Abstract
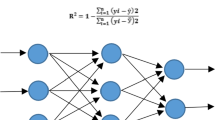
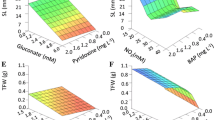
Doubled haploids, subsequent to haploid induction, have wide range of applications in basic and applied plant studies. Various parameters can affect the efficiency of haploid induction through an anther culture of tomato. The hybrid system of image processing-artificial neural network (ANN) was used to better understand callus induction and regeneration in an anther culture of tomato. The effect of parameters such as plant genotype, the concentrations of 2,4-dichlorophenoxyacetic acid (2,4-D) and kinetin (Kin) plant growth regulators, the concentration of gum arabic (GA) additive, the cold pretreatment duration, and flower length on callus induction percentage and number of regenerated calli in an anther culture of tomato were studied using multiple linear regression (MLR) and ANN models. The precise flower bud length was measured using an image processing technique. The 4′,6-diamidino-2-phenylindole (DAPI) analysis showed that the flowers with 5–6.9 mm length had the highest percentage of the mid- to late-uninucleate microspore stage. The best ANN model for both callus induction percentage and number of regenerated calli was a model with one hidden layer, 12–15 neurons in the first hidden layer, Levenberg–Marquardt learning algorithm, and Tan-Sigmoid transfer function in hidden layer, based on the root mean square error (RMSE), mean absolute error (MAE), and coefficient of determination (R2) statistics. The scatter plot of measured values versus the predicted values showed the superiority of the ANN to MLR model to predict the callus induction percentage in an anther culture of tomato. The sensitivity analysis of MLR and ANN models revealed the plant genotype and 2,4-D concentration as the most important factors affecting both callus induction percentage and number of regenerated calli. Since tomato is a recalcitrant plant to androgenesis-based pathway of haploid induction, therefore the results of the present study can be helpful to develop an efficient haploid induction protocol in tomato through an anther culture pathway.









Similar content being viewed by others
Abbreviations
- ANN:
-
Artificial neural network
- BBBM:
-
Biotechnology-based breeding method
- CIP:
-
Callus induction percentage
- DH:
-
Doubled haploid
- GA:
-
Gum arabic
- IP:
-
Image processing
- Kin:
-
Kinetin
- MAE:
-
Mean absolute error
- MLP:
-
Multi-layer perceptron
- MLR:
-
Multiple linear regression
- NRC:
-
Number of regenerated calli
- PGRs:
-
Plant growth regulators
- TCB:
-
Tissue culture based
- QTL:
-
Quantitative trait loci
- RMSE:
-
Root mean square error
- R 2 :
-
Coefficient of determination
- 2,4-D:
-
2,4-Dichlorophenoxyacetic acid
References
Abdipour M, Ramazani SHR, Younessi-Hmazekhanlu M, Niazian M (2018) Modeling oil content of sesame (Sesamum indicum L.) using artificial neural network and multiple linear regression approaches. J Am Oil Chem Soc 95(3):283–297. https://doi.org/10.1002/aocs.12027
Abdipour M, Younessi-Hmazekhanlu M, Ramazani SHR (2019) Artificial neural networks and multiple linear regression as potential methods for modeling seed yield of safflower (Carthamus tinctorius L.). Ind Crop Prod 127:185–194. https://doi.org/10.1016/j.indcrop.2018.10.050
Abdollahi MR, Rashidi S (2017) Production and conversion of haploid embryos in chickpea (Cicer arietinum L.) anther cultures using high 2,4-D and silver nitrate containing media. Plant Cell Tissue Organ Cult 133(1):39–49. https://doi.org/10.1007/s11240-017-1359-4
