Abstract
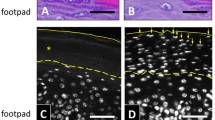
Transglutaminases (TGs) are a family of enzymes that catalyse the formation of isopeptide bonds between the γ-carboxamide groups of glutamine residues and the ε-amino groups of lysine residues leading to cross-linking reactions among proteins. Four members, TG1, TG2, TG3, and TG5, of the nine mammalian enzymes are expressed in the skin. TG1, TG3 and TG5 crosslinking properties are fundamental for cornified envelope assembly. In contrast, the role of TG2 in keratinization has never been studied at biochemical level in vivo. In this study, taking advantage of the TG2 knock-out (KO) and TG1 heterozygous mice, we generated and characterized the epidermis of TG1–TG2 double knock-out (DKO) mice. We performed morphological analysis of the epidermis and evaluation of the expression of differentiation markers. In addition, we performed analysis of the amino acid composition from isolated corneocytes. We found a significant change in amino acid composition in TG1KO cornified cell envelopes (CEs) while TG2KO amino acid composition was similar to wild-type CEs. Our results confirm a key role of TG1 in skin differentiation and CE assembly and demonstrate that TG2 is not essential for CE assembly and skin formation.



Similar content being viewed by others
Abbreviations
- TGs:
-
Transglutaminases
- ECM:
-
Extracellular matrix
- KO:
-
Knock-out mouse
- DKO:
-
Double knock-out mouse
- CE:
-
Cornified cell envelope
References
Aeschlimann D, Paulsson M (1991) Cross-linking of laminin-nidogen complexes by tissue transglutaminase. A novel mechanism for basement membrane stabilization. J Biol Chem 266(23):15308–15317
Candi E, Melino G, Mei G, Tarcsa E, Chung SI, Marekov LN, Steinert PM (1995) Biochemical, structural, and transglutaminase substrate properties of human loricrin, the major epidermal cornified cell envelope protein. J Biol Chem 270(44):26382–26390
Candi E, Melino G, Lahm A, Ceci R, Rossi A, Kim IG, Ciani B, Steinert PM (1998) Transglutaminase 1 mutations in lamellar ichthyosis. Loss of activity due to failure of activation by proteolytic processing. J Biol Chem 273(22):13693–13702
Candi E, Tarcsa E, Idler WW, Kartasova T, Marekov LN, Steinert PM (1999) Transglutaminase cross-linking properties of the small proline-rich 1 family of cornified cell envelope proteins. Integration with loricrin. J Biol Chem 274(11):7226–7237
Candi E, Oddi S, Terrinoni A, Paradisi A, Ranalli M, Finazzi-Agro A, Melino G (2001) Transglutaminase 5 cross-links loricrin, involucrin, and small proline-rich proteins in vitro. J Biol Chem 276(37):35014–35023. doi:10.1074/jbc.M010157200
Candi E, Oddi S, Paradisi A, Terrinoni A, Ranalli M, Teofoli P, Citro G, Scarpato S, Puddu P, Melino G (2002) Expression of transglutaminase 5 in normal and pathologic human epidermis. J Invest Dermatol 119(3):670–677. doi:10.1046/j.1523-1747.2002.01853.x
Candi E, Schmidt R, Melino G (2005) The cornified envelope: a model of cell death in the skin. Nat Rev Mol Cell Biol 6(4):328–340. doi:10.1038/nrm1619
Craiglow BG (2013) Ichthyosis in the newborn. Semin Perinatol 37(1):26–31. doi:10.1053/j.semperi.2012.11.001
De Laurenzi V, Melino G (2001) Gene disruption of tissue transglutaminase. Mol Cell Biol 21(1):148–155. doi:10.1128/MCB.21.1.148-155.2001
Dolynchuk KN, Bendor-Samuel R, Bowness JM (1994) Effect of putrescine on tissue transglutaminase activity in wounds: decreased breaking strength and increased matrix fucoprotein solubility. Plast Reconstr Surg 93(3):567–573
Etoh Y, Simon M, Green H (1986) Involucrin acts as a transglutaminase substrate at multiple sites. Biochem Biophys Res Commun 136(1):51–56
Fischer J (2009) Autosomal recessive congenital ichthyosis. J Invest Dermatol 129(6):1319–1321. doi:10.1038/jid.2009.57
Gentile V, Thomazy V, Piacentini M, Fesus L, Davies PJ (1992) Expression of tissue transglutaminase in Balb-C 3T3 fibroblasts: effects on cellular morphology and adhesion. J Cell Biol 119(2):463–474
