-sectional observational study on the prevalence of prior major osteoporotic fractures in adults presenting with hip fracture-limitations and scope for fracture liaison services in prevention of hip fracture. Osteoporos Int 29:109–114
Bynum JPW, Bell JE, Cantu RV, Wang Q, McDonough CM, Carmichael D, Tosteson TD, Tosteson ANA (2016) Second fractures among older adults in the year following hip, shoulder, or wrist fracture. Osteoporos Int 27:2207–2215
Dyer SM, Crotty M, Fairhall N, Magaziner J, Beaupre LA, Cameron ID, Sherrington C; Fragility Fracture Network (FFN) Rehabilitation Research Special Interest Group (2016) A critical review of the long-term disability outcomes following hip fracture. BMC Geriatr 16:158
Papadimitriou N, Tsilidis KK, Orfanos P, Benetou V, Ntzani EE et al (2017) Burden of hip fracture using disability-adjusted life-years: a pooled analysis of prospective cohorts in the CHANCES consortium. Lancet Public Health 2:e239–e246
Borgström F, Karlsson L, Ortsäter G, Norton N, Halbout P, Cooper C, Lorentzon M, McCloskey EV, Harvey NC, Javaid MK, Kanis JA; International Osteoporosis Foundation (2020) Fragility fractures in Europe: burden, management and opportunities. Arch Osteoporos 15:59
Schemitsch E, Adachi JD, Brown JP, Tarride JE, Burke N, Oliveira T, Slatkovska L (2021) Hip fracture predicts subsequent hip fracture: a retrospective observational study to support a call to early hip fracture prevention efforts in post-fracture patients. Osteoporos Int 11:1–10
Saito T, Sterbenz JM, Malay S, Zhong L, MacEachern MP, Chung KC (2017) Effectiveness of anti-osteoporotic drugs to prevent secondary fragility fractures: systematic review and meta-analysis. Osteoporos Int 28:3289–3300
Wang PW, Li YZ, Zhuang HF, Yu HM, Cai SQ, Xu H, Chen ZH, Lin JK, Yao XD (2019) Anti-osteoporosis medications associated with decreased mortality after hip fracture. Orthop Surg 11:777–783
Dobre R, Niculescu DA, Petca RC, Popescu RI, Petca A, Poiană C (2021) Adherence to anti-osteoporotic treatment and clinical implications after hip fracture: a systematic review. J Pers Med 11:341
Soen S, Fukunaga M, Sugimoto T, Sone T, Fujiwara S, Endo N, Gorai I, Shiraki M, Hagino H, Hosoi T, Ohta H, Yoneda T, Tomomitsu T, Japanese Society for Bone and Mineral Research and Japan Osteoporosis Society Joint Review Committee for the Revision of the Diagnostic Criteria for Primary Osteoporosis (2013) Diagnostic criteria for primary osteoporosis: year 2012 revision. J Bone Miner Metab 31:247–257
Japan Osteoporosis Society (2015) Guidelines on the prevention and treatment of osteoporosis 2015. The committee for development of the guidelines on the prevention and treatment of osteoporosis. (in Japanese). http://www.josteo.com/ja/guideline/doc/15_1.pdf. Accessed 22 Sep 2022
Nakatoh S, Fujimori K, Ishii S, Tamaki J, Okimoto N, Ogawa S, Iki M (2021) Insufficient increase in bone mineral density testing rates and pharmacotherapy after hip and vertebral fracture: analysis of the National Database of Health Insurance Claims and Specific Health Checkups of Japan. Arch Osteoporos 16:130
Chen YC, Lin WC (2017) Poor 1st-year adherence to anti-osteoporotic therapy increases the risk of mortality in patients with magnetic resonance imaging-proven acute osteoporotic vertebral fractures. Patient Prefer Adherence 11:839–843
Kanis JA, Cooper C, Hiligsmann M, Rabenda V, Reginster JY, Rizzoli R (2011) Partial adherence: a new perspective on health economic assessment in osteoporosis. Osteoporos Int 22:2565–2573
Nakatoh S, Fujimori K, Ishii S, Tamaki J, Okimoto N, Ogawa S, Iki M (2021) Insufficient persistence to pharmacotherapy in Japanese patients with osteoporosis: an analysis of the National Database of Health Insurance Claims and Specific Health Checkups in Japan. Arch Osteoporos 16:131
Barrionuevo P, Kapoor E, Asi N, Alahdab F, Mohammed K, Benkhadra K, Almasri J, Farah W, Sarigianni M, Muthusamy K, Al Nofal A, Haydour Q, Wang Z, Murad MH (2019) Efficacy of pharmacological therapies for the prevention of fractures in postmenopausal women: a network meta-analysis. J Clin Endocrinol Metab 104:1623–1630
Simpson EL, Martyn-St James M, Hamilton J, Wong R, Gittoes N, Selby P, Davis S (2020) Clinical effectiveness of denosumab, raloxifene, romosozumab, and teriparatide for the prevention of osteoporotic fragility fractures: a systematic review and network meta-analysis. Bone 130:115081
Nakatoh S, Fujimori K, Ishii S, Tamaki J, Okimoto N, Ogawa S, Iki M (2022) Association of pharmacotherapy with the second hip fracture incidence in women: a retrospective analysis of the National Database of Health Insurance Claims and Specific Health Checkups of Japan. Geriatr Gerontol Int 22:930–937