Adhikari PB, Kang WH (2017) Association of floral bud and anther size with microspore developmental stage in Campari tomato. Korean J Hortic Sci 35(5):608–617
Ahmadi B, Shariatpanahi ME, Asghari-Zakaria R, Zare N, Azadi P (2015) Efficient microspore embryogenesis induction in tomato (Lycopersicon esculentum Mill.) using shed microspore culture. J Pure Appl Microbiol 9(2):21–29
Arab MM, Yadollahi A, Shojaeiyan A, Ahmadi H (2016) Artificial neural network genetic algorithm as powerful tool to predict and optimize in vitro proliferation mineral medium for G× N15 rootstock. Front Plant Sci 7(1526). https://doi.org/10.3389/fpls.2016.01526
Bal U, Abak K (2007) Haploidy in tomato (Lycopersicon esculentum Mill.): a critical review. Euphytica 158(1–2):1–9. https://doi.org/10.1007/s10681-007-9427-1
Britt AB, Kuppu S (2016) Cenh3: an emerging player in haploid induction technology. Front Plant Sci 7:357. https://doi.org/10.3389/fpls.2016.00357
Cheema DS, Dhaliwal MS (2005) Hybrid tomato breeding. J New Seeds 6(2–3):1–14. https://doi.org/10.1300/J153v06n02_01
Chegini GR, Khazaei J, Ghobadian B, Goudarzi AM (2008) Prediction of process and product parameters in an orange juice spray dryer using artificial neural networks. J Food Eng (4, 4):534–543. https://doi.org/10.1016/j.jfoodeng.2007.06.007
Dong YQ, Zhao WX, Li XH, Liu XC, Gao NN, Huang JH, Wang WY, Xu XL, Tang ZH (2016) Androgenesis, gynogenesis, and parthenogenesis haploids in cucurbit species. Plant Cell Rep 35(10):1991–2019. https://doi.org/10.1007/s00299-016-2018-7
Dwivedi SL, Britt AB, Tripathi L, Sharma S, Upadhyaya HD, Ortiz R (2015) Haploids: constraints and opportunities in plant breeding. Biotechnol Adv 33(6):812–829. https://doi.org/10.1016/j.biotechadv.2015.07.001
Emamgholizadeh S, Parsaeian M, Baradaran M (2015) Seed yield prediction of sesame using artificial neural network. Eur J Agron 68:89–96. https://doi.org/10.1016/j.eja.2015.04.010
Erzin Y, Rao BH, Singh DN (2008) Artificial neural network models for predicting soil thermal resistivity. Int J Therm Sci 47(10):1347–1358. https://doi.org/10.1016/j.ijthermalsci.2007.11.001
Evans MM (2007) The indeterminate gametophyte1 gene of maize encodes a LOB domain protein required for embryo sac and leaf development. Plant Cell 19(1):46–62. https://doi.org/10.1105/tpc.106.047506
Farjam A, Omid M, Akram A, Fazel Niari Z (2014) A neural network based modeling and sensitivity analysis of energy inputs for predicting seed and grain corn yields. J Agric Sci Technol 16(4):767–778
Fayos O, Vallés MP, Garcés-Claver A, Mallor C, Castillo AM (2015) Doubled haploid production from Spanish onion (Allium cepa L.) germplasm: embryogenesis induction, plant regeneration and chromosome doubling. Front Plant Sci 6(384). https://doi.org/10.3389/fpls.2015.00384
Germanà MA (2006) Doubled haploid production in fruit crops. Plant Cell Tissue Organ Cult 86(2):131–146. https://doi.org/10.1007/s11240-006-9088-0
Germanà MA (2011) Anther culture for haploid and doubled haploid production. Plant Cell Tissue Organ Cult 104(3):283–300. https://doi.org/10.1007/s11240-010-9852-z
Gilles LM, Khaled A, Laffaire JB, Chaignon S, Gendrot G, Laplaige J, Bergès H, Beydon G, Bayle V, Barret P, Comadran J (2017) Loss of pollen-specific phospholipase NOT LIKE DAD triggers gynogenesis in maize. EMBO J 36:707–717. https://doi.org/10.15252/embj.201796603