Greenberg CS, Birckbichler PJ, Rice RH (1991) Transglutaminases: multifunctional cross-linking enzymes that stabilize tissues. FASEB J 5(15):3071–3077
Griffin M, Casadio R, Bergamini CM (2002) Transglutaminases: nature’s biological glues. Biochem J 368(Pt 2):377–396. doi:10.1042/BJ20021234
Haroon ZA, Hettasch JM, Lai TS, Dewhirst MW, Greenberg CS (1999) Tissue transglutaminase is expressed, active, and directly involved in rat dermal wound healing and angiogenesis. FASEB J 13(13):1787–1795
Huber M, Rettler I, Bernasconi K, Frenk E, Lavrijsen SP, Ponec M, Bon A, Lautenschlager S, Schorderet DF, Hohl D (1995) Mutations of keratinocyte transglutaminase in lamellar ichthyosis. Science 267(5197):525–528
Jarnik M, Kartasova T, Steinert PM, Lichti U, Steven AC (1996) Differential expression and cell envelope incorporation of small proline-rich protein 1 in different cornified epithelia. J Cell Sci 109(Pt 6):1381–1391
Kalinin A, Marekov LN, Steinert PM (2001) Assembly of the epidermal cornified cell envelope. J Cell Sci 114(Pt 17):3069–3070
Kalinin AE, Kajava AV, Steinert PM (2002) Epithelial barrier function: assembly and structural features of the cornified cell envelope. BioEssays 24(9):789–800. doi:10.1002/bies.10144
Kuramoto N, Takizawa T, Takizawa T, Matsuki M, Morioka H, Robinson JM, Yamanishi K (2002) Development of ichthyosiform skin compensates for defective permeability barrier function in mice lacking transglutaminase 1. J Clin Invest 109(2):243–250. doi:10.1172/JCI13563
Lorand L, Graham RM (2003) Transglutaminases: crosslinking enzymes with pleiotropic functions. Nat Rev Mol Cell Biol 4(2):140–156. doi:10.1038/nrm1014
Matsuki M, Yamashita F, Ishida-Yamamoto A, Yamada K, Kinoshita C, Fushiki S, Ueda E, Morishima Y, Tabata K, Yasuno H, Hashida M, Iizuka H, Ikawa M, Okabe M, Kondoh G, Kinoshita T, Takeda J, Yamanishi K (1998) Defective stratum corneum and early neonatal death in mice lacking the gene for transglutaminase 1 (keratinocyte transglutaminase). Proc Natl Acad Sci USA 95(3):1044–1049
Michel S, Schmidt R, Shroot B, Reichert U (1988) Morphological and biochemical characterization of the cornified envelopes from human epidermal keratinocytes of different origin. J Invest Dermatol 91(1):11–15
Nanda N, Iismaa SE, Owens WA, Husain A, Mackay F, Graham RM (2001) Targeted inactivation of Gh/tissue transglutaminase II. J Biol Chem 276(23):20673–20678. doi:10.1074/jbc.M010846200
Nemes Z, Steinert PM (1999) Bricks and mortar of the epidermal barrier. Exp Mol Med 31(1):5–19. doi:10.1038/emm.1999.2
Nemes Z, Marekov LN, Fesus L, Steinert PM (1999a) A novel function for transglutaminase 1: attachment of long-chain omega-hydroxyceramides to involucrin by ester bond formation. Proc Natl Acad Sci USA 96(15):8402–8407
Nemes Z, Marekov LN, Steinert PM (1999b) Involucrin cross-linking by transglutaminase 1. Binding to membranes directs residue specificity. J Biol Chem 274(16):11013–11021
Raghunath M, Hopfner B, Aeschlimann D, Luthi U, Meuli M, Altermatt S, Gobet R, Bruckner-Tuderman L, Steinmann B (1996) Cross-linking of the dermo-epidermal junction of skin regenerating from keratinocyte autografts. Anchoring fibrils are a target for tissue transglutaminase. J Clin Invest 98(5):1174–1184. doi:10.1172/JCI118901
Reichert U, Michel S, Schmidt C (2012) The cornified envelope: a key structure of terminally differentiating keratinocytes. In: Darmon M, Blumenberg M (eds) Molecular biology of the skin: the keratinocyte, vol 1. Academic Press, San Diego, pp 107–146
Rice RH, Green H (1977) The cornified envelope of terminally differentiated human epidermal keratinocytes consists of cross-linked protein. Cell 11(2):417–422
Rorke EA, Eckert RL (1991) Stable expression of transfected human involucrin gene in various cell types: evidence for in situ cross-linking by type I and type II transglutaminase. J Invest Dermatol 97(3):543–548
Russell LJ, DiGiovanna JJ, Rogers GR, Steinert PM, Hashem N, Compton JG, Bale SJ (1995) Mutations in the gene for transglutaminase 1 in autosomal recessive lamellar ichthyosis. Nat Genet 9(3):279–283. doi:10.1038/ng0395-279