Ministry of Health, Labour, and Welfare (2020) The popularization and state of electronic receipt claims. https://www.mhlw.go.jp/file/06-Seisakujouhou-12400000-Hokenkyoku/0000099002.pdf. (in Japanese) Accessed 22 Sep 2022
The Japanese Orthopaedic Association (2020) Results of National Survey of hip fracture in 2020. (in Japanese). https://www.joa.or.jp/member/committee/osteoporosis/pdf/femur20.pdf. Accessed 22 Sep 2022
Liu S, Zhu Y, Chen W, Sun T, Cheng J, Zhang Y (2015) Risk factors for the second contralateral hip fracture in elderly patients: a systematic review and meta-analysis. Clin Rehabil 29:285–294
Sheikh HQ, Hossain FS, Khan S, Usman M, Kapoor H, Aqil A (2019) Short-term risk factors for a second hip fracture in a UK population. Eur J Orthop Surg Traumatol 29:1055–1060
Conley RB, Adib G, Adler RA, Åkesson KE, Alexander IM et al (2020) Secondary fracture prevention: consensus clinical recommendations from a multistakeholder coalition. J Bone Miner Res 35:36–52
Barron RL, Oster G, Grauer A, Crittenden DB, Weycker D (2020) Determinants of imminent fracture risk in postmenopausal women with osteoporosis. Osteoporos Int 31:2103–2111
Yeam CT, Chia S, Tan HCC, Kwan YH, Fong W, Seng JJB (2018) A systematic review of factors affecting medication adherence among patients with osteoporosis. Osteoporos Int 29:2623–2637
van Geel TA, van Helden S, Geusens PP, Winkens B, Dinant GJ (2009) Clinical subsequent fractures cluster in time after first fractures. Ann Rheum Dis 68:99–102
Johansson H, Siggeirsdóttir K, Harvey NC, Odén A, Gudnason V, McCloskey E, Sigurdsson G, Kanis JA (2017) Imminent risk of fracture after fracture. Osteoporos Int 28:775–780
Kanis JA, Johansson H, Odén A, Harvey NC, Gudnason V, Sanders KM, Sigurdsson G, Siggeirsdottir K, Fitzpatrick LA, Borgström F, McCloskey EV (2018) Characteristics of recurrent fractures. Osteoporos Int 29:1747–1757
Acknowledgements
We thank Enago (https://www.enago.jp) for their English-language editing services.
Funding
This study was partly supported by the 28th Pfizer Health Research Grant, 52nd (2019) Medical Research Grant from Taijyu Life Welfare Foundation and 2020 Research Grant from Japan Osteoporosis Foundation. The funding bodies had no role in the study design, data collection and analysis, decision to publish, or manuscript preparation.
Author information
Authors and Affiliations
Contributions
SN: study conception and design, data analysis and interpretation, article drafting, critical revisions of the article for important intellectual content, and final approval. KF: full access to all study data and responsible for the integrity of data and accuracy of data analysis. SI, JT, NO, JO, and MI: study conception and design, critical revisions of the article for important intellectual content, and final approval.
Corresponding author
Ethics declarations
Conflict of interest
S. Nakatoh has received payments for lectures, including speakers' bureau fees, from Asahi-Kasei Pharmaceutical Co., Ltd.; Amgen K.K.; and Astellas Pharma Inc. N. Okimoto has received consulting fees from Asahi-Kasei Pharmaceutical Co., Ltd. and Teijin Pharma Ltd. N. Okimoto has received payments for lectures, including speakers' bureau fees from Asahi-Kasei Pharmaceutical Co., Ltd.; Amgen Astellas Bio Pharma K.K.; Astellas Pharma Inc.; Chugai Pharmaceutical Co.; Daiichi-Sankyo Co., Ltd.; Eisai Co., Ltd.; Eli Lilly Japan; and Teijin Pharma Ltd. The other authors have no conflicts of interest to declare.
Ethical approval
The study was conducted according to the principles of the Declaration of Helsinki and was approved by the Ethics Committee of Kindai University Faculty of Medicine (approval number: 31–065).
Consent to participate and consent for publication
The study data were completely anonymized. All data used in our analysis were deidentified by the Ministry of Health, Labour and Welfare. This study was performed in accordance with the guidelines of the Ministry of Information Security. Thus, informed consent was not required.
Informed consent
The data in this study were completely anonymous. All data used in our analysis were deidentified by the Ministry of Health, Labour and Welfare, and this study followed the guidelines of the Ministry of Information Security. Thus, informed consent was not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Nakatoh, S., Fujimori, K., Ishii, S. et al. Association between pharmacotherapy and secondary hip fracture in a real-world setting: a nationwide database study. J Bone Miner Metab 41, 248–257 (2023). https://doi.org/10.1007/s00774-023-01411-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00774-023-01411-4