Gonzalo MJ, Claveria E, Monforte AJ, Dolcet-Sanjuan R (2011) Parthenogenic haploids in melon: generation and molecular characterization of a doubled haploid line population. J Am Soc Hortic Sci 136(2):145–154. https://doi.org/10.21273/JASHS.136.2.145
Gresshoff PM, Doy CH (1972) Development and differentiation of haploid Lycopersicon esculentum (tomato). Planta 107(2):161–170. https://doi.org/10.1007/BF00387721
Gupta SD, Pattanayak AK (2017) Intelligent image analysis (IIA) using artificial neural network (ANN) for non-invasive estimation of chlorophyll content in micropropagated plants of potato. In Vitro Cell Dev Biol Plant 53(6):520–526. https://doi.org/10.1007/s11627-017-9825-6
Gurushidze M, Hensel G, Hiekel S, Schedel S, Valkov V, Kumlehn J (2014) True-breeding targeted gene knock-out in barley using designer TALE-nuclease in haploid cells. PLoS One 9(3):e92046. https://doi.org/10.1371/journal.pone.0092046
Heidari-Zefreh AA, Shariatpanahi ME, Mousavi A, Kalatejari S (2018) Enhancement of microspore embryogenesis induction and plantlet regeneration of sweet pepper (Capsicum annuum L.) using putrescine and ascorbic acid. Protoplasma 256(1):13–24. https://doi.org/10.1007/s00709-018-1268-3
Kasperbauer MJ, Wilson HM (1979) Haploid plant production and use. In: Durbin RD (ed) Nicotiana procedures for experimental use. U.S. Dept. Agr. Technol. Bul. 1586: pp 33–39
Kelliher T, Starr D, Richbourg L, Chintamanani S, Delzer B, Nuccio ML, Green J, Chen Z, McCuiston J, Wang W, Liebler T (2017) MATRILINEAL, a sperm-specific phospholipase, triggers maize haploid induction. Nature 542(7639):105–109. https://doi.org/10.1038/nature20827
Khan H, Bhardwaj SC, Gangwar OP, Prasad P, Rathore R (2017) Efficiency of double haploid production in wheat through wide hybridization and embryo rescue. Indian J Genet Pl Br 77(3):428–430. https://doi.org/10.5958/0975-6906.2017.00059.1
Labbani Z, Richard N, De Buyser J, Picard E (2005) Chlorophyllian durum wheat plants obtained by isolated microspores culture: importance of the pre-treatments. C R Biol 328(8):713–723. https://doi.org/10.1016/j.crvi.2005.05.009
Liu C, Li X, Meng D, Zhong Y, Chen C, Dong X, Xu X, Chen B, Li W, Li L, Tian X (2017) A 4-bp insertion at ZmPLA1 encoding a putative phospholipase A generates haploid induction in maize. Mol Plant 10(3):520–522. https://doi.org/10.1016/j.molp.2017.01.011
Makowska K, Kałużniak M, Oleszczuk S, Zimny J, Czaplicki A, Konieczny R (2017) Arabinogalactan proteins improve plant regeneration in barley (Hordeum vulgare L.) anther culture. Plant Cell Tissue Organ Cult 131(2):247–257. https://doi.org/10.1007/s11240-017-1280-x
Mansouri A, Fadavi A, Mortazavian SMM (2015) Effects of length and position of hypocotyl explants on Cuminum cyminum L. callogensis by image processing analysis. Plant Cell Tissue Organ Cult 121(3):657–666. https://doi.org/10.1007/s11240-015-0736-0
Mansouri A, Fadavi A, Mortazavian SMM (2016) An artificial intelligence approach for modeling volume and fresh weight of callus—a case study of cumin (Cuminum cyminum L.). J Theor Biol 397:199–205. https://doi.org/10.1016/j.jtbi.2016.03.009
MATLAB and Statistics Toolbox Release (2018) The MathWorks, Inc., Natick, Massachusetts, United States
Mehrotra S, Prakash O, Mishra BN, Dwevedi B (2008) Efficiency of neural networks for prediction of in vitro culture conditions and inoculum properties for optimum productivity. Plant Cell Tissue Organ Cult 95(1):29–35. https://doi.org/10.1007/s11240-008-9410-0