Sandler B, Hashimoto K (1998) Collodion baby and lamellar ichthyosis. J Cutan Pathol 25(2):116–121
Simon M, Green H (1988) The glutamine residues reactive in transglutaminase-catalyzed cross-linking of involucrin. J Biol Chem 263(34):18093–18098
Steinert PM (2000) The complexity and redundancy of epithelial barrier function. J Cell Biol 151(2):F5–F8
Steinert PM, Marekov LN (1995) The proteins elafin, filaggrin, keratin intermediate filaments, loricrin, and small proline-rich proteins 1 and 2 are isodipeptide cross-linked components of the human epidermal cornified cell envelope. J Biol Chem 270(30):17702–17711
Steinert PM, Marekov LN (1997) Direct evidence that involucrin is a major early isopeptide cross-linked component of the keratinocyte cornified cell envelope. J Biol Chem 272(3):2021–2030
Steinert PM, Chung SI, Kim SY (1996) Inactive zymogen and highly active proteolytically processed membrane-bound forms of the transglutaminase 1 enzyme in human epidermal keratinocytes. Biochem Biophys Res Commun 221(1):101–106. doi:10.1006/bbrc.1996.0552
Stephens P, Grenard P, Aeschlimann P, Langley M, Blain E, Errington R, Kipling D, Thomas D, Aeschlimann D (2004) Crosslinking and G-protein functions of transglutaminase 2 contribute differentially to fibroblast wound healing responses. J Cell Sci 117(Pt 15):3389–3403. doi:10.1242/jcs.01188
Steven AC, Steinert PM (1994) Protein composition of cornified cell envelopes of epidermal keratinocytes. J Cell Sci 107(Pt 2):693–700
Steven AC, Bisher ME, Roop DR, Steinert PM (1990) Biosynthetic pathways of filaggrin and loricrin—two major proteins expressed by terminally differentiated epidermal keratinocytes. J Struct Biol 104(1–3):150–162
Takahashi M, Tezuka T, Kakegawa H, Katunuma N (1994) Linkage between phosphorylated cystatin alpha and filaggrin by epidermal transglutaminase as a model of cornified envelope and inhibition of cathepsin L activity by cornified envelope and the conjugated cystatin alpha. FEBS Lett 340(3):173–176
Tarcsa E, Candi E, Kartasova T, Idler WW, Marekov LN, Steinert PM (1998) Structural and transglutaminase substrate properties of the small proline-rich 2 family of cornified cell envelope proteins. J Biol Chem 273(36):23297–23303
Tatsukawa H, Abe N, Ohashi S, Hitomi K (2015) Distribution of transglutaminase family members in mouse whole body sections. Biochem Biophys Res Commun 467(4):1046–1051. doi:10.1016/j.bbrc.2015.10.001
Telci D, Griffin M (2006) Tissue transglutaminase (TG2)—a wound response enzyme. Front Biosci 11:867–882
Terrinoni A, Serra V, Codispoti A, Talamonti E, Bui L, Palombo R, Sette M, Campione E, Didona B, Annicchiarico-Petruzzelli M, Zambruno G, Melino G, Candi E (2012) Novel transglutaminase 1 mutations in patients affected by lamellar ichthyosis. Cell Death Dis 3:e416. doi:10.1038/cddis.2012.152
Verderio E, Nicholas B, Gross S, Griffin M (1998) Regulated expression of tissue transglutaminase in Swiss 3T3 fibroblasts: effects on the processing of fibronectin, cell attachment, and cell death. Exp Cell Res 239(1):119–138. doi:10.1006/excr.1997.3874
Verderio EA, Johnson T, Griffin M (2004) Tissue transglutaminase in normal and abnormal wound healing: review article. Amino Acids 26(4):387–404. doi:10.1007/s00726-004-0094-4
Acknowledgements
This work was mainly supported by “Ricerca Corrente RC-1” Italian Ministry of Health to AT, and partially supported by the Medical Research Council (MRC), “Fondazione Roma” NCD Grant to GM and EC.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Handling Editors: S. Beninati, M. Piacentini, C. M. Bergamini.
C. Pitolli and V. Pietroni contributed equally.
Rights and permissions
About this article
Cite this article
Pitolli, C., Pietroni, V., Marekov, L. et al. Characterization of TG2 and TG1–TG2 double knock-out mouse epidermis. Amino Acids 49, 635–642 (2017). https://doi.org/10.1007/s00726-016-2356-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-016-2356-3