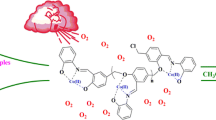
Abstract
A new polymer-supported Cu(II) Schiff base complex has been synthesized and characterized by elemental (including metal) analysis, FT-IR spectroscopy, UV–Vis diffuse reflectance spectroscopy, thermogravimetric analysis, and scanning electron microscopy. The catalytic performance of this complex was evaluated in the epoxidation of styrene in acetonitrile/N,N-dimethylformamide (9:1) mixture with 70% tert-butyl hydroperoxide as an oxidizing agent under liquid phase reaction conditions for selective synthesis of styrene oxide. Suitable reaction conditions have been optimized by considering the effects of various reaction parameters such as temperature, reaction time, solvent, oxidant, catalyst amount, and styrene to hydroperoxide molar ratio for the maximum conversion of styrene as well as selectivity of styrene oxide. We have also investigated the epoxidation reaction of various olefins under the optimized reaction conditions. Comparison between catalytic activities of the polymer-supported Cu(II) Schiff base complex and its homogeneous analogue showed that the polymer-supported catalyst was more active. This heterogeneous complex was reused for five times. The selectivity of the heterogeneous catalyst does not change even after five times of reusing.
Graphical Abstract







Similar content being viewed by others
References
McGarrigle EM, Gilheany DG (2005) Chem Rev 105:1563
Blaser HU, Pugin B, Spindler F (2005) J Mol Catal A Chem 231:1
Shulpin GB (2002) J Mol Catal A: Chem 189:39
Backvall JE (2004) Modern oxidation methods. Wiley-VCH, Weinheim, p 21
Haines AH (1985) Methods for the oxidation of organic compounds. Academic Press, New York
Xia QH, Ge HQ, Ye CP, Liu ZM, Su KX (2005) Chem Rev 105:1603
Yudin AK (2006) Aziridines and epoxides in organic synthesis. Wiley-VCH, Weinheim, p 185
Swern D (ed) (1971) Organic Peroxide, vol 2. Wiley-Interscience, New York, p 265
Barf GA, Sheldon RA (1995) J Mol Catal A: Chem 102:23
Mukaiyama T, Yamada T (1995) Bull Chem Soc Jpn 68:17
Bregeault JM (2003) Dalton Trans 17:3289
Herrmann WA, Fischer RW, Marz DW (1991) Angew Chem Int Ed 30:1638
Maschmeyer T, Rey F, Sankar G (1995) Nature 378:159
Behrens CH, Sharpless KB (1983) Aldrichimica Acta 16:67
Mohajer D, Tangestaninejad S (1993) J Chem Soc, Chem Commun 3:240
Calvante RM, O’Shea VAP, Campos-Martin JM, Fierro JLG, Gutiérrez-Puebla E (2004) J Mol Catal A: Chem 214:269
Booth RJ, Hodges JC (1999) Acc Chem Res 32:18
Gupta KC, Sutar AK (2008) React Funct Polym 68:12
Miller MM, Sherrington DC, Simpson S (1994) J Chem Soc Perkin Trans 2:2091
Valodkar VB, Tembe GL, Ram RN, Rama HS (2003) Catal Lett 90:91
Kotov SV, Balbolov E (2001) J Mol Catal A: Chem 176:41
Arnold U, Fan F, Habicht W, Döring M (2007) J Catal 245:55
Jia M, Seifert A, Thiel WR (2004) J Catal 221:319
Bakala PC, Briot E, Salles L, Brégeault JM (2006) Appl Catal A: Gen 300:91
Karandikar P, Chandwadkar AJ, Agashe M, Ramgir NS, Sivasanker S (2006) Appl Catal A: Gen 297:220
Handzlik J, Ogonowski J, Stoch J, Mikołajczyk M (2005) Catal Lett 101:65
Gigante B, Corma A, Garcia H, Sabater MJ (2000) Catal Lett 68:113
Tangestaninejad S, Habibi MH, Mirkhani V, Moghadam M, Grivani G (2006) Inorg Chem Comm 9:575
Mbeleck R, Ambroziak K, Saha B, Sherrington DC (2007) React Funct Polym 67:1448
Sureshan CA, Bhattacharya PK (1998) J Mol Catal A: Chem 136:285
Nam W, Kim HJ, Kim SH, Ho RYN, Valentine JS (1996) Inorg Chem 35:1045
Pietikäinen P (1999) Tetrahedron Lett 40:1001
Fraile JM, Garcia JI, Mayoral JA, Vispe E (2000) J Catal 189:40
Maurya MR, Chandrakar AK, Chand S (2007) J Mol Catal A: Chem 270:225
Karandikara P, Dhany KC, Deshpande S, Chandwadkar AJ, Sivasanker S, Agashe M (2004) Cat Comm 5:69
Srinivas D, Sivasanker S (2003) Catal Surv Asia 7:121
Sui Y, Zenga X, Fang X, Fub X, Xiao Y, Chend L, Li M, Chenga S (2007) J Mol Catal A: Chem 270:61
Angelino MD, Laibinis PE (1998) Macromolecules 31:7581
Ferraro JR (1971) Low Frequency Vibration of Inorganic and Coordination Compounds. Plenum Press, New York
Sallam SA, Orabi AS (2002) Transition Met Chem 27:447
Kandil SS, El-Hefnawy GB (2003) Transition Met Chem 28:168
Solomon EI, Sundaram UM, Machonkin TE (1996) Chem Rev 96:2563
Burk PL, Osborn JA, Youinou MT, Agnus Y, Louis R, Weiss R (1981) J Am Chem Soc 103:1273
Lever ABP (1984) Inorganic electronic spectroscopy, 2nd edn. Elsevier, Amsterdam
Salavati-Niasari M (2004) J Mol Catal A: Chem 217:87
Yonemitsu M, Tanaka Y, Iwamoto M (1998) J Catal 178:207
Arends IWCE, Sheldon RA (2002) Topics Catal 19:133
Pillai UR, Sahle-Demessie E (2002) Chem Commun 18:2142
Karandikar P, Agashe M, Vijayamohanan K, Chandwadkar A (2004) Appl Catal A: Gen 257:133
Brown K, Zolezzi S, Aguirre P, Venegas-Yazigi D, Paredes-Garcia V, Baggio R, Novak MA, Spodine E (2009) Dalton Trans 8:1422–1427
Mijanuddin M, Jana AD, Drew MGB, Hong CS, Chattopadhyay B, Mukherjee M, Nandi M, Bhaumik A, Helliwell M, Mostafa G, Ali M (2009) Polyhedron 28:665
Roy P, Dhara K, Manassero M, Banerjee P (2008) Inorg Chem Comm 11:265
Seelan S, Sinha AK, Srinivas D, Srivasankar S (2000) J Mol Catal A: Chem 157:163
Vogel AI (1989) Textbook of practical organic chemistry (quantitative analysis), 5th edn. Longman, London
Frechet JM, Schuerch C (1971) J Am Chem Soc 93:492
Acknowledgments
S.M.I. acknowledges the Department of Science and Technology (DST), Council of Scientific and Industrial Research (CSIR), and University Grant Commission (UGC), New Delhi, India for funding.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Islam, S.M., Roy, A.S., Mondal, P. et al. Olefin epoxidation with tert-butyl hydroperoxide catalyzed by functionalized polymer-supported copper(II) Schiff base complex. Monatsh Chem 143, 815–823 (2012). https://doi.org/10.1007/s00706-011-0641-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-011-0641-0