Mishra VK, Bajpai R, Chaturvedi R (2017) An efficient and reproducible method for development of androgenic haploid plants from in vitro anther cultures of Camellia assamica ssp. assamica (Masters). In Vitro Cell Dev Biol Plant 53(3):239–248. https://doi.org/10.1007/s11627-017-9811-z
Motallebi-Azar A (2010) Androgenic response of tomato (Lycopersicon esculentum Mill.) lines and their hybrids to anther culture. Russ Agric Sci 36(4):250–258. https://doi.org/10.3103/S1068367410040087
Nezami Alanagh E, Garoosi G, Haddad R, Maleki S, Landín M, Gallego PP (2014) Design of tissue culture media for efficient Prunus rootstock micropropagation using artificial intelligence models. Plant Cell Tissue Organ Cult 117:349–359. https://doi.org/10.1007/s11240-014-0444-1
Niazian M, Sadat-Noori SA, Abdipour M, Tohidfar M, Mortazavian SMM (2018a) Image processing and artificial neural network-based models to measure and predict physical properties of embryogenic callus and number of somatic embryos in ajowan (Trachyspermum ammi (L.) Sprague). In Vitro Cell Dev Biol Plant 54(1):54–68. https://doi.org/10.1007/s11627-017-9877-7
Niazian M, Sadat-Noori SA, Abdipour M (2018b) Modeling the seed yield of Ajowan (Trachyspermum ammi L.) using artificial neural network and multiple linear regression models. Ind Crop Prod 117:224–234. https://doi.org/10.1016/j.indcrop.2018.03.013
Niazian M, Sadat-Noori SA, Abdipour M (2018c) Artificial neural network and multiple regression analysis models to predict essential oil content of ajowan (Carum copticum L.). J Appl Res Med Aromat Plants 9:124–131. https://doi.org/10.1016/j.jarmap.2018.04.001
Nowaczyk L, Nowaczyk P, Olszewska D (2016) Treating donor plants with 2,4-dichlorophenoxyacetic acid can increase the effectiveness of induced androgenesis in Capsicum spp. Sci Hortic 205:1–6. https://doi.org/10.1016/j.scienta.2016.03.044
Pechan PM, Keller WA (1988) Identification of potentially embryogenic microspores in Brassica napus. Physiol Plant 74(2):377–384. https://doi.org/10.1111/j.1399-3054.1988.tb00646.x
Prasad A, Prakash O, Mehrotra S, Khan F, Mathur AK, Mathur A (2017) Artificial neural network-based model for the prediction of optimal growth and culture conditions for maximum biomass accumulation in multiple shoot cultures of Centella asiatica. Protoplasma 254(1):335–341. https://doi.org/10.1007/s00709-016-0953-3
Ravi M, Chan SW (2010) Haploid plants produced by centromere-mediated genome elimination. Nature 464(7288):615–618. https://doi.org/10.1038/nature08842
Rosenblatt FX (1961) Principles of neurodynamics: perceptrons and the theory of brain mechanisms. Cornell Aeronautical Lab Inc Buffalo NY. Spartan Books, Washington DC
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ: 25 years of image analysis. Nat Methods 9(7):671–675. https://doi.org/10.1038/nmeth.2089
Seguí-Simarro JM, Nuez F (2005) Meiotic metaphase I to telophase II as the most responsive stage during microspore development for callus induction in tomato (Solanum lycopersicum) anther cultures. Acta Physiol Plant 27(4):675–685. https://doi.org/10.1007/s11738-005-0071-x
Seguí-Simarro JM, Nuez F (2008) How microspores transform into haploid embryos: changes associated with embryogenesis induction and microspore-derived embryogenesis. Physiol Plant 134(1):1–12. https://doi.org/10.1111/j.1399-3054.2008.01113.x
Shariatpanahi ME, Ahmadi B (2016) Isolated microspore culture and its applications in plant breeding and genetics. In: Plant tissue culture: propagation, conservation and crop improvement. Springer, Singapore, pp 487–507. https://doi.org/10.1007/978-981-10-1917-3_21
Shariatpanahi ME, Bal U, Heberle-Bors E, Touraev A (2006) Stresses applied for the re-programming of plant microspores towards in vitro embryogenesis. Physiol Plant 127(4):519–534. https://doi.org/10.1111/j.1399-3054.2006.00675.x
Shen Y, Pan G, Lübberstedt T (2015) Haploid strategies for functional validation of plant genes. Trends Biotechnol 33(10):611–620. https://doi.org/10.1016/j.tibtech.2015.07.005
Shtereva LA, Zagorska NA, Dimitrov BD, Kruleva MM, Oanh HK (1998) Induced androgenesis in tomato (Lycopersicon esculentum Mill). II. Factors affecting induction of androgenesis. Plant Cell Rep 18(3–4):312–317. https://doi.org/10.1007/s002990050578
Sood S, Dwivedi S (2015) Doubled haploid platform: an accelerated breeding approach for crop improvement. In: Bahadur B, Venkat Rajam M, Sahijram L, Krishnamurthy K (eds) Plant biology and biotechnology, Volume II: plant genomics and biotechnology. Springer, New Delhi, pp 89–111. https://doi.org/10.1007/978-81-322-2283-5_5
Summers WL, Jaramillo J, Bailey T (1992) Microspore developmental stage and anther length influence the induction of tomato anther callus. Hort Sci 27(7):838–840. https://doi.org/10.21273/HORTSCI.27.7.838
Thomas P, Mythili JB (1995) Development of cultured tomato anther to a fruit-like structure accompanied by in vitro ripening. Curr Sci 69(2):94–95
Tufail M, Ormsbee L, Teegavarapu R (2008) Artificial intelligence-based inductive models for prediction and classification of fecal coliform in surface waters. J Environ Eng 134(9):789–799. https://doi.org/10.1061/(ASCE)0733-9372(2008)134:9(789)
Wang GF, Qin HY, Sun D, Fan ST, Yang YM, Wang ZX, Xu PL, Zhao Y, Liu YX, Ai J (2018) Haploid plant regeneration from hardy kiwifruit (Actinidia arguta Planch.) anther culture. Plant Cell Tissue Organ Cult 134:1–14. https://doi.org/10.1007/s11240-018-1396-7
Wu J, Zhang X, Nie Y, Jin S, Liang S (2004) Factors affecting somatic embryogenesis and plant regeneration from a range of recalcitrant genotypes of Chinese cottons (Gossypium hirsutum L.). In Vitro Cell Dev Biol Plant 40:371–375. https://doi.org/10.1079/IVP2004535
Zagorska NA, Shtereva A, Dimitrov BD, Kruleva MM (1998) Induced androgenesis in tomato (Lycopersicon esculentum Mill.). II. Influence of genotype on androgenetic ability. Plant Cell Rep 17(12):968–973. https://doi.org/10.1007/s002990050519
Zheng MY, Konzak CF (1999) Effect of 2,4-dichlorophenoxyacetic acid on callus induction and plant regeneration in anther culture of wheat (Triticum aestivum L.). Plant Cell Rep 19(1):69–73. https://doi.org/10.1007/s002990050712
Author information
Authors and Affiliations
Contributions
M.N. conceived the idea, performed the anther culture and laboratory works along with ANN and IP analyses, and wrote the manuscript; M.E.S contributed in the idea and corrected the manuscript; M.A. contributed in ANN, IP, and other statistical analyses; and M.O. conducted the DAPI experiments.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest to disclose.
Additional information
Handling Editor: Peter Nick
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Niazian, M., Shariatpanahi, M.E., Abdipour, M. et al. Modeling callus induction and regeneration in an anther culture of tomato (Lycopersicon esculentum L.) using image processing and artificial neural network method. Protoplasma 256, 1317–1332 (2019). https://doi.org/10.1007/s00709-019-01379-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-019-01379